Advanced One-Step Synthesis of 2-(4-tert-butyl-phenyl) Malonic Mononitrile Ester for Agrochemicals
Introduction to Patent CN102140071A
The pharmaceutical and agrochemical industries are constantly seeking more efficient pathways to produce complex intermediates, and patent CN102140071A presents a significant breakthrough in the synthesis of 2-(4-tert-butyl-phenyl) malonic mononitrile (2-methoxyl) ethyl ester. This specific compound serves as a critical precursor for Cyflumetofen, a novel and highly effective miticide developed for controlling harmful mites on fruit trees, vegetables, and ornamental plants without cross-resistance issues. The patent details a transformative shift from a cumbersome, moisture-sensitive two-step process to a streamlined, robust one-step acylation reaction. By leveraging 4-tert-butylbenzyl cyanide and 2-methoxyethyl chloroformate as primary raw materials, this methodology not only simplifies the operational workflow but also drastically enhances the overall yield and purity profile of the final intermediate. For global supply chain managers and R&D directors, understanding this technological pivot is essential for securing a reliable agrochemical intermediate supplier capable of meeting the rigorous demands of modern pesticide manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the industrial preparation of this key ester intermediate was plagued by a convoluted two-step synthetic route that imposed severe operational constraints and quality risks. The traditional first step involved reacting 4-tert-butylbenzyl cyanide with dimethyl carbonate in the presence of sodium methylate at elevated temperatures ranging from 90°C to 130°C. This was followed by a second transesterification step requiring aluminum isopropoxide as a catalyst and excessive ethylene glycol monomethyl ether. A critical flaw in this legacy process is the extreme sensitivity of the reagents—specifically sodium methylate and aluminum isopropoxide—to atmospheric moisture. Even trace amounts of water, particularly during humid summer months, could hydrolyze the catalysts, leading to incomplete reactions, difficult purification, and ultimately, reaction failure. Furthermore, the cumulative yield of this two-step process was suboptimal, with the first step yielding approximately 80.9% and the second step around 84.3%, resulting in significant material loss and increased waste generation.
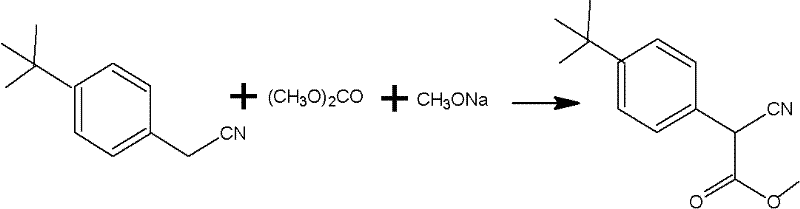
The Novel Approach
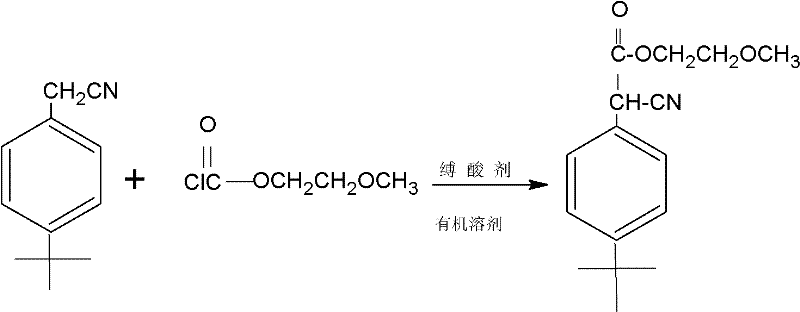
In stark contrast to the legacy methodology, the novel approach described in the patent utilizes a direct, one-step acylation strategy that fundamentally reshapes the production landscape for this agrochemical intermediate. By reacting 4-tert-butylbenzyl cyanide directly with 2-methoxyethyl chloroformate in the presence of an organic acid-binding agent, the process eliminates the need for harsh alkaline conditions and moisture-sensitive metal catalysts. The reaction proceeds under mild conditions, with the addition of the chloroformate occurring at a controlled temperature of 15°C to 30°C, followed by a maintenance phase at 30°C to 50°C. This gentle thermal profile not only preserves the integrity of the reactants but also minimizes the formation of thermal degradation byproducts. The result is a brownish-red oily product obtained after simple filtration and solvent removal, boasting yields consistently above 95% across various embodiments. This transition from a fragile, multi-stage process to a robust, single-vessel operation represents a paradigm shift in cost reduction in agrochemical manufacturing.
Mechanistic Insights into Acid-Binding Agent Catalyzed Acylation
The core chemical transformation in this novel process is a nucleophilic substitution where the activated methylene group of the nitrile attacks the carbonyl carbon of the chloroformate. The presence of the electron-withdrawing cyano group (-CN) adjacent to the benzyl position significantly increases the acidity of the alpha-protons, allowing for facile deprotonation by the organic base. Unlike the conventional method which relied on strong inorganic bases like sodium methylate that necessitated strict anhydrous conditions, this new route employs organic acid-binding agents such as triethylamine, DMAP (4-dimethylaminopyridine), or anhydrous carbonates. These agents serve a dual purpose: they generate the reactive carbanion species necessary for the nucleophilic attack and simultaneously scavenge the hydrochloric acid byproduct formed during the displacement of the chloride leaving group. This in-situ neutralization prevents the accumulation of acidic species that could otherwise catalyze the decomposition of the sensitive chloroformate or the product ester.
From an impurity control perspective, this mechanistic pathway offers superior selectivity. The mild reaction temperatures (15°C to 50°C) effectively suppress side reactions such as the hydrolysis of the chloroformate or the polymerization of the nitrile species, which are common pitfalls in high-temperature processes. Additionally, the use of non-polar or moderately polar organic solvents like toluene, xylene, or cyclohexane facilitates the precipitation of the amine hydrochloride salts (e.g., triethylamine hydrochloride) as solids. This physical property allows for the easy removal of the primary byproduct via simple filtration, thereby streamlining the downstream workup. The resulting filtrate contains the crude product with high purity, requiring only solvent evaporation to obtain the final intermediate, thus eliminating the need for complex aqueous extractions or column chromatography often associated with lower-yielding routes.
How to Synthesize 2-(4-tert-butyl-phenyl) Malonic Mononitrile Ester Efficiently
Implementing this synthesis on a commercial scale requires precise adherence to the stoichiometric ratios and thermal profiles outlined in the patent to maximize efficiency and safety. The process begins with the careful selection of the acid-binding agent and solvent system, which dictates the reaction kinetics and the ease of byproduct removal. Operators must ensure that the addition rate of the chloroformate is controlled to manage the exotherm, preventing local hot spots that could degrade the reagents. The following guide summarizes the critical operational parameters derived from the patent embodiments, providing a roadmap for process engineers to replicate the high yields observed in the laboratory setting. For detailed standard operating procedures and safety data sheets, please refer to the technical documentation provided below.
- Charge a reaction flask with 4-tert-butylbenzyl cyanide, an organic solvent (such as toluene or xylene), and an acid-binding agent like triethylamine or DMAP.
- Slowly drip 2-methoxyethyl chloroformate into the mixture while maintaining the temperature between 15°C and 30°C to control exothermicity.
- Maintain the reaction temperature at 30°C to 50°C until completion, filter out solid salts, wash the filtrate, and remove the solvent to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers tangible strategic advantages that extend far beyond simple chemistry. The elimination of the two-step sequence inherently reduces the total processing time, equipment occupancy, and labor hours required per batch, leading to substantial cost savings in manufacturing overhead. By removing the dependency on aluminum isopropoxide and sodium methylate, companies can avoid the logistical challenges and price volatility associated with these specialized, moisture-sensitive reagents. Instead, the process relies on commodity chemicals like triethylamine and common solvents, which are readily available from multiple global suppliers, thereby enhancing supply chain resilience and reducing the risk of production stoppages due to raw material shortages.
- Cost Reduction in Manufacturing: The consolidation of two reaction steps into a single vessel operation dramatically lowers energy consumption by eliminating the need for prolonged heating cycles up to 130°C and subsequent cooling phases. Furthermore, the high yield (>95%) significantly reduces the cost of goods sold (COGS) by minimizing raw material waste and maximizing the output per unit of input. The simplicity of the workup, which involves mere filtration and solvent distillation rather than complex aqueous extractions and drying steps, further reduces utility costs and waste disposal fees, contributing to a leaner and more profitable production model.
- Enhanced Supply Chain Reliability: One of the most critical supply chain benefits is the robustness of the process against environmental variables. The conventional method's susceptibility to humidity meant that production schedules were often at the mercy of seasonal weather patterns, particularly in regions with high summer moisture. The new method's tolerance to ambient conditions ensures consistent batch-to-batch quality and predictable lead times, regardless of the season. This reliability allows supply chain planners to maintain lower safety stock levels and commit to tighter delivery windows with confidence, knowing that the production process is not prone to sudden failures caused by trace moisture ingress.
- Scalability and Environmental Compliance: The use of recyclable organic solvents like toluene and xylene, combined with the generation of solid amine salts that can be easily separated and potentially regenerated, aligns well with modern green chemistry principles. The process avoids the generation of large volumes of aluminum-containing wastewater, which is difficult and expensive to treat. This simplified waste profile facilitates easier regulatory compliance and reduces the environmental footprint of the manufacturing site. Additionally, the straightforward nature of the reaction makes it highly scalable from pilot plant quantities to multi-ton commercial production without the need for specialized high-pressure or cryogenic equipment.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific applications, we have compiled answers to common inquiries regarding the synthesis and handling of this intermediate. These insights are derived directly from the experimental data and technical specifications found in the patent literature, addressing concerns about scalability, purity, and process safety. Understanding these nuances is vital for making informed decisions about integrating this intermediate into broader agrochemical synthesis pipelines.
Q: Why is the new one-step method superior to the conventional two-step process regarding environmental stability?
A: The conventional method relies on sodium methylate and aluminum isopropoxide, both of which are extremely sensitive to moisture, often leading to reaction failure in humid conditions. The novel method utilizes organic acid-binding agents and chloroformates, which significantly reduces sensitivity to atmospheric humidity, ensuring consistent batch quality even in summer months.
Q: What yield improvements can be expected with this novel synthetic route?
A: While the traditional two-step process typically achieves yields around 80-84% per step (resulting in a lower overall yield), the patented one-step acylation method consistently demonstrates yields exceeding 95%, as evidenced by multiple embodiments in the patent data.
Q: Which solvents are compatible with this synthesis for large-scale production?
A: The process is highly versatile regarding solvent choice, successfully utilizing common industrial solvents such as toluene, xylene, cyclohexane, and petroleum benzine, allowing procurement teams to optimize costs based on regional availability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(4-tert-butyl-phenyl) Malonic Mononitrile Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields demonstrated in patent CN102140071A can be reliably replicated on an industrial scale. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of 2-(4-tert-butyl-phenyl) malonic mononitrile (2-methoxyl) ethyl ester meets the exacting standards required for the synthesis of high-performance miticides like Cyflumetofen.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains and reduce costs through advanced process chemistry. By leveraging our expertise in this specific one-step acylation technology, we can offer a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact our technical procurement team today to request specific COA data, discuss route feasibility assessments, and explore how our optimized manufacturing processes can drive value for your agrochemical projects.
