Advanced Catalytic Synthesis of 2-Alkyl-1,3-Propanediol for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to synthesize critical building blocks, and the recent disclosure in patent CN111018664B presents a transformative approach to producing 2-alkyl-1,3-propanediol compounds. These diols serve as indispensable intermediates in the manufacture of advanced liquid crystal materials and various active pharmaceutical ingredients, where structural precision and purity are paramount. Traditionally, the synthesis of these valuable diols has been plagued by inefficient multi-step processes and costly reagents, creating bottlenecks for large-scale production. This new methodology introduces a highly selective catalytic system that utilizes readily available 2-alkylacroleins and simple alcohols to construct the carbon backbone with exceptional efficiency. By leveraging novel phthalimide and naphthalimide derivatives as catalysts, the process achieves total yields exceeding 70 percent under mild reaction conditions, representing a significant leap forward in synthetic organic chemistry. For R&D directors and procurement specialists alike, this technology offers a compelling solution to reduce dependency on scarce raw materials while simultaneously lowering the environmental footprint of chemical manufacturing operations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 2-alkyl-1,3-propanediols has relied heavily on the alkylation of diethyl malonate, a pathway that is fraught with significant economic and technical disadvantages. As illustrated in the traditional reaction scheme below, the initial alkylation step invariably generates substantial quantities of diethyl 2,2-di(alkyl)malonate as a stubborn byproduct, which is chemically similar to the desired mono-alkylated intermediate and extremely difficult to separate. 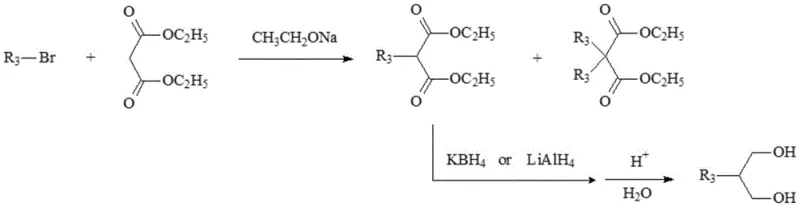
This separation challenge not only drives up processing costs due to the need for rigorous purification but also results in a yield loss of at least 10 percent at this early stage. Furthermore, the subsequent reduction of the ester groups requires the use of potent and expensive reducing agents such as sodium borohydride, lithium aluminum hydride, or red aluminum in at least two molar equivalents. The handling of these pyrophoric or moisture-sensitive reagents necessitates specialized equipment and strict safety protocols, further inflating the operational expenditure. Consequently, the conventional route suffers from poor atom economy, high waste generation, and a complex workup procedure that hinders its viability for cost-sensitive, large-volume applications in the competitive global market.
The Novel Approach
In stark contrast, the innovative method disclosed in the patent utilizes a direct addition reaction between 2-alkylacrolein and an alcohol compound, catalyzed by specifically designed imide derivatives. This strategy bypasses the problematic malonate alkylation entirely, utilizing inexpensive and abundant starting materials that are commercially available on a multi-ton scale. The core of this breakthrough lies in the use of catalysts 1A, 1B, or 1C, which facilitate the formation of the beta-aldehyde ether intermediate with high regioselectivity and minimal side reactions. Unlike the harsh conditions of the old method, this addition proceeds smoothly at temperatures ranging from 0 to 80 degrees Celsius, significantly reducing energy consumption. The subsequent conversion to the final diol involves a straightforward catalytic hydrogenation followed by ether deprotection, eliminating the need for stoichiometric metal hydrides. This streamlined two-step sequence not only simplifies the process flow but also drastically improves the overall mass balance, making it an ideal candidate for green chemistry initiatives and sustainable manufacturing practices.
Mechanistic Insights into Phthalimide-Catalyzed Addition
The success of this synthetic route hinges on the unique structural properties of the catalysts employed, specifically the 3,4-di(formate)-phthalimide and related naphthalimide compounds. These catalysts are engineered with ester functionalities that play a dual role: enhancing solubility in organic media during the reaction phase and facilitating recovery post-reaction. The mechanism involves the activation of the alcohol nucleophile by the imide catalyst, which then attacks the beta-position of the 2-alkylacrolein in a conjugate addition manner. 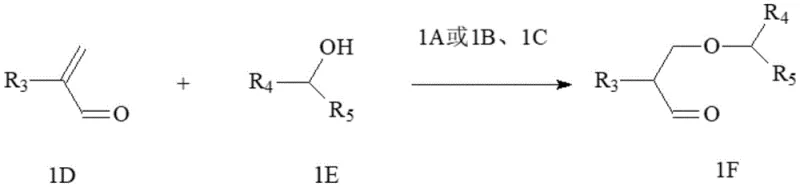
This catalytic cycle is remarkably robust, tolerating a wide range of substituents on both the acrolein and the alcohol, including aromatic, aliphatic, and heteroatom-containing groups. Crucially, the catalyst design addresses a common pitfall in previous attempts where strong acids or bases led to polymerization or acetal formation. By maintaining a neutral to mildly acidic/basic environment depending on the specific catalyst variant, the system prevents the decomposition of the sensitive beta-aldehyde ether intermediate. Furthermore, the solubility switch mechanism allows for easy catalyst recycling; post-reaction hydrolysis converts the soluble ester-catalyst into an insoluble carboxylic acid form, which precipitates out and can be filtered off. This recovered material can then be re-esterified to regenerate the active catalyst, ensuring that the expensive catalytic component does not contribute significantly to the variable cost of goods sold.
Following the formation of the intermediate, the process employs standard catalytic hydrogenation using nickel or palladium on carbon to reduce the aldehyde moiety to a primary alcohol. This step is highly efficient and operates under moderate hydrogen pressures between 0.4 and 8.0 MPa. The final deprotection of the ether bond, whether via acid hydrolysis for alkyl ethers or hydrogenolysis for benzyl ethers, proceeds cleanly to yield the target 2-alkyl-1,3-propanediol. The entire sequence is designed to minimize impurity carryover, ensuring that the final crude product is amenable to simple purification methods like distillation or crystallization to reach purity specifications above 98 percent.
How to Synthesize 2-Alkyl-1,3-Propanediol Efficiently
Implementing this synthesis protocol requires careful attention to the preparation of the reaction mixture and the control of hydrogenation parameters to ensure optimal yield and safety. The process begins with the precise mixing of the catalyst and alcohol solvent, followed by the controlled addition of the reactive acrolein species to manage exotherms. Detailed standardized operating procedures for each stage, including specific molar ratios and temperature profiles, are essential for reproducibility. For a comprehensive guide on executing this synthesis with maximum efficiency, please refer to the step-by-step instructions provided below.
- Mix the specific phthalimide-based catalyst (1A, 1B, or 1C) with the chosen alcohol compound and stir at 0-80°C while adding 2-alkylacrolein.
- Isolate the beta-aldehyde ether intermediate (1F) after reaction completion, typically achieving yields over 80%.
- Subject the intermediate to catalytic hydrogenation (Ni or Pd/C) followed by acid hydrolysis or hydrogenolysis to remove the ether protecting group.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers profound advantages that directly address the pain points of procurement managers and supply chain directors in the fine chemical sector. By shifting away from the diethyl malonate route, manufacturers can eliminate the reliance on volatile and expensive reducing agents, leading to a substantial reduction in raw material costs. The ability to recycle the catalyst further enhances the economic viability of the process, as the effective cost per kilogram of catalyst usage becomes negligible over multiple batches. Moreover, the use of common alcohols and hydrogen gas as reagents ensures a stable and secure supply chain, mitigating risks associated with the sourcing of specialized or regulated chemicals. This stability is crucial for long-term production planning and contract fulfillment in the pharmaceutical industry.
- Cost Reduction in Manufacturing: The elimination of stoichiometric reducing agents like lithium aluminum hydride represents a massive saving in reagent costs, as these materials are not only expensive but also generate significant amounts of hazardous waste that require costly disposal. Additionally, the high yield of the first step, often exceeding 80 percent, minimizes the loss of valuable starting materials, thereby improving the overall material throughput. The simplified workup procedure, which avoids complex chromatographic separations required for removing malonate byproducts, reduces labor hours and solvent consumption, contributing to a leaner and more cost-effective manufacturing operation.
- Enhanced Supply Chain Reliability: The raw materials for this process, specifically 2-alkylacroleins and various alcohols, are commodity chemicals produced by numerous global suppliers, ensuring high availability and competitive pricing. This diversification of supply sources reduces the risk of production stoppages due to single-source dependencies. Furthermore, the mild reaction conditions allow for the use of standard stainless steel reactors without the need for exotic alloys or specialized lining, making the technology easily transferable to existing manufacturing facilities without significant capital investment in new infrastructure.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from laboratory to pilot scales with consistent results. The absence of heavy metal contaminants from stoichiometric reductants simplifies the purification of the final API intermediate, ensuring compliance with strict regulatory limits on residual metals. The recyclable nature of the catalyst aligns with green chemistry principles, significantly reducing the E-factor (mass of waste per mass of product) of the synthesis. This environmental benefit is increasingly important for meeting corporate sustainability goals and adhering to tightening environmental regulations in major chemical production hubs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and technical disclosures within the patent documentation to provide clarity on process capabilities and limitations. Understanding these details is vital for technical teams evaluating the feasibility of adopting this route for their specific product portfolios.
Q: What are the primary advantages of this new synthesis method over the traditional diethyl malonate route?
A: The new method eliminates the formation of difficult-to-separate diethyl 2,2-di(alkyl)malonate byproducts and avoids the use of expensive stoichiometric reducing agents like lithium aluminum hydride, resulting in significantly lower production costs and higher total yields exceeding 70%.
Q: How is the novel phthalimide catalyst recovered and reused?
A: The catalyst features ester groups that ensure solubility during reaction; post-reaction, acid or base hydrolysis converts these esters into insoluble carboxylic acids, allowing for simple filtration and subsequent re-esterification to regenerate the active catalyst.
Q: What purity levels can be achieved with this process?
A: Through standard purification techniques such as extraction, distillation, or crystallization, the final 2-alkyl-1,3-propanediol product can consistently achieve purity levels greater than 98%, meeting stringent pharmaceutical intermediate specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Alkyl-1,3-Propanediol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes for high-value intermediates like 2-alkyl-1,3-propanediol. Our team of expert chemists has extensively evaluated this patented technology and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovation to the global market. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. Our facility is equipped to handle the specific hydrogenation and deprotection steps safely and efficiently, ensuring a consistent supply of high-quality intermediates for your downstream applications.
We invite you to collaborate with us to leverage this cost-effective synthesis method for your projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to contact us directly to obtain specific COA data for our pilot batches and to discuss route feasibility assessments for your unique molecular targets. Let us help you optimize your supply chain and reduce your manufacturing costs with our advanced chemical solutions.
