Advanced Synthesis of Perhaloalkyl Thioethers: A Cost-Effective Route for High-Purity Agrochemical Intermediates
Advanced Synthesis of Perhaloalkyl Thioethers: A Cost-Effective Route for High-Purity Agrochemical Intermediates
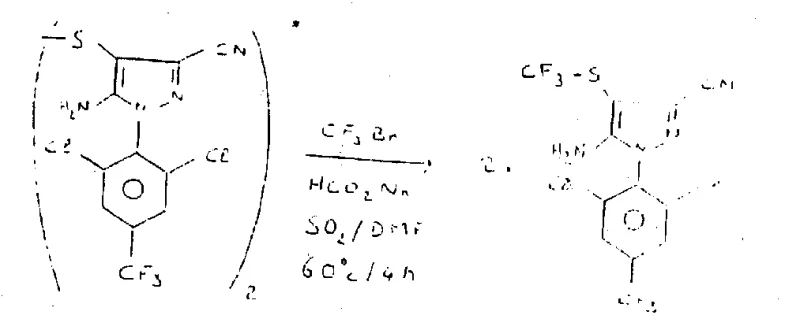
The landscape of fine chemical manufacturing is constantly evolving, driven by the need for more economical and safer synthetic routes for complex intermediates. Patent CN1043499A introduces a groundbreaking methodology for the preparation of whole haloalkyl thioethers, specifically addressing the longstanding challenges associated with introducing perfluoroalkyl groups into organic molecules. This technology represents a paradigm shift from traditional multi-step syntheses that rely on expensive and hazardous reagents, offering a direct, one-pot solution that utilizes industrially abundant perfluoroalkyl bromides. For R&D directors and procurement specialists in the agrochemical and pharmaceutical sectors, this innovation provides a critical pathway to reduce raw material costs while enhancing the purity profile of key intermediates such as pyrazole derivatives.
The core of this invention lies in the strategic use of a reducing agent system, comprising metals like zinc or sulfur-based reductants such as hyposulfites, in the presence of sulfur dioxide. This combination effectively activates the relatively inert carbon-bromine bond in perfluoroalkyl bromides, allowing them to react directly with disulfides. Unlike prior art which often necessitated the use of perfluoroalkyl iodides due to their higher reactivity, this method unlocks the potential of cheaper bromides without sacrificing yield. The implications for commercial scale-up are profound, as it replaces scarce iodine-based feedstocks with widely available bromine-based gases, thereby stabilizing the supply chain against volatile commodity pricing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of perfluoroalkyl thioethers has been plagued by significant economic and safety hurdles that hinder efficient commercial production. Conventional pathways frequently relied on the chlorination of methyl thio derivatives followed by fluorination, a tedious multi-step process that generated substantial waste and required harsh reaction conditions. Alternatively, methods utilizing trifluoromethanesulfenyl chloride (CF3SCl) or organo-manganese compounds introduced severe safety risks due to the toxicity and instability of these reagents. Furthermore, the reliance on perfluoroalkyl iodides (such as CF3I) as the fluorinated source imposed a heavy financial burden, as iodides are exponentially more expensive than their bromide counterparts and often subject to supply constraints. These legacy methods also struggled with selectivity, often producing mixtures of mono- and di-substituted products that required complex purification protocols, ultimately driving up the cost of goods sold for the final active pharmaceutical or agrochemical ingredient.
The Novel Approach
The methodology disclosed in the patent offers a transformative solution by enabling the direct reaction of disulfides with perfluoroalkyl bromides under reductive conditions. This approach eliminates the need for pre-functionalized, hazardous intermediates like CF3SCl and bypasses the expensive iodide route entirely. By employing a robust reducing system—typically zinc dust with sulfur dioxide or alkali metal hyposulfites—the process generates the reactive perfluoroalkyl radical or anion in situ, which immediately couples with the disulfide bond. This results in a streamlined, atom-economical transformation that can be performed in common polar aprotic solvents like dimethylformamide (DMF). The versatility of this method is demonstrated by its ability to accommodate a wide range of substrates, from simple phenyl disulfides to complex heterocyclic systems, delivering high-purity thioethers with minimal byproduct formation.
Mechanistic Insights into Reductive Trifluoromethylation
The mechanistic underpinning of this synthesis involves the generation of a nucleophilic or radical perfluoroalkyl species through single-electron transfer (SET) processes facilitated by the metal/reductant system. When zinc or hyposulfite interacts with sulfur dioxide in a polar medium, it creates a highly reducing environment capable of cleaving the carbon-halogen bond of the perfluoroalkyl bromide. This activation step is critical, as the C-Br bond in compounds like CF3Br is stronger and less reactive than the C-I bond found in traditional reagents. The resulting trifluoromethyl radical or anion then attacks the disulfide linkage, leading to the scission of the S-S bond and the formation of the new C-S bond. This mechanism is particularly advantageous because it proceeds under mild thermal conditions, typically between 20°C and 100°C, minimizing thermal degradation of sensitive functional groups often present in advanced intermediates.
From an impurity control perspective, this mechanism offers superior selectivity compared to free-radical halogenation methods. The controlled generation of the perfluoroalkyl species ensures that the reaction is directed specifically towards the sulfur center, reducing the incidence of unwanted side reactions on the aromatic or heterocyclic rings. For instance, in the synthesis of pyrazole derivatives, the method preserves the integrity of the amino and cyano groups, which are susceptible to degradation under harsher conditions. The use of hyposulfites or formates as co-reductants further buffers the reaction medium, preventing the accumulation of acidic byproducts that could catalyze decomposition. This inherent cleanliness of the reaction profile translates directly to simplified downstream processing, as the crude product often requires only basic extraction and crystallization to meet stringent purity specifications required for regulatory filings.
How to Synthesize Perhaloalkyl Thioethers Efficiently
The operational simplicity of this patented process makes it highly attractive for pilot plant and commercial manufacturing environments. The procedure generally involves charging a pressure-rated reactor with the disulfide substrate, a polar solvent such as DMF or DMSO, and the chosen reducing agent system. For gaseous reagents like trifluoromethyl bromide, the reactor is pressurized to levels between 1 to 50 atmospheres, ensuring sufficient concentration of the reagent in the liquid phase to drive the reaction to completion. The detailed standardized synthesis steps, including specific molar ratios and workup procedures optimized for different substrates, are outlined below to guide process development teams in replicating these high-yielding transformations.
- Prepare the reaction mixture by combining the disulfide substrate, a polar aprotic solvent such as DMF, and a reducing agent system comprising zinc dust or sodium hyposulfite.
- Introduce sulfur dioxide gas or a sulfinate salt to activate the reducing environment, ensuring the reactor is purged of oxygen to prevent side reactions.
- Pressurize the reactor with perfluoroalkyl bromide (e.g., CF3Br) and maintain the temperature between 20°C and 100°C until conversion is complete, followed by standard aqueous workup.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The primary driver of cost reduction is the substitution of raw materials; replacing perfluoroalkyl iodides with bromides results in a drastic decrease in feedstock expenses, as bromides are produced on a much larger industrial scale for refrigerant and fire suppression applications. Additionally, the elimination of hazardous reagents like trifluoromethanesulfenyl chloride reduces the costs associated with specialized handling, storage, and waste disposal compliance. The robustness of the reaction conditions also implies lower energy consumption, as the process does not require cryogenic temperatures or extended heating periods, further contributing to a leaner manufacturing cost structure.
- Cost Reduction in Manufacturing: The shift from iodide-based reagents to bromide-based reagents fundamentally alters the cost basis of the synthesis. Since perfluoroalkyl bromides are commoditized industrial gases, their price stability is far superior to that of specialized iodides. Furthermore, the one-pot nature of the reaction consolidates multiple synthetic steps into a single unit operation, reducing labor hours, solvent usage, and reactor occupancy time. This consolidation minimizes the cumulative yield losses typically seen in multi-step sequences, ensuring that more of the starting material is converted into saleable product, thereby maximizing the return on investment for every batch produced.
- Enhanced Supply Chain Reliability: Relying on niche reagents like CF3I creates vulnerability in the supply chain, as these materials are often sourced from limited suppliers with long lead times. In contrast, CF3Br and zinc dust are globally available commodities with established logistics networks. This abundance ensures that production schedules are not disrupted by raw material shortages. Moreover, the tolerance of the process to various reducing agents (zinc, hyposulfite, formate) provides operational flexibility; if one reagent faces temporary supply constraints, the protocol can be adjusted to utilize an alternative without compromising the reaction outcome, thus safeguarding continuous manufacturing operations.
- Scalability and Environmental Compliance: The environmental profile of this method aligns well with modern green chemistry initiatives. By avoiding chlorination steps and toxic sulfenyl chlorides, the process generates a cleaner waste stream that is easier to treat. The solvents used, such as DMF, are recoverable and recyclable, reducing the overall solvent footprint. From a scalability standpoint, the reaction kinetics are well-suited for large-scale pressure reactors, and the exothermic nature of the reaction is manageable with standard cooling systems. This ease of scale-up allows manufacturers to respond rapidly to market demand surges without the need for extensive process re-engineering or new capital equipment investments.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this perhaloalkyl thioether synthesis technology. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a realistic overview of what process development teams can expect when transitioning from laboratory scale to commercial production. Understanding these nuances is essential for accurate risk assessment and project planning.
Q: Why is CF3Br preferred over CF3I in this synthesis?
A: CF3Br is significantly more cost-effective and industrially available compared to CF3I. The patented method utilizes a specific reducing system (Zn/SO2 or Hyposulfite) that activates the stronger C-Br bond efficiently, eliminating the need for expensive iodides while maintaining high yields.
Q: What are the safety advantages of this new method?
A: Traditional methods often require hazardous intermediates like trifluoromethanesulfenyl chloride (CF3SCl) or harsh chlorination steps. This novel approach operates under milder conditions using stable disulfides and gaseous CF3Br, significantly reducing the handling of toxic and corrosive reagents.
Q: Can this process be scaled for commercial production?
A: Yes, the process is designed for industrial scalability. It utilizes common solvents like DMF and robust reducing agents like zinc or V-Brite B. The reaction tolerates pressures up to 50 atm, making it suitable for large-scale pressure reactors used in fine chemical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Perhaloalkyl Thioether Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes in the development of next-generation agrochemicals and pharmaceuticals. Our technical team has extensively analyzed the methodologies described in CN1043499A and possesses the expertise to implement this reductive trifluoromethylation technology effectively. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless. Our facilities are equipped with state-of-the-art pressure reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of perhaloalkyl thioether intermediate delivered meets the highest quality standards required by global regulatory bodies.
We invite you to collaborate with us to leverage this cost-effective technology for your specific project needs. Whether you require custom synthesis of complex pyrazole derivatives or bulk production of fluorinated intermediates, our team is ready to provide a Customized Cost-Saving Analysis tailored to your bill of materials. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can optimize your supply chain and reduce your overall production costs.
