Advanced Iodine-Mediated Synthesis of Chiral Pyrazole Spirofuran Compounds for Commercial API Production
Advanced Iodine-Mediated Synthesis of Chiral Pyrazole Spirofuran Compounds for Commercial API Production
The pharmaceutical industry continuously seeks robust and scalable methodologies for constructing complex chiral scaffolds, particularly those found in bioactive natural products and clinical drugs. Patent CN109020987B introduces a groundbreaking iodine-mediated preparation method for chiral pyrazole spirofuran compounds, addressing critical challenges in asymmetric synthesis. These spirocyclic structures are pivotal in medicinal chemistry, serving as core skeletons for phosphodiesterase PDE4 inhibitors used in treating heart failure and asthma, as well as potent antimicrobial agents. The disclosed technology leverages a dual-catalytic system involving chiral squaric acid catalysts and iodine sources to achieve high yields and exceptional stereoselectivity under mild conditions. For a reliable pharmaceutical intermediate supplier, mastering such transformations is essential to meet the stringent purity and regulatory demands of global drug developers. This report analyzes the technical merits and commercial implications of this novel synthetic route, highlighting its potential to streamline the manufacturing of high-value heterocyclic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing chiral spirocyclic frameworks often rely on harsh reaction conditions, stoichiometric amounts of toxic reagents, or expensive transition metal catalysts that complicate downstream processing. Many existing methods suffer from poor atom economy, generating significant quantities of inorganic salt waste that increases post-treatment costs and environmental burden. Furthermore, achieving high enantioselectivity in spirocyclization reactions frequently requires cryogenic temperatures or prolonged reaction times, which are energetically inefficient and difficult to scale up for industrial production. The presence of heavy metal residues in the final product is a major concern for pharmaceutical applications, necessitating additional purification steps that reduce overall yield and increase lead time. These limitations underscore the urgent need for greener, more efficient catalytic systems that can deliver high-purity chiral intermediates without compromising on scalability or cost-effectiveness.
The Novel Approach
The invention described in CN109020987B overcomes these hurdles by employing an iodine-mediated strategy coupled with organocatalysis. As illustrated in the reaction scheme below, the process begins with a Michael addition between an o-hydroxy nitroolefin and a pyrazolone, catalyzed by a chiral squaric acid derivative. This is followed by an iodine-promoted cyclization step that efficiently closes the spiro ring system. 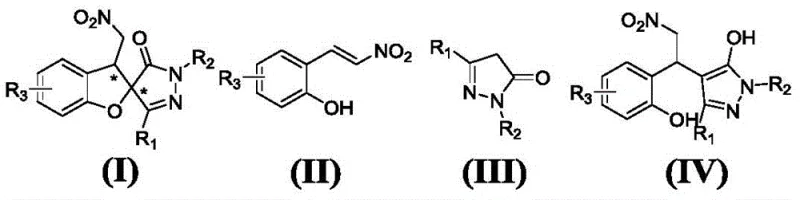 This tandem approach operates under remarkably mild conditions, typically ranging from -60°C to 60°C, and utilizes readily available iodine sources such as potassium iodide or elemental iodine. The elimination of transition metals not only reduces raw material costs but also simplifies the purification workflow, making it highly attractive for cost reduction in API manufacturing. The versatility of this method allows for the introduction of diverse functional groups, enabling the rapid generation of compound libraries for drug discovery programs while maintaining excellent diastereo- and enantioselectivity.
This tandem approach operates under remarkably mild conditions, typically ranging from -60°C to 60°C, and utilizes readily available iodine sources such as potassium iodide or elemental iodine. The elimination of transition metals not only reduces raw material costs but also simplifies the purification workflow, making it highly attractive for cost reduction in API manufacturing. The versatility of this method allows for the introduction of diverse functional groups, enabling the rapid generation of compound libraries for drug discovery programs while maintaining excellent diastereo- and enantioselectivity.
Mechanistic Insights into Squaric Acid-Catalyzed Asymmetric Cyclization
The success of this synthetic route hinges on the precise interplay between the chiral squaric acid catalyst and the iodine mediator. The chiral squaric acid catalysts, derived from cinchona alkaloids, function by activating the nitroolefin substrate through hydrogen bonding interactions, thereby facilitating the initial asymmetric Michael addition with high stereocontrol. 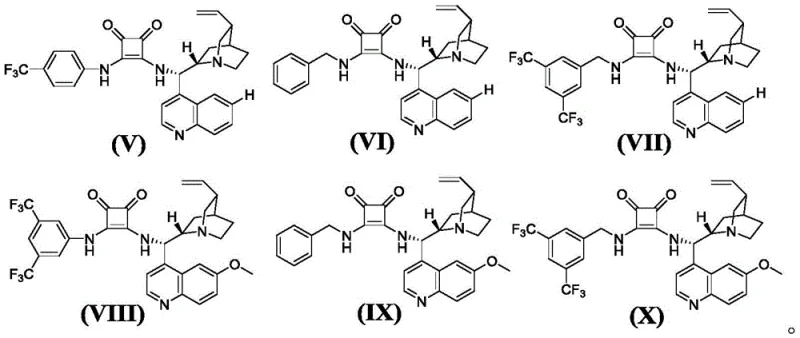 The specific spatial arrangement of the catalyst creates a chiral environment that directs the approach of the nucleophile, ensuring the formation of the desired enantiomer. Following the Michael addition, the introduction of the iodine source triggers an intramolecular cyclization. The iodine species likely acts as a Lewis acid or an electrophilic activator, promoting the attack of the phenolic hydroxyl group onto the activated intermediate to form the spiro-furan ring. This mechanism avoids the use of strong bases or acids that could lead to racemization or decomposition of sensitive functional groups. Understanding these mechanistic nuances is crucial for optimizing reaction parameters such as solvent choice, temperature, and stoichiometry to maximize yield and optical purity.
The specific spatial arrangement of the catalyst creates a chiral environment that directs the approach of the nucleophile, ensuring the formation of the desired enantiomer. Following the Michael addition, the introduction of the iodine source triggers an intramolecular cyclization. The iodine species likely acts as a Lewis acid or an electrophilic activator, promoting the attack of the phenolic hydroxyl group onto the activated intermediate to form the spiro-furan ring. This mechanism avoids the use of strong bases or acids that could lead to racemization or decomposition of sensitive functional groups. Understanding these mechanistic nuances is crucial for optimizing reaction parameters such as solvent choice, temperature, and stoichiometry to maximize yield and optical purity.
Impurity control is another critical aspect where this mechanism excels. The high selectivity of the squaric acid catalyst minimizes the formation of diastereomeric byproducts, which are often difficult to separate. Additionally, the mild nature of the iodine-mediated cyclization prevents side reactions such as polymerization or over-oxidation that are common in harsher oxidative cyclization protocols. The result is a cleaner reaction profile, which translates directly to reduced purification costs and higher throughput in a commercial setting. For R&D teams focused on process development, this level of control over the impurity profile is invaluable for meeting strict regulatory specifications for pharmaceutical intermediates.
How to Synthesize Chiral Pyrazole Spirofuran Efficiently
Implementing this synthesis requires careful attention to the sequential addition of reagents and the selection of appropriate solvents. The process is divided into two distinct stages: the initial organocatalytic Michael addition and the subsequent iodine-mediated cyclization. Solvents such as dichloromethane, toluene, or ethyl acetate are commonly employed, with the choice depending on the solubility of the specific substrates and the desired reaction rate. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are outlined in the guide below to ensure reproducibility and safety.
- Mix o-hydroxy nitroolefin, pyrazolone, and chiral squaric acid catalyst in organic solvent at -60 to 60°C for 1-48 hours to form the intermediate.
- Remove solvent and add iodine source and base to the intermediate mixture in fresh organic solvent.
- React at -60 to 60°C for 1-48 hours, then perform extraction and column chromatography to isolate the pure chiral spiro compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this iodine-mediated synthesis offers substantial strategic benefits. The shift from metal-catalyzed to organocatalytic processes significantly de-risks the supply chain by removing dependence on volatile precious metal markets and complex recycling protocols. This transition aligns perfectly with the industry's move towards sustainable and green chemistry practices, enhancing the long-term viability of the manufacturing process.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the associated removal steps leads to direct cost savings. Furthermore, the use of inexpensive iodine sources and common organic solvents reduces raw material expenditure. The high yields reported in the patent examples mean less starting material is wasted, improving the overall material efficiency and lowering the cost per kilogram of the final intermediate.
- Enhanced Supply Chain Reliability: The reagents required for this synthesis, including nitroolefins, pyrazolones, and squaric acid catalysts, are commercially available and stable, ensuring a consistent supply. The robustness of the reaction conditions allows for flexible scheduling and reduces the risk of batch failures due to sensitive operating parameters. This reliability is crucial for maintaining continuous production schedules and meeting tight delivery deadlines for downstream API manufacturers.
- Scalability and Environmental Compliance: The mild reaction temperatures and absence of hazardous heavy metals simplify the scale-up process from laboratory to pilot and commercial plant scales. Waste streams are easier to treat and dispose of, reducing environmental compliance costs. The atom economy of the reaction is superior to many traditional methods, minimizing the generation of chemical waste and supporting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These insights are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for process chemists and project managers.
Q: What are the key advantages of the iodine-mediated method for spiro compounds?
A: The method offers mild reaction conditions, high product yields, and excellent stereoselectivity without requiring expensive transition metal catalysts, simplifying downstream purification.
Q: Which catalysts are preferred for this asymmetric synthesis?
A: Chiral squaric acid catalysts derived from cinchona alkaloids, specifically compounds represented by formulas (V) to (X), with formula (VII) being particularly effective.
Q: What is the scope of substituents for the pyrazole spirofuran products?
A: The method tolerates a wide range of substituents including alkyl, phenyl, halogenated phenyl, and methoxy groups on both the nitroolefin and pyrazolone starting materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Pyrazole Spirofuran Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, leveraging advanced technologies like the iodine-mediated spirocyclization to deliver high-quality intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to optimize your supply chain and reduce costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis for your specific project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our expertise can accelerate your drug development timeline and enhance your competitive edge in the global market.
