Advanced Asymmetric Synthesis of Chiral Dihydrofuran Compounds for Pharmaceutical Applications
The pharmaceutical and agrochemical industries continuously seek robust methodologies for constructing complex heterocyclic scaffolds, particularly those exhibiting significant biological activity. Patent CN107216297B introduces a groundbreaking asymmetric synthesis method for chiral dihydrofuran compounds, addressing critical limitations in current manufacturing protocols. Dihydrofuran skeletons are ubiquitous in nature, serving as core structures in potent bioactive molecules such as Obtusafuran, which demonstrates strong cytotoxicity, and Conocarpan, known for its antibacterial properties. 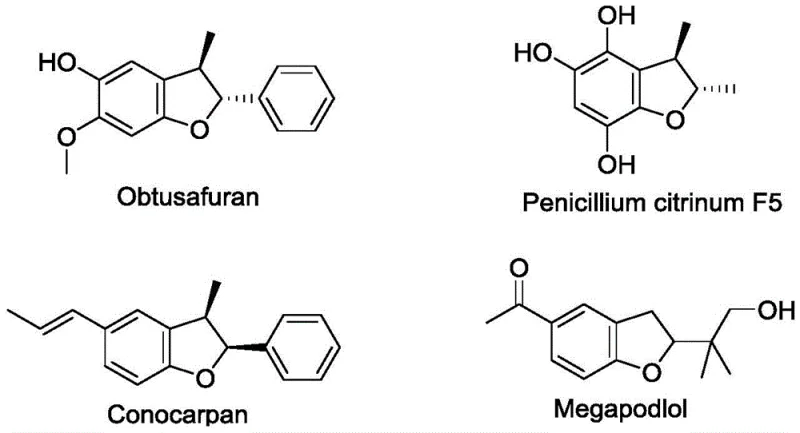 The ability to synthesize these motifs with high stereochemical fidelity is paramount for drug discovery and development. This patent details a novel catalytic system that leverages chiral squaric acid catalysts to drive a domino Michael addition-cyclization sequence, offering a streamlined pathway to high-value intermediates.
The ability to synthesize these motifs with high stereochemical fidelity is paramount for drug discovery and development. This patent details a novel catalytic system that leverages chiral squaric acid catalysts to drive a domino Michael addition-cyclization sequence, offering a streamlined pathway to high-value intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of functionalized dihydrofurans has relied heavily on the use of bromo-nitroolefins as key electrophilic partners. Prominent research groups, such as Rueping et al. in 2010 and the Aleman group in 2013, developed protocols utilizing thiourea or squaric acid catalysts with these brominated substrates. However, these legacy methods suffer from significant drawbacks that hinder their industrial applicability. The reliance on bromo-substituted starting materials often necessitates harsher reaction conditions and introduces additional synthetic steps for halogen removal or functionalization. Furthermore, the enantioselectivity achieved in these traditional routes is frequently inconsistent, with reported ee values ranging broadly from 66% to 92%, which is often insufficient for stringent pharmaceutical purity requirements without extensive and costly recrystallization processes.
The Novel Approach
The methodology disclosed in CN107216297B represents a paradigm shift by eliminating the need for bromo-nitroolefins entirely. Instead, it employs readily accessible non-bromo substituted nitroolefins in conjunction with a specialized chiral squaric acid catalyst. 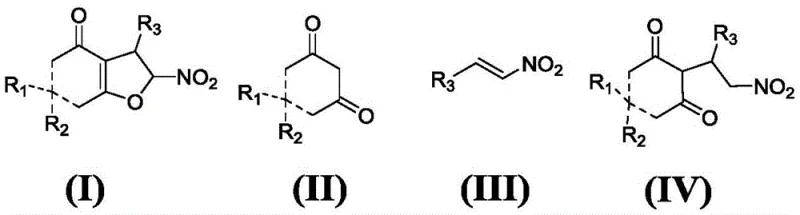 This innovative two-step sequence begins with a highly stereoselective Michael addition to form an intermediate, followed by an iodine-mediated cyclization. The result is a dramatic improvement in performance metrics, with the new process delivering products with up to 99% enantiomeric excess (ee) and greater than 99:1 diastereomeric ratio (dr). By operating under mild temperatures between -20°C and 60°C, this approach not only enhances safety but also significantly reduces energy consumption compared to high-temperature alternatives.
This innovative two-step sequence begins with a highly stereoselective Michael addition to form an intermediate, followed by an iodine-mediated cyclization. The result is a dramatic improvement in performance metrics, with the new process delivering products with up to 99% enantiomeric excess (ee) and greater than 99:1 diastereomeric ratio (dr). By operating under mild temperatures between -20°C and 60°C, this approach not only enhances safety but also significantly reduces energy consumption compared to high-temperature alternatives.
Mechanistic Insights into Squaric Acid-Catalyzed Cyclization
The core of this technological breakthrough lies in the unique activation mode provided by the chiral squaric acid catalysts. These organocatalysts, specifically selected from the series of compounds labeled (V) through (XIII), function through a dual hydrogen-bonding mechanism. 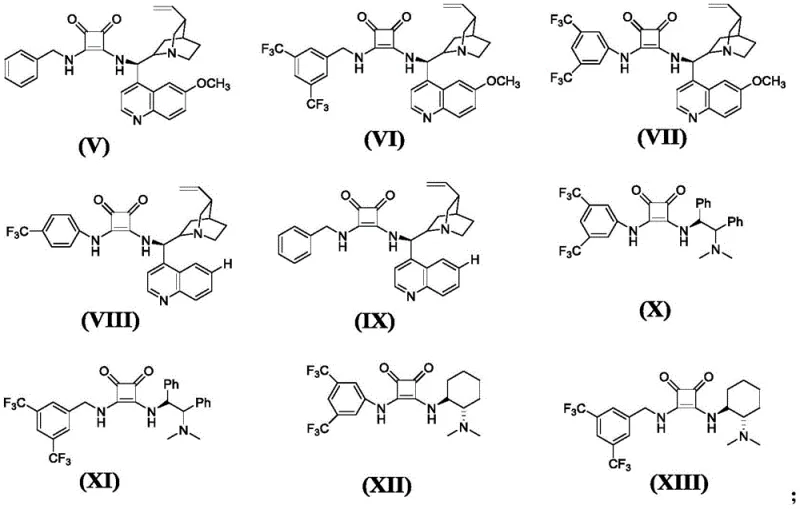 In the initial step, the catalyst simultaneously activates the 1,3-cyclohexanedione derivative and the nitroolefin substrate. This bifunctional activation aligns the reactants in a precise spatial orientation within the chiral pocket of the catalyst, ensuring that the nucleophilic attack occurs exclusively from one face of the molecule. This rigorous control over the transition state geometry is what enables the exceptional enantioselectivity observed, effectively suppressing the formation of unwanted stereoisomers that typically plague racemic syntheses.
In the initial step, the catalyst simultaneously activates the 1,3-cyclohexanedione derivative and the nitroolefin substrate. This bifunctional activation aligns the reactants in a precise spatial orientation within the chiral pocket of the catalyst, ensuring that the nucleophilic attack occurs exclusively from one face of the molecule. This rigorous control over the transition state geometry is what enables the exceptional enantioselectivity observed, effectively suppressing the formation of unwanted stereoisomers that typically plague racemic syntheses.
Following the initial carbon-carbon bond formation, the reaction proceeds to a cyclization phase mediated by an iodine source additive. The presence of the iodine species facilitates an intramolecular nucleophilic attack by the carbonyl oxygen onto the activated nitro-alkene moiety, closing the furan ring. Crucially, the chirality established in the first step is preserved and reinforced during this cyclization event. The subsequent addition of a basic substance neutralizes acidic byproducts and drives the equilibrium toward the final stable dihydrofuran product. This mechanistic elegance ensures that the impurity profile remains clean, with minimal formation of regioisomers or polymeric byproducts, thereby simplifying downstream purification.
How to Synthesize Chiral Dihydrofuran Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and solvent selection to maximize yield and optical purity. The process is designed to be operationally simple, avoiding the need for inert atmosphere techniques in many cases, which facilitates easier scale-up. The patent outlines a clear protocol where the Michael adduct is generated first, followed by the direct addition of cyclization reagents to the same pot or a subsequent vessel. For detailed operational parameters regarding specific substrates and catalyst loadings, please refer to the standardized synthesis guide below.
- Mix 1,3-cyclohexanedione derivatives with nitroolefins and a chiral squaric acid catalyst in an organic solvent at -20 to 60°C to form the Michael adduct intermediate.
- Add an iodine source additive and a basic substance to the intermediate mixture in a second solvent system, reacting at -20 to 60°C to induce cyclization.
- Perform post-treatment including extraction, solvent removal, and column chromatography to isolate the final chiral dihydrofuran compound with high enantiomeric excess.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis method offers compelling advantages that directly address the pain points of procurement managers and supply chain directors. The shift away from specialized brominated starting materials to commodity nitroolefins fundamentally alters the cost structure of the supply chain. By utilizing simpler, more abundant raw materials, manufacturers can mitigate the risks associated with supply volatility and price fluctuations of exotic reagents. Furthermore, the high selectivity of the reaction minimizes waste generation, aligning with modern green chemistry principles and reducing the burden on waste treatment facilities.
- Cost Reduction in Manufacturing: The elimination of bromo-nitroolefins removes a significant cost driver, as halogenated specialty chemicals often command premium pricing due to complex synthesis and handling requirements. Additionally, the high enantioselectivity (up to 99% ee) drastically reduces the need for expensive chiral resolution steps or repeated recrystallizations, which are traditionally resource-intensive. The mild reaction conditions also translate to lower energy costs for heating and cooling, contributing to a leaner overall manufacturing budget.
- Enhanced Supply Chain Reliability: The reliance on 1,3-cyclohexanediones and simple nitroolefins ensures a robust supply base, as these are bulk chemicals produced by multiple global vendors. This diversification of raw material sources protects against single-supplier bottlenecks. Moreover, the tolerance of the reaction to various functional groups on the aromatic ring allows for a flexible substrate scope, meaning the same core process can be adapted for a wide library of derivatives without requalifying entirely new supply chains for each variant.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability potential due to its straightforward workup procedures involving standard extraction and chromatography. The absence of heavy metal catalysts, which are common in alternative transition-metal catalyzed routes, simplifies regulatory compliance regarding residual metal limits in APIs. This metal-free organocatalytic approach inherently produces a cleaner product stream, reducing the environmental footprint and facilitating faster regulatory approval timelines for new drug applications.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this asymmetric synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity for technical teams evaluating this route for potential adoption.
Q: What is the primary advantage of this synthesis method over conventional routes?
A: Unlike conventional methods that require bromo-nitroolefins and often yield moderate enantioselectivity (66-92% ee), this novel approach utilizes non-bromo substituted nitroolefins with chiral squaric acid catalysts, achieving up to 99% ee and excellent diastereoselectivity under mild conditions.
Q: What types of catalysts are employed in this asymmetric synthesis?
A: The process utilizes specific chiral squaric acid catalysts, specifically compounds designated as formulas (V) through (XIII) in the patent documentation, which facilitate hydrogen bond catalysis for precise stereocontrol.
Q: Is this method suitable for large-scale pharmaceutical manufacturing?
A: Yes, the method features mild reaction temperatures ranging from -20°C to 60°C and uses readily available starting materials like 1,3-cyclohexanediones and nitroolefins, making it highly scalable and cost-effective for commercial production of API intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Dihydrofuran Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this asymmetric synthesis technology for the next generation of therapeutic agents. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market supply is seamless. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, including precise measurement of enantiomeric excess and diastereomeric ratios, guaranteeing that every batch meets the highest international standards.
We invite you to collaborate with us to leverage this advanced chemistry for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating how this route can optimize your bill of materials. Please contact our technical procurement team today to request specific COA data for related intermediates and comprehensive route feasibility assessments to accelerate your development timeline.
