Scalable Production of Tunable Bifunctional L-Proline Organocatalysts for Advanced Asymmetric Synthesis
Scalable Production of Tunable Bifunctional L-Proline Organocatalysts for Advanced Asymmetric Synthesis
The landscape of asymmetric synthesis has undergone a paradigm shift with the emergence of organocatalysis, moving away from the historical dominance of expensive and toxic transition metal complexes. Patent CN100361971C introduces a groundbreaking class of bifunctional L-proline derivatives that offer adjustable catalytic activity, specifically designed to overcome the limitations of earlier catalytic systems. These compounds represent a significant technological leap for the production of complex polyhydroxyl compounds and chiral intermediates essential in modern drug discovery. By leveraging a unique structural motif that combines a proline core with a functionalized cyclohexane diamine scaffold, this technology enables high-selectivity transformations under remarkably mild conditions. For R&D directors and process chemists, this patent provides a robust blueprint for developing greener, more efficient synthetic routes that align with modern regulatory standards for impurity control and environmental sustainability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of complex chiral molecules relied heavily on metal-based catalysis, which presents inherent challenges for large-scale pharmaceutical manufacturing. Traditional methods often require rigorous exclusion of moisture and oxygen, necessitating specialized equipment and increasing operational expenditures significantly. Furthermore, the removal of trace metal residues from the final active pharmaceutical ingredient (API) is a costly and time-consuming bottleneck, often requiring multiple purification steps such as chromatography or specialized scavenging resins. Earlier generations of organocatalysts, while metal-free, frequently suffered from narrow substrate scopes, exhibiting poor activity towards less reactive ketones like cyclohexanone or cyclopentanone. This limitation forced manufacturers to rely on more expensive or hazardous activated donors, complicating the supply chain and increasing the overall cost of goods sold. The inability to fine-tune the catalyst structure for specific substrates often resulted in suboptimal yields and selectivity, hindering the economic viability of many potential drug candidates.
The Novel Approach
The innovative strategy outlined in the patent data utilizes a modular synthetic approach to create bifunctional catalysts with tunable electronic and steric properties. By varying the R1 substituent on the acyl group attached to the cyclohexane diamine backbone, chemists can precisely adjust the catalyst's acidity and hydrogen-bonding capability to match specific reaction requirements. This adaptability allows for high conversion rates and excellent stereoselectivity even with challenging, low-activity substrates that previously failed under standard proline catalysis. The synthesis route itself is streamlined, utilizing readily available starting materials like (1R,2R)-diaminocyclohexane and protected proline derivatives, which ensures a reliable supply chain for raw materials. This novel approach not only enhances reaction efficiency but also simplifies downstream processing, as the absence of metals eliminates the need for complex heavy metal clearance protocols. Consequently, this technology offers a compelling value proposition for procurement teams seeking to reduce manufacturing costs while maintaining the highest purity standards.
Mechanistic Insights into Bifunctional Organocatalysis
The superior performance of these L-proline derivatives stems from their ability to operate via a dual-activation mechanism, simultaneously activating both the nucleophile and the electrophile in the aldol reaction. The secondary amine of the proline ring forms an enamine intermediate with the ketone donor, lowering the energy barrier for nucleophilic attack. Concurrently, the amide NH group on the cyclohexane scaffold acts as a hydrogen bond donor, stabilizing the transition state and orienting the aldehyde acceptor for optimal stereocontrol. This bifunctional cooperation creates a highly organized chiral environment that dictates the facial selectivity of the reaction, resulting in high enantiomeric excess (ee) values often exceeding 90%. The rigidity of the cyclohexane ring further restricts conformational freedom, ensuring that the catalytic pocket remains well-defined throughout the reaction cycle. Understanding this mechanistic nuance is critical for process optimization, as it explains why these catalysts maintain high activity across a diverse range of aromatic and aliphatic aldehydes.
Impurity control is another critical aspect where this mechanistic design excels, particularly regarding the suppression of side reactions common in aldol chemistry. The specific spatial arrangement of the functional groups minimizes non-productive background reactions and prevents the formation of unwanted diastereomers. By carefully selecting the R1 group, such as electron-withdrawing trifluoromethyl or halogen substituents, the acidity of the hydrogen bond donor can be enhanced, further accelerating the reaction rate without compromising selectivity. This level of control is vital for meeting the stringent impurity profiles required by global regulatory agencies. For quality assurance teams, the predictability of the impurity spectrum simplifies method development and validation, reducing the time to market for new drug substances. The robustness of the catalytic cycle ensures consistent batch-to-batch reproducibility, a key metric for successful technology transfer from pilot plant to commercial manufacturing.
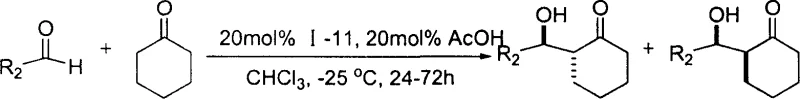
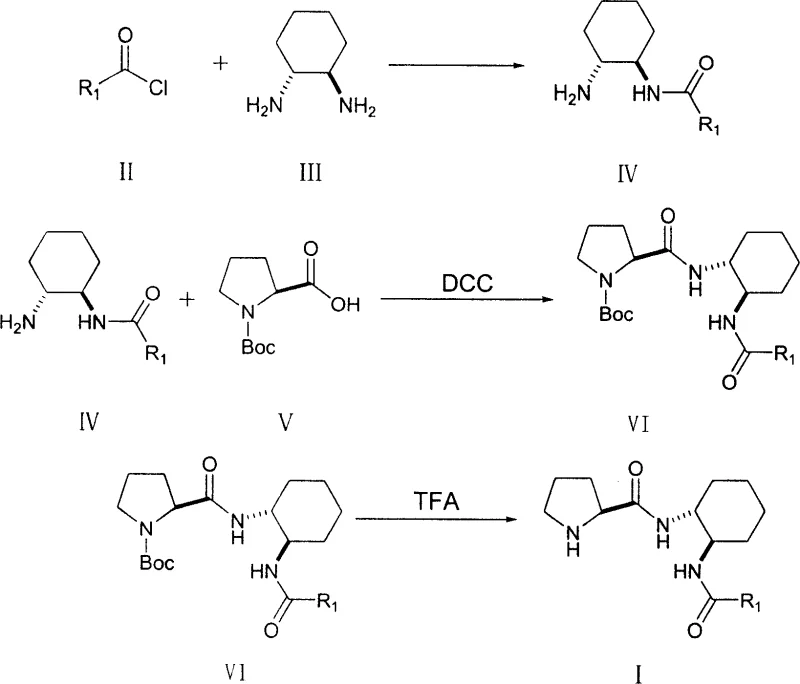
How to Synthesize Bifunctional L-Proline Derivatives Efficiently
The preparation of these high-value organocatalysts follows a logical three-step sequence that is amenable to scale-up in standard multipurpose reactors. The process begins with the formation of a mono-amide intermediate, followed by peptide coupling with a protected proline species, and concludes with a deprotection step to reveal the active catalyst. Each stage utilizes common organic solvents and reagents, avoiding the need for exotic or highly hazardous chemicals that would complicate safety assessments. The detailed procedural parameters, including stoichiometry, temperature controls, and purification methods, are optimized to maximize yield and minimize waste generation. For process engineers looking to implement this technology, the following guide outlines the critical operational windows and workup procedures necessary to achieve pharmaceutical-grade quality. Adhering to these standardized protocols ensures that the final catalyst exhibits the precise stereochemical integrity required for high-performance asymmetric synthesis.
- React acid chloride with (1R,2R)-diaminocyclohexane in dichloromethane at room temperature to form the amide intermediate.
- Couple the amide intermediate with Boc-protected L-proline using DCC as a coupling agent in anhydrous dichloromethane.
- Remove the Boc protecting group using trifluoroacetic acid (TFA) to yield the final bifunctional L-proline derivative catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this bifunctional organocatalyst technology offers substantial strategic advantages for supply chain resilience and cost management. The elimination of precious metal catalysts removes exposure to volatile commodity prices associated with rhodium, palladium, or iridium, stabilizing long-term production costs. Furthermore, the simplified purification workflow reduces the consumption of silica gel and solvents during column chromatography, directly lowering the cost of goods sold and minimizing hazardous waste disposal fees. The use of stable, shelf-stable intermediates allows for flexible inventory management, enabling manufacturers to stockpile key precursors without degradation concerns. This flexibility is crucial for mitigating supply chain disruptions and ensuring continuous production schedules for high-demand API intermediates. By integrating this technology, companies can achieve a more sustainable manufacturing footprint while enhancing their competitive positioning in the global fine chemicals market.
- Cost Reduction in Manufacturing: The transition to metal-free organocatalysis fundamentally alters the cost structure of asymmetric synthesis by removing the most expensive line items from the bill of materials. Without the need for costly transition metal salts or ligands, the raw material costs are significantly reduced, and the associated expenses for metal scavenging resins are completely eliminated. Additionally, the mild reaction conditions, often proceeding at room temperature or slightly below, result in lower energy consumption for heating and cooling compared to traditional high-pressure or cryogenic metal-catalyzed processes. The higher selectivity of these bifunctional catalysts also means fewer by-products are formed, leading to improved overall yields and reduced loss of valuable starting materials. These cumulative efficiencies translate into a leaner, more cost-effective manufacturing process that improves profit margins without sacrificing product quality.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as L-proline, diamines, and simple acid chlorides ensures a robust and diversified supply base that is less susceptible to geopolitical bottlenecks. Unlike specialized metal catalysts that may have single-source suppliers or long lead times, the precursors for these organocatalysts are widely available from multiple global vendors. This redundancy in the supply chain mitigates the risk of production stoppages due to raw material shortages. Moreover, the stability of the final catalyst products allows for extended storage periods, facilitating just-in-time delivery models and reducing the need for expedited shipping. For supply chain heads, this reliability translates into predictable lead times and the ability to commit to long-term supply agreements with confidence, strengthening relationships with downstream pharmaceutical partners.
- Scalability and Environmental Compliance: The synthetic route described in the patent is inherently scalable, utilizing unit operations that are standard in the fine chemical industry, such as liquid-liquid extraction and crystallization. The absence of heavy metals simplifies the environmental permitting process and reduces the regulatory burden associated with wastewater treatment and effluent monitoring. This alignment with green chemistry principles not only lowers compliance costs but also enhances the corporate sustainability profile, which is increasingly important for securing contracts with environmentally conscious multinational corporations. The ability to scale from gram-scale laboratory experiments to multi-ton commercial production without significant process re-engineering accelerates the time to market for new products. This scalability ensures that the technology can grow with demand, supporting both early-stage clinical trials and full-scale commercial launches seamlessly.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic technology in industrial settings. These answers are derived directly from the experimental data and scope defined in the patent documentation, providing clarity on performance metrics and operational feasibility. Understanding these details is essential for making informed decisions about process adoption and resource allocation. The responses cover critical aspects ranging from substrate compatibility to purification strategies, ensuring that all stakeholders have a comprehensive understanding of the technology's capabilities.
Q: What is the primary advantage of these bifunctional L-proline derivatives over traditional metal catalysts?
A: These organocatalysts eliminate the need for toxic transition metals, simplifying purification processes and ensuring the final product meets stringent heavy metal limits required for pharmaceutical applications.
Q: Can the catalytic activity be adjusted for different substrates?
A: Yes, the R1 group on the benzoyl or acyl moiety can be varied (e.g., methyl, chloro, trifluoromethyl) to tune the electronic and steric properties, optimizing activity for less reactive donors like cyclohexanone.
Q: Is this synthesis route suitable for large-scale commercial production?
A: The process utilizes standard reagents like DCC and TFA under mild conditions (room temperature), avoiding extreme pressures or temperatures, which facilitates straightforward scale-up from laboratory to industrial tonnage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Proline Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced organocatalysis in driving the next generation of pharmaceutical innovation. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to industrial manufacturing is seamless and efficient. We are committed to delivering high-purity L-proline derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in process optimization allows us to fine-tune reaction parameters to maximize yield and minimize impurities, guaranteeing a consistent supply of high-quality catalysts for your critical synthetic steps. By partnering with us, you gain access to a reliable supply chain partner dedicated to supporting your R&D and commercial goals with technical excellence.
We invite you to engage with our technical procurement team to discuss how these bifunctional catalysts can be integrated into your existing workflows to drive efficiency and cost savings. Request a Customized Cost-Saving Analysis today to quantify the potential economic benefits for your specific application. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your project requirements, ensuring that you have all the information needed to make strategic sourcing decisions. Let us help you unlock the full potential of asymmetric organocatalysis for your upcoming projects.
