Advanced Manufacturing of High-Purity Guaiacol Glycidyl Ether for Pharmaceutical Intermediates
The pharmaceutical industry continuously demands intermediates with exceptional purity profiles to ensure the safety and efficacy of final Active Pharmaceutical Ingredients (APIs). Patent CN101838249A introduces a groundbreaking methodology for the preparation of high-purity guaiacol glycidyl ether, a critical building block for cardiovascular medications such as Ranolazine and Moprolol. This technical disclosure addresses long-standing challenges in etherification and epoxidation processes by replacing energy-intensive distillation with a sophisticated recrystallization protocol. By achieving gas chromatography (GC) purity levels exceeding 99.5 percent, this innovation sets a new benchmark for quality in the synthesis of epoxy-based pharmaceutical intermediates. The process not only enhances product specifications but also streamlines the operational workflow, making it highly attractive for large-scale industrial adoption by forward-thinking chemical manufacturers.
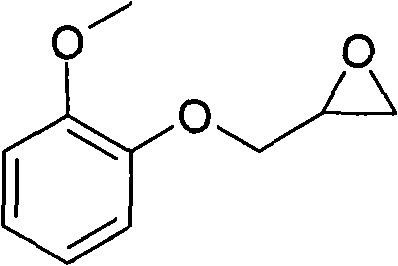
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of guaiacol glycidyl ether has been plagued by significant technical and environmental drawbacks inherent to traditional purification techniques. Prior art, such as that described in patent GB2216520, relies heavily on the use of methylene chloride for extraction followed by high-vacuum fractionation. This approach necessitates the consumption of vast quantities of chlorinated solvents, posing severe environmental hazards and increasing waste disposal costs substantially. Furthermore, the requirement for high-temperature vacuum distillation places extreme stress on production equipment and often induces thermal polymerization of the sensitive epoxy product. Other methods, like those utilizing toluene as a solvent referenced in WO2008047388, suffer from emulsification issues during aqueous washing due to density differences, leading to difficult phase separations and product losses. These conventional routes typically struggle to surpass 96 percent purity, leaving residual impurities that can complicate downstream drug synthesis and regulatory approval processes.
The Novel Approach
The methodology outlined in CN101838249A represents a paradigm shift by eliminating the need for extraneous organic solvents and harsh thermal purification steps. Instead of relying on distillation, this novel approach leverages the physical property differences between the solid product and liquid impurities through a targeted recrystallization strategy. By utilizing excess epichlorohydrin as both a reactant and the reaction medium, the process inherently reduces solvent inventory and simplifies the recovery of unreacted materials. The introduction of a crystallization step using C1-C4 lower alcohols, such as ethanol or methanol, allows for the precise exclusion of liquid by-products at ambient or slightly elevated temperatures. This gentle purification technique prevents thermal degradation and polymerization, ensuring that the final crystalline product maintains structural integrity while achieving purity levels greater than 99.5 percent. The result is a robust, scalable process that significantly lowers the barrier to entry for high-quality intermediate manufacturing.
Mechanistic Insights into Base-Catalyzed Condensation and Crystallization
The core chemical transformation involves a two-stage reaction mechanism initiated by the nucleophilic attack of the phenolic hydroxyl group of guaiacol on the epichlorohydrin molecule. Under the influence of an inorganic base, such as sodium hydroxide or potassium hydroxide, and facilitated by a phase-transfer catalyst like tetrabutylammonium bromide, the phenoxide ion attacks the less hindered carbon of the epichlorohydrin epoxide ring or displaces the chloride depending on the specific pathway conditions, eventually forming the glycidyl ether linkage. The reaction is carefully controlled within a temperature range of 30 to 60 degrees Celsius to balance reaction kinetics with selectivity, preventing the homopolymerization of epichlorohydrin. Following the condensation, the reaction mixture undergoes a ring-closing or stabilization phase where the epoxy group is preserved. The use of a biphasic system with aqueous alkali ensures that inorganic salts and water-soluble by-products are effectively partitioned away from the organic phase containing the desired intermediate.
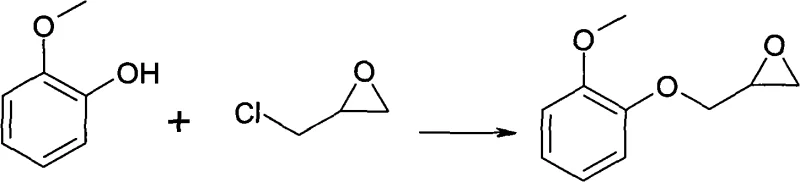
Impurity control is fundamentally achieved through the thermodynamic principles of crystallization rather than kinetic separation via distillation. Since the target guaiacol glycidyl ether exists as a solid at room temperature while the major impurities (unreacted guaiacol, oligomers, and chlorohydrin derivatives) remain liquid, cooling the solution in a selective alcohol solvent induces the nucleation of pure crystals. The addition of crystal seeds further governs the growth rate, ensuring that impurities are excluded from the crystal lattice and remain in the mother liquor. This mechanism is far superior to distillation for heat-sensitive epoxides, as it operates at significantly lower temperatures, thereby preserving the delicate oxirane ring structure. The choice of solvent polarity is critical; lower alcohols provide the optimal solubility gradient where the product is soluble when hot but precipitates efficiently upon cooling, dragging down the overall impurity profile to negligible levels suitable for stringent pharmaceutical applications.
How to Synthesize Guaiacol Glycidyl Ether Efficiently
The synthesis protocol detailed in the patent offers a clear roadmap for transitioning from laboratory benchtop experiments to pilot plant operations with minimal friction. The process begins with the dissolution of guaiacol in a molar excess of epichlorohydrin, creating a homogeneous reaction environment that drives the equilibrium toward product formation. Subsequent addition of the aqueous base and catalyst initiates the exothermic condensation, which must be managed through controlled dosing to maintain the optimal thermal window. Once the reaction reaches completion, the workup involves a straightforward phase separation and the recovery of the excess epichlorohydrin via reduced pressure distillation, which can be recycled for subsequent batches. The resulting crude material is then subjected to the critical recrystallization step, where solvent volume and cooling rates are tuned to maximize yield and crystal quality. For a comprehensive understanding of the specific operational parameters, including exact stoichiometric ratios and temperature ramps, please refer to the standardized synthesis guide below.
- Condense guaiacol with excess epichlorohydrin using an inorganic base and phase-transfer catalyst at 30-60°C.
- Separate layers and recover excess epichlorohydrin via reduced pressure distillation to obtain the crude product.
- Recrystallize the crude product using a C1-C4 lower alcohol solvent (e.g., ethanol or methanol) to achieve >99.5% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology translates directly into enhanced operational efficiency and risk mitigation. By removing the dependency on chlorinated solvents like methylene chloride, manufacturers can significantly reduce their environmental compliance burden and associated waste treatment costs. The elimination of high-vacuum distillation equipment lowers capital expenditure requirements and reduces maintenance downtime, as the process relies on standard crystallization vessels that are easier to operate and clean. Furthermore, the ability to recycle excess epichlorohydrin directly within the process loop minimizes raw material consumption, driving down the variable cost per kilogram of the final intermediate. These factors combine to create a more resilient supply chain capable of meeting the rigorous quality standards of global pharmaceutical clients without the volatility associated with complex solvent management.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the simplification of the unit operations and the reduction in solvent procurement. By utilizing the reactant itself as the solvent, the need for purchasing, storing, and disposing of separate organic solvents is drastically curtailed. Additionally, the avoidance of high-energy distillation steps reduces utility consumption, specifically steam and cooling water, leading to a leaner manufacturing cost structure. The high yield and purity achieved through recrystallization also minimize the loss of valuable starting materials, ensuring that a greater proportion of input costs are converted into saleable high-value product.
- Enhanced Supply Chain Reliability: From a logistics perspective, the robustness of this synthetic route ensures consistent batch-to-batch quality, which is paramount for maintaining uninterrupted API production schedules. The use of common, commercially available reagents such as sodium hydroxide and ethanol reduces the risk of supply disruptions associated with specialty chemicals. Moreover, the simplified purification process shortens the overall cycle time per batch, allowing for increased throughput and faster response to market demand fluctuations. This reliability makes the manufacturer a more dependable partner for long-term supply agreements with major pharmaceutical companies.
- Scalability and Environmental Compliance: The transition from laboratory to commercial scale is seamless due to the reliance on standard crystallization technology rather than specialized high-vacuum distillation columns. This scalability ensures that production volumes can be ramped up to meet clinical trial demands or commercial launch requirements without significant process re-engineering. Environmentally, the reduction in volatile organic compound (VOC) emissions and hazardous waste generation aligns with modern green chemistry principles and increasingly strict global environmental regulations. This compliance future-proofs the manufacturing site against tightening regulatory frameworks, securing long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this high-purity synthesis method. These insights are derived directly from the comparative data and experimental examples provided in the patent documentation, offering clarity on how this approach outperforms legacy technologies. Understanding these nuances is essential for technical teams evaluating process transfers or seeking to optimize their current intermediate sourcing strategies. The answers highlight the specific advantages in purity, yield, and operational simplicity that define this innovative manufacturing route.
Q: Why is recrystallization preferred over high-vacuum distillation for this intermediate?
A: High-temperature distillation often leads to product polymerization and requires harsh equipment conditions. Recrystallization using lower alcohols avoids thermal degradation, ensuring higher purity (>99.5%) and better yield without complex vacuum systems.
Q: How does this method reduce environmental impact compared to conventional routes?
A: Conventional methods utilize large volumes of chlorinated solvents like methylene chloride or toluene. This novel approach uses excess epichlorohydrin as both reactant and solvent, which is recovered and reused, significantly minimizing hazardous waste and solvent consumption.
Q: What represents the critical quality attribute for this API intermediate?
A: The critical quality attribute is the GC purity, which must exceed 99.5% to ensure the safety and efficacy of downstream drugs like Ranolazine. The patented crystallization step effectively removes liquid impurities that remain after the initial condensation reaction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Guaiacol Glycidyl Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of your final drug product is only as good as the intermediates you source. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent concept to industrial reality is seamless. We are committed to delivering high-purity guaiacol glycidyl ether that meets stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our facility is designed to handle complex chemistries with the utmost care, guaranteeing supply continuity and consistency for your critical pharmaceutical projects.
We invite you to collaborate with us to leverage this advanced manufacturing technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized process can enhance your project's economics and timeline. Let us be your partner in delivering excellence in pharmaceutical intermediate manufacturing.
