Advanced Copper-Catalyzed Diaryl Ether Synthesis for Commercial Scale Manufacturing
Advanced Copper-Catalyzed Diaryl Ether Synthesis for Commercial Scale Manufacturing
The landscape of fine chemical manufacturing is constantly evolving, driven by the relentless pursuit of more efficient, cost-effective, and environmentally sustainable synthetic routes. A pivotal advancement in this domain is detailed in patent CN101445437A, which discloses an improved process for the catalytic synthesis of diaryl ethers. This technology addresses long-standing challenges in forming carbon-oxygen bonds, specifically targeting the limitations of classical Ullmann couplings and modern palladium-catalyzed methods. By leveraging a novel catalyst system comprising a copper(I) salt and a 1-substituted imidazole, this methodology enables the coupling of aryl or heteroaryl bromides with phenols under significantly milder conditions. For global procurement leaders and R&D directors, this represents a strategic opportunity to optimize the supply chain for critical pharmaceutical intermediates and agrochemical building blocks, ensuring both economic viability and technical robustness in high-volume production scenarios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of diaryl ethers has been dominated by the classical Ullmann condensation, a reaction that, while effective, suffers from severe practical drawbacks that hinder its application in modern commercial settings. Traditional protocols typically require stoichiometric or even super-stoichiometric amounts of copper metal or copper salts, leading to significant waste generation and difficult downstream purification processes due to heavy metal contamination. Furthermore, these reactions often necessitate extremely high temperatures, frequently exceeding 200°C, which poses safety risks and limits the functional group tolerance of the substrates. Alternatively, the industry has shifted towards palladium-catalyzed Buchwald-Hartwig type couplings to mitigate these thermal issues. However, these palladium systems introduce their own set of economic and logistical burdens, primarily the reliance on expensive precious metal catalysts and sophisticated, often air-sensitive phosphine ligands. The high cost of palladium and the complexity of ligand synthesis create a substantial barrier to entry for cost-sensitive large-scale manufacturing, making the search for a robust base-metal alternative a critical priority for supply chain optimization.
The Novel Approach
The methodology outlined in the patent data presents a transformative solution by utilizing a catalytic amount of copper in conjunction with simple, commercially available 1-substituted imidazoles. This approach effectively bridges the gap between the low cost of classical copper chemistry and the high efficiency of modern palladium catalysis. The system operates at moderate temperatures, typically ranging from 120°C to 140°C, which is significantly lower than classical Ullmann conditions yet sufficiently energetic to drive the coupling of challenging substrates. Crucially, the catalyst loading is drastically reduced to as low as 1 mol% to 10 mol%, representing a massive reduction in metal usage compared to stoichiometric methods. The use of 1-butylimidazole or similar derivatives as ligands stabilizes the copper center, facilitating the oxidative addition and reductive elimination steps essential for the catalytic cycle. This innovation allows for the successful coupling of a wide array of substrates, including electron-deficient and electron-rich aryl bromides, as well as sterically hindered phenols, without the need for exotic reagents or cryogenic conditions.
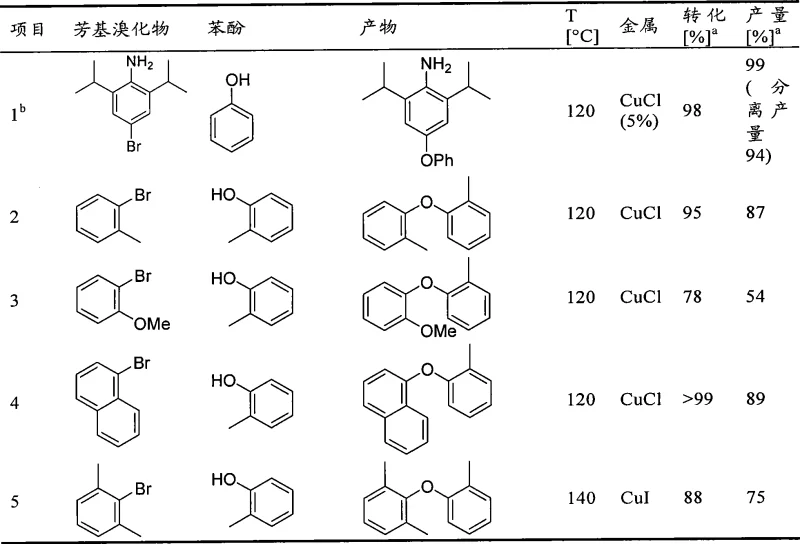
The versatility of this novel catalytic system is vividly demonstrated by the broad substrate scope detailed in the experimental data. As illustrated in the reaction table, the protocol successfully accommodates diverse structural motifs, ranging from simple phenyl rings to complex naphthyl and heteroaryl systems. For instance, the coupling of 4-bromo-2,6-diisopropylaniline with phenol proceeds with exceptional efficiency, achieving a 99% yield at 120°C, showcasing the system's ability to overcome significant steric hindrance that typically plagues nucleophilic aromatic substitutions. Similarly, the reaction tolerates various functional groups, including amino, methoxy, and nitro groups, without requiring extensive protection-deprotection strategies. This high level of chemoselectivity is particularly advantageous for the synthesis of complex pharmaceutical intermediates where orthogonal reactivity is paramount. The data further confirms that both copper(I) chloride and copper(I) iodide serve as effective precursors, providing flexibility in raw material sourcing for manufacturing plants.
Mechanistic Insights into Cu/Imidazole-Catalyzed Etherification
Understanding the mechanistic underpinnings of this copper-catalyzed transformation is essential for R&D teams aiming to implement this technology at scale. The reaction is believed to proceed through a catalytic cycle where the 1-substituted imidazole acts as a monodentate or hemilabile ligand, coordinating to the copper(I) center to form an active catalytic species. This coordination enhances the solubility of the copper salt in organic solvents like toluene or xylene and prevents the aggregation of copper into inactive clusters, a common deactivation pathway in classical Ullmann reactions. The active copper-ligand complex facilitates the oxidative addition of the aryl bromide, generating an aryl-copper(III) intermediate. Subsequent coordination of the phenoxide anion, generated in situ by the base (typically potassium carbonate), leads to the formation of the diaryl ether product via reductive elimination, regenerating the copper(I) catalyst. The presence of the imidazole ligand lowers the activation energy for these elementary steps, thereby allowing the reaction to proceed at the observed moderate temperatures of 120°C to 140°C. This mechanistic efficiency is what enables the high turnover numbers and excellent yields observed across the diverse substrate library.
Furthermore, the impurity profile of the resulting diaryl ethers is significantly improved compared to traditional methods, a critical factor for regulatory compliance in pharmaceutical manufacturing. The high selectivity of the copper/imidazole system minimizes the formation of homocoupling byproducts (biaryls) and dehalogenated side products, which are common impurities in less controlled radical pathways. The use of mild bases like potassium carbonate or potassium acetate, rather than strong alkoxides, further reduces the risk of ester hydrolysis or other base-sensitive side reactions. This clean reaction profile simplifies the downstream purification process, often allowing for direct crystallization or simple chromatographic separation, thereby reducing solvent consumption and processing time. For quality control teams, this translates to a more consistent and predictable product specification, reducing the risk of batch failures and ensuring a reliable supply of high-purity intermediates for downstream drug substance synthesis.
How to Synthesize Diaryl Ethers Efficiently
Implementing this synthetic route requires careful attention to reaction parameters to maximize yield and minimize impurities. The general procedure involves charging a reactor with the aryl bromide, phenol, copper catalyst, and base under an inert atmosphere to prevent catalyst oxidation. The choice of solvent is flexible, with toluene and xylene being preferred for their ability to sustain the required reaction temperatures while maintaining good solubility for the organic substrates. The addition of the 1-substituted imidazole ligand is critical, with molar ratios typically ranging from 50 mol% to 200 mol% relative to the substrate. Detailed standardized operating procedures for scaling this reaction from gram to kilogram scale are essential for technology transfer.
- Charge a reaction vessel with potassium carbonate, copper(I) chloride, the phenol substrate, and the aryl bromide under an inert argon atmosphere.
- Add the solvent, typically toluene or xylene, along with the 1-substituted imidazole ligand such as 1-butylimidazole.
- Heat the mixture to between 120°C and 140°C and stir until conversion is complete, followed by standard aqueous workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed process offers compelling economic and logistical advantages over legacy technologies. The primary driver for cost reduction is the substitution of expensive palladium catalysts with abundant and inexpensive copper salts. Copper is orders of magnitude cheaper than palladium, and the ligand system utilizes simple imidazole derivatives that are commodity chemicals, unlike the complex, multi-step synthesized phosphine ligands required for palladium catalysis. This shift in raw material costs directly impacts the bill of materials, leading to substantial cost savings in the manufacturing of diaryl ether intermediates. Additionally, the lower catalyst loading reduces the burden on metal scavenging and removal processes, which are often costly and time-consuming steps required to meet strict residual metal specifications in pharmaceutical products. The elimination of these purification bottlenecks streamlines the production workflow and reduces overall processing time.
- Cost Reduction in Manufacturing: The economic benefits extend beyond just the price of the catalyst. The ability to run the reaction at moderate temperatures (120°C - 140°C) reduces energy consumption compared to the extreme heating required for classical Ullmann couplings. Furthermore, the high conversion rates and selectivity minimize the loss of valuable starting materials, improving the overall atom economy of the process. By avoiding the use of specialized, air-sensitive reagents, the process also reduces the need for specialized handling equipment and inert gas consumption, further lowering operational expenditures. These cumulative efficiencies result in a significantly leaner cost structure for the production of high-value fine chemicals.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the reliance on commodity chemicals enhances the resilience of the manufacturing process. Copper salts and imidazoles are widely available from multiple global suppliers, mitigating the risk of supply disruptions that can occur with single-source specialty ligands or precious metals. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality or environmental conditions, ensuring consistent batch-to-batch performance. This reliability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery requirements of downstream pharmaceutical customers. The scalability of the process ensures that supply can be rapidly ramped up to meet market demand without the need for extensive process re-optimization.
- Scalability and Environmental Compliance: The environmental footprint of this process is markedly lower than traditional methods, aligning with increasingly stringent global sustainability regulations. The reduction in heavy metal usage decreases the toxicity of the waste stream, simplifying wastewater treatment and disposal. The use of common organic solvents like toluene allows for efficient solvent recovery and recycling, minimizing volatile organic compound (VOC) emissions. Moreover, the high efficiency of the reaction reduces the volume of chemical waste generated per kilogram of product. These factors contribute to a more sustainable manufacturing profile, which is becoming a key differentiator in supplier selection criteria for major multinational corporations committed to green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed diaryl ether synthesis. These insights are derived directly from the experimental data and technical specifications provided in the patent literature, offering a clear understanding of the process capabilities and limitations. Understanding these details is vital for project managers evaluating the feasibility of integrating this technology into existing production lines.
Q: What represents the key innovation in this copper-catalyzed diaryl ether synthesis?
A: The key innovation lies in the use of inexpensive 1-substituted imidazoles as ligands combined with catalytic amounts of copper salts, replacing expensive palladium systems or harsh stoichiometric copper conditions.
Q: Does this method tolerate sterically hindered substrates?
A: Yes, the patent data demonstrates high conversion rates even for sterically demanding substrates such as 2,6-diisopropyl-4-bromoaniline and 1-bromonaphthalene, achieving yields up to 99%.
Q: Is this process suitable for large-scale industrial production?
A: Absolutely. The use of robust copper catalysts, common solvents like toluene, and moderate temperatures (120-140°C) makes the process highly scalable and economically viable for tonnage production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diaryl Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes in the modern pharmaceutical supply chain. Our technical team has extensively evaluated the copper-catalyzed diaryl ether synthesis described in patent CN101445437A and confirmed its potential for delivering high-purity intermediates at a competitive cost. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of materials. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the exacting standards required for API synthesis. We are committed to leveraging advanced catalytic technologies to drive value for our partners.
We invite potential collaborators to engage with our technical procurement team to discuss how this innovative chemistry can be tailored to your specific project needs. Whether you require a Customized Cost-Saving Analysis for an existing product line or need to develop a new route for a complex intermediate, our experts are ready to assist. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on real-world performance metrics. Partner with us to secure a sustainable and efficient supply chain for your critical diaryl ether intermediates.
