Revolutionizing Alpha-Azaspiro Drug Template Production: A High-Yield Commercial Strategy
The pharmaceutical industry's relentless pursuit of novel bioactive scaffolds has placed alpha-azaspiro drug templates at the forefront of medicinal chemistry research. As detailed in patent CN101081851B, these unique spiro-fused nitrogen heterocycles serve as exceptional Type II beta-turn mimics, offering rigid conformational constraints that significantly enhance binding affinity and metabolic stability in peptide-based therapeutics. The structural diversity of these templates, ranging from [4.4] to [6.7] spiro systems, allows for the rapid generation of compound libraries via combinatorial chemistry, accelerating the discovery of potent drug candidates. However, the historical bottleneck in accessing these valuable intermediates has been the inefficiency of traditional synthetic routes, which often suffer from prohibitive costs and low throughput. This technical insight report analyzes the breakthrough methodology disclosed in the patent, providing a roadmap for R&D directors and procurement specialists to leverage this technology for cost-effective API intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
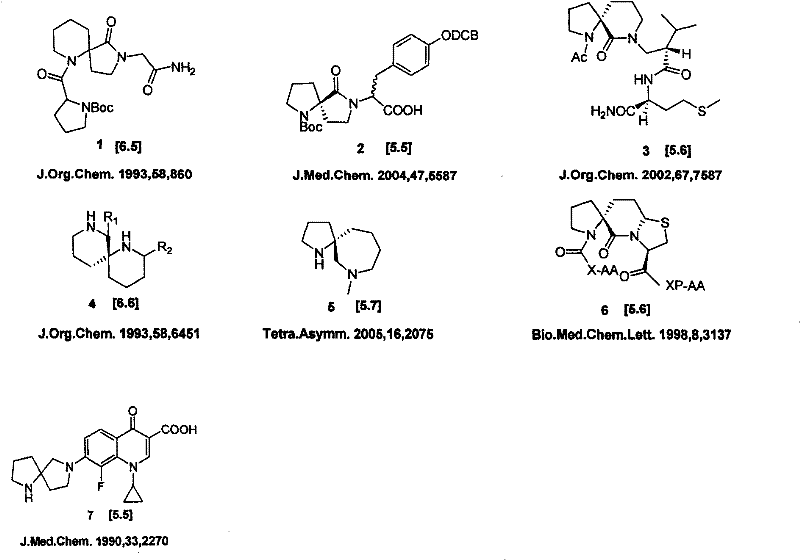
The Limitations of Conventional Methods
Prior to the innovations described in CN101081851B, the synthesis of alpha-azaspiro frameworks was plagued by convoluted multi-step sequences that severely impacted commercial viability. Literature precedents, such as those reported in J. Med. Chem. (1990, 33, 2270) and J. Org. Chem. (1993, 58, 860), typically required between 5 to 8 distinct reaction steps to construct the core spiro skeleton. These legacy routes often relied on inefficient ring-closing strategies, such as ester aminolysis or complex condensation reactions involving alcohol and amide functionalities, which inherently suffered from poor atom economy. Furthermore, the overall yields for these conventional processes were dismally low, frequently hovering between 4% and 21%, rendering them unsuitable for the mass preparation required in modern drug development pipelines. The reliance on expensive reagents and the generation of significant chemical waste further exacerbated the cost burden, creating a substantial barrier for procurement managers aiming to secure reliable supplies of these critical building blocks.
The Novel Approach
The patented methodology introduces a paradigm shift by streamlining the synthesis into a concise four-step sequence that dramatically improves efficiency and scalability. By strategically utilizing a nitrile intermediate as a pivot point for ring construction, the inventors have eliminated unnecessary functional group manipulations that characterized previous attempts. The core innovation lies in the direct conversion of protected proline or pipecolic acid derivatives into nitriles, followed by a highly selective alkylation and a final reductive cyclization. This approach not only reduces the step count but also boosts the overall yield to a commercially attractive range of 27% to 35%. The use of mild reaction conditions and readily available catalysts ensures that the process is robust and adaptable to large-scale reactors, addressing the critical need for a reliable alpha-azaspiro drug template supplier capable of meeting industrial demand without compromising on purity or delivery timelines.
Mechanistic Insights into Reductive Cyclization and Nitrile Alkylation
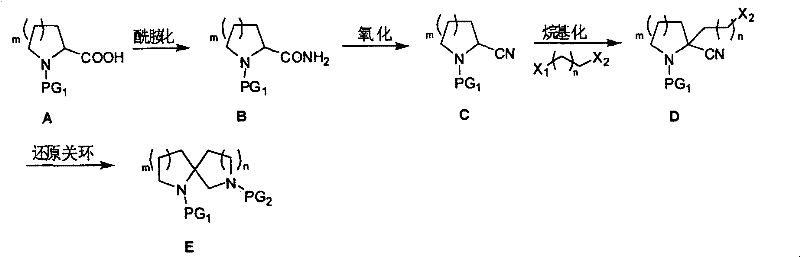
The mechanistic elegance of this synthesis rests on the precise control of reactivity at the alpha-position of the cyclic amino acid derivative. The process initiates with the protection of the nitrogen atom, typically using Boc or Cbz groups, followed by amidation to install the necessary nitrogen handle. The subsequent dehydration of this amide to a nitrile using trifluoroacetic anhydride (TFAA) is a critical transformation; it activates the alpha-carbon for deprotonation by strong bases like Lithium Diisopropylamide (LDA). This generates a stabilized carbanion that acts as a potent nucleophile, attacking dihaloalkanes to extend the carbon chain with a terminal halide. This alkylation step is pivotal as it pre-organizes the molecule for the final cyclization event, ensuring the correct ring size is formed based on the length of the dihaloalkane spacer used. The precision of this alkylation dictates the structural integrity of the final spiro system, minimizing the formation of regioisomeric impurities that often plague less controlled radical cyclization methods.
The final reductive cyclization represents the culmination of the synthetic strategy, where the nitrile group and the terminal halide are simultaneously transformed to close the second ring. Under hydrogenation conditions using catalysts such as Raney Nickel, Palladium on Carbon, or Platinum Dioxide in the presence of ammonia, the nitrile is reduced to an amine while the halide undergoes nucleophilic substitution. This tandem reduction-cyclization mechanism is highly efficient, driven by the thermodynamic stability of the newly formed spiro-fused ring system. The presence of ammonia in the reaction mixture is crucial as it suppresses intermolecular polymerization and ensures that the intramolecular attack prevails, leading to high selectivity for the desired spiro product. This mechanistic pathway avoids the use of hazardous hydride reagents or complex transition metal catalysts often seen in cross-coupling approaches, thereby simplifying the purification process and enhancing the safety profile of the manufacturing operation.
How to Synthesize Alpha-Azaspiro Drug Templates Efficiently
Implementing this synthesis requires strict adherence to the optimized reaction parameters outlined in the patent to ensure maximum yield and purity. The process begins with the preparation of the nitrile intermediate, which serves as the universal precursor for various spiro derivatives. Operators must maintain low temperatures during the LDA-mediated alkylation to prevent side reactions, followed by careful workup to isolate the alkylated nitrile. The final hydrogenation step is flexible regarding the catalyst choice, allowing facilities to utilize existing hydrogenation infrastructure with Raney Ni or Pd/C. For a detailed breakdown of the specific reagents, stoichiometry, and isolation procedures for each stage of this four-step sequence, please refer to the standardized protocol below.
- Amidation: Protect the amino group of proline or pipecolic acid derivatives and react with active esters and ammonia to form the amide intermediate.
- Oxidation: Dehydrate the amide using trifluoroacetic anhydride (TFAA) and triethylamine to generate the critical nitrile intermediate.
- Alkylation: Deprotonate the nitrile with LDA and react with dihaloalkanes to introduce the side chain required for ring closure.
- Reductive Cyclization: Perform catalytic hydrogenation using Raney Nickel or Pd/C in the presence of ammonia to close the spiro ring.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers tangible strategic benefits that extend beyond simple chemical transformation. The reduction in reaction steps from nearly eight down to four directly correlates with a significant decrease in processing time and resource consumption, effectively lowering the cost of goods sold (COGS). By eliminating the need for exotic reagents and complex purification stages associated with older methods, manufacturers can achieve substantial cost savings in raw material procurement and waste disposal. This efficiency gain is particularly vital in the current economic climate, where optimizing the supply chain for high-purity pharmaceutical intermediates is essential for maintaining competitive pricing structures without sacrificing quality standards.
- Cost Reduction in Manufacturing: The streamlined four-step process drastically reduces the operational overhead associated with multi-step synthesis. By consolidating the synthetic pathway, facilities can minimize solvent usage, energy consumption for heating and cooling cycles, and labor hours required for intermediate isolations. The avoidance of expensive transition metal catalysts, replacing them with cost-effective options like Raney Nickel, further drives down the direct material costs. Additionally, the higher overall yield means that less starting material is required to produce the same amount of final product, effectively amplifying the purchasing power of the procurement budget and reducing the financial risk associated with raw material price volatility.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures a more predictable and stable supply of alpha-azaspiro templates. Traditional methods with low yields and multiple steps are prone to bottlenecks, where a failure in any single step can halt the entire production batch. In contrast, this high-yielding process with mild conditions offers greater process reliability, reducing the likelihood of batch failures and ensuring consistent delivery schedules. The use of commodity chemicals and standard catalysts means that the supply chain is less vulnerable to disruptions caused by the scarcity of specialized reagents, providing a secure source of critical intermediates for long-term drug development projects.
- Scalability and Environmental Compliance: From a sustainability and scale-up perspective, this methodology is exceptionally well-suited for industrial expansion. The reaction conditions are mild, typically operating at ambient or moderately elevated temperatures and pressures, which reduces the engineering complexity required for scaling from kilogram to tonne quantities. Furthermore, the improved atom economy and reduced waste generation align with green chemistry principles, simplifying environmental compliance and waste treatment protocols. This facilitates the commercial scale-up of complex pharmaceutical intermediates, allowing manufacturers to rapidly respond to market demand while adhering to stringent environmental regulations and corporate sustainability goals.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is crucial for stakeholders evaluating its integration into their supply chain. The following questions address common inquiries regarding the versatility, scalability, and quality control aspects of the patented method. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What are the key advantages of this synthesis method over conventional routes?
A: This method reduces the reaction steps from 5-8 down to just 4, significantly improving overall yield from typically <21% to approximately 27-35%, while avoiding harsh reagents.
Q: Can this process be scaled for industrial production?
A: Yes, the process utilizes mild conditions and common catalysts like Raney Nickel, making it highly suitable for large-scale commercial manufacturing without specialized high-pressure equipment.
Q: What types of spiro structures can be generated using this template?
A: The method is versatile, capable of producing various ring sizes including [4.4], [4.5], [5.5], and [5.6] spiro systems by varying the starting amino acid and the dihaloalkane chain length.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Azaspiro Drug Template Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality spiro scaffolds play in the development of next-generation therapeutics. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the structural integrity and impurity profile of every batch. Our capability to implement the efficient four-step synthesis described in CN101081851B allows us to offer a competitive advantage in both lead time and cost structure for our global partners.
We invite you to collaborate with us to optimize your supply chain for these valuable intermediates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing expertise can accelerate your drug discovery timeline while maximizing value.
