Revolutionizing Polyimide Precursors: A Two-Pot Strategy for Thioether Dianhydride Manufacturing
Introduction to Advanced Thioether Dianhydride Technology
The landscape of high-performance polymer manufacturing is undergoing a significant transformation driven by the need for more efficient and sustainable synthetic routes for critical monomers. Central to this evolution is the production of thioether dianhydride, a pivotal building block for polythioetherimides known for their exceptional thermomechanical stability and solubility. As detailed in patent CN102659728A, a groundbreaking methodology has emerged that challenges the status quo of multi-step synthesis. This innovation leverages a telescoped "two-pot" strategy that consolidates what were traditionally four distinct reaction stages into a seamless, continuous process. By integrating anhydride end-group protection directly with the sulfur coupling event, and subsequently merging hydrolysis with dehydration, this approach drastically minimizes unit operations. The structural integrity of the target molecule, characterized by the sulfur bridge linking two phthalic anhydride moieties, is preserved with high fidelity throughout this streamlined workflow.
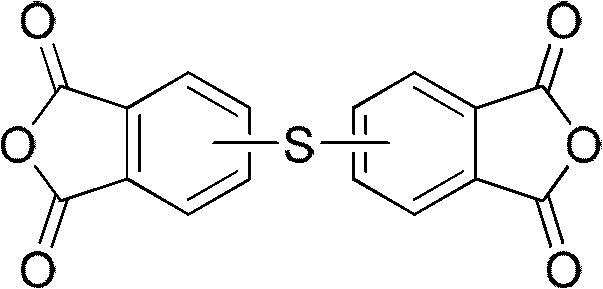
For R&D directors and process engineers, the implications of this patent extend far beyond simple yield improvements; it represents a fundamental shift in how we approach complex aromatic substitution reactions. The ability to manipulate the substitution pattern of the sulfur atom on the benzene ring—allowing for 3,3', 4,4', or mixed 3,4' configurations—provides unparalleled flexibility in tuning the final polymer properties. Whether the goal is to enhance the glass transition temperature or improve melt processability, controlling the isomeric purity at the monomer stage is paramount. This patent provides the blueprint for achieving such control while simultaneously addressing the economic and environmental pressures facing modern chemical manufacturing. As a reliable supplier of specialty chemical intermediates, understanding these mechanistic nuances is essential for delivering value to downstream polymer producers who demand both consistency and cost-efficiency in their supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thioether dianhydride has been plagued by inefficiencies inherent to linear, multi-step batch processing. Traditional protocols, such as those cited in earlier US patents, typically mandate a rigorous four-step sequence: initial protection of the anhydride group, followed by a separate coupling reaction, then a distinct hydrolysis and acidification phase, and finally, a standalone dehydration step to regenerate the anhydride functionality. Each of these discrete stages necessitates comprehensive post-processing workups, including filtration, extensive washing, and drying of intermediate solids. These repetitive isolation procedures are not merely time-consuming; they are the primary drivers of yield erosion, as mechanical losses accumulate with every transfer and purification event. Furthermore, the reliance on stoichiometric amounts of reagents without effective recovery mechanisms leads to inflated raw material costs and substantial waste generation. The cumulative effect is a process that is economically burdensome and environmentally taxing, struggling to meet the stringent efficiency metrics required for large-scale commercial production of high-purity electronic or aerospace grade materials.
The Novel Approach
In stark contrast, the methodology disclosed in CN102659728A introduces a paradigm shift through the concept of "one-pot" telescoping within a two-vessel framework. This innovative route utilizes readily available starting materials, specifically 3-chlorophthalic anhydride or 4-chlorophthalic anhydride, which serve as the foundational scaffolds for the molecular architecture.
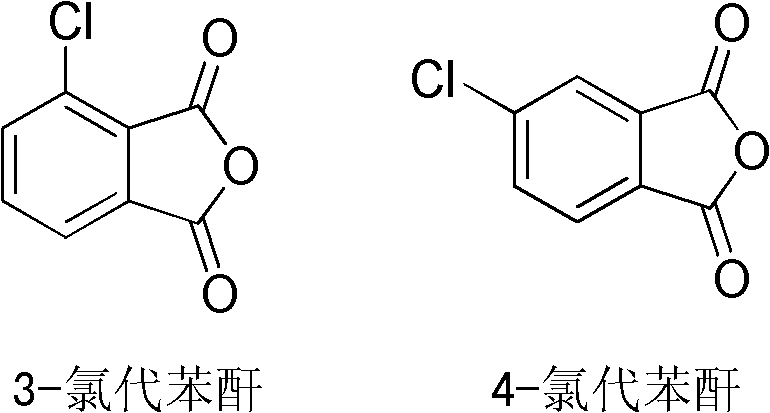
By reacting these chlorinated precursors with equimolar amounts of aniline in a carefully selected binary solvent system comprising polar aprotic and benzene-type solvents, the process achieves simultaneous protection and coupling in the "first pot." This eliminates the need to isolate the protected intermediate before introducing the sulfur source. Subsequently, the "second pot" operation seamlessly combines the hydrolysis of the imide protective groups with the dehydration of the resulting tetraacid. This consolidation means that the intermediate thioether tetraacid does not need to be isolated, dried, or purified before cyclization. The result is a dramatic reduction in cycle time and a significant enhancement in overall throughput. For procurement managers, this translates to a leaner manufacturing footprint where capital expenditure on equipment and operational expenditure on labor and energy are substantially optimized without compromising the chemical integrity of the final product.
Mechanistic Insights into Aniline-Mediated Protection and Sulfur Coupling
The core chemical ingenuity of this process lies in the dual role of aniline as a transient protecting group and the strategic selection of solvent systems to facilitate nucleophilic aromatic substitution. In the initial phase, aniline reacts with the chlorophthalic anhydride to form an N-phenyl phthalimide derivative. This transformation is critical because it masks the reactive anhydride carbonyls, preventing them from participating in unwanted side reactions or premature polymerization during the harsh conditions required for the subsequent sulfur coupling. The reaction is conducted at elevated temperatures ranging from 100°C to 200°C, ensuring complete conversion while maintaining the solubility of the intermediates. Following this protection step, a sulfur-containing coupling agent, such as sodium sulfide or elemental sulfur, is introduced directly into the same reaction vessel. The nucleophilic sulfur species attacks the chloro-substituted aromatic ring, displacing the chloride ion and forming the crucial carbon-sulfur-carbon bridge. The presence of the polar aprotic solvent, such as NMP or DMF, stabilizes the transition state and enhances the nucleophilicity of the sulfur anion, driving the reaction to completion with high selectivity.
Following the coupling, the transition to the "second pot" involves a base-mediated hydrolysis that serves a dual purpose: regenerating the carboxylic acid groups and releasing the aniline protecting group. This step is particularly elegant from a green chemistry perspective because the liberated aniline can be recovered and recycled, effectively closing the loop on one of the major reagent costs. Once the tetraacid is formed via acidification, the addition of a water-carrying agent like xylene or ortho-dichlorobenzene facilitates azeotropic distillation. Heating the mixture to 130°C to 200°C drives the dehydration equilibrium forward, cyclizing the tetraacid back into the thermodynamically stable dianhydride form. This mechanism ensures that the final product possesses the requisite purity for high-performance applications, as the telescoped nature of the reaction minimizes exposure to atmospheric moisture and contaminants that could degrade the anhydride rings. For quality control teams, this robust mechanism offers a predictable impurity profile, simplifying the analytical validation required for regulatory compliance in sensitive industries.
How to Synthesize Thioether Dianhydride Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize the benefits of the telescoped design. The protocol begins with the preparation of the reaction mixture in the first vessel, where stoichiometry and temperature ramps are critical for avoiding the formation of oligomeric byproducts. Detailed standard operating procedures regarding solvent ratios, specifically the volume ratio of polar aprotic to benzene solvents which can range from 1:20 to 20:1, must be strictly adhered to ensure optimal solubility throughout the temperature cycle. The subsequent addition of the sulfur coupling agent must be timed precisely after the initial protection phase is confirmed complete to prevent competitive side reactions. Following the coupling, the transfer to the hydrolysis phase involves careful pH management to ensure quantitative recovery of the aniline while fully converting the imide to the acid. For scale-up engineers, understanding these thermal and chemical boundaries is essential for designing reactors that can handle the exothermic nature of the coupling and the high-temperature requirements of the dehydration step safely and efficiently.
- First Pot Reaction: Mix chlorophthalic anhydride with aniline in a polar aprotic/benzene solvent system. Heat to 100-200°C for protection and coupling, then add a sulfur source (e.g., Na2S) to form N,N'-disubstituted thioetherimide.
- Workup: Precipitate the intermediate imide by pouring into an acidic or alcoholic solution, followed by filtration and washing to remove impurities before the second stage.
- Second Pot Reaction: Hydrolyze the imide in alkaline solution with heat to recover aniline. Acidify to form tetraacid, then dehydrate using a water-carrying agent (e.g., xylene) at 130-200°C to yield the final dianhydride.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented two-pot methodology offers profound advantages that resonate deeply with procurement strategies focused on cost reduction and supply chain resilience. The most immediate impact is seen in the drastic simplification of the manufacturing workflow. By collapsing four discrete unit operations into two, the process inherently reduces the consumption of utilities such as steam for heating and cooling water for condensation. This energy efficiency directly correlates to a lower cost of goods sold (COGS), allowing suppliers to offer more competitive pricing structures without sacrificing margin. Furthermore, the elimination of intermediate isolation steps means that there is significantly less handling of solid materials, which reduces labor costs and minimizes the risk of cross-contamination between batches. For supply chain heads, this streamlined process translates to shorter lead times and increased production capacity within existing facility footprints, enabling a more agile response to fluctuating market demands for polyimide precursors.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the reduction in processing steps and the recovery of valuable reagents. Unlike traditional methods that consume stoichiometric amounts of protecting groups, this novel approach allows for the recovery and reuse of aniline, effectively lowering the net raw material consumption per kilogram of product. Additionally, the ability to recycle solvents such as toluene and xylene, which are used as water-carrying agents and co-solvents, further diminishes the operational expenditure associated with waste disposal and fresh solvent procurement. The cumulative effect of these efficiencies is a substantial reduction in the overall manufacturing cost, making high-purity thioether dianhydride more accessible for cost-sensitive applications in the electronics and automotive sectors.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by complex processes that have multiple points of failure. By reducing the number of discrete steps and intermediate handlings, the risk of batch failures due to human error or equipment malfunction is significantly mitigated. The robustness of the "one-pot" coupling and "one-pot" cyclization ensures a more consistent output quality, reducing the need for rework or off-spec inventory management. Moreover, the use of commodity chemicals like chlorophthalic anhydrides and aniline as feedstocks ensures that the supply chain is not dependent on exotic or scarce reagents, thereby insulating the production schedule from upstream volatility. This reliability is crucial for long-term contracts with major polymer manufacturers who require guaranteed delivery schedules to maintain their own production lines.
- Scalability and Environmental Compliance: As regulatory pressures regarding industrial waste intensify, the environmental profile of a chemical process becomes a key differentiator. This method generates significantly less "three wastes" (wastewater, waste gas, and solid waste) compared to the prior art, primarily due to the reduced number of washing and filtration steps. The minimized solvent usage and the potential for closed-loop solvent recovery systems align perfectly with modern green chemistry principles and environmental, social, and governance (ESG) goals. For facilities operating under strict environmental permits, this technology offers a pathway to increase production volume without proportionally increasing waste discharge, thus facilitating easier permitting for capacity expansion and ensuring long-term operational compliance in regulated markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding the nuances of isomer control, yield optimization, and waste management is critical for stakeholders evaluating this technology for integration into their existing portfolios. We encourage technical teams to review these details closely to assess the feasibility of adopting this streamlined approach for their specific manufacturing requirements.
Q: How does the new two-pot method improve yield compared to traditional four-step processes?
A: The patented telescoped approach eliminates intermediate isolation steps, reducing mechanical losses. Experimental data shows total yields improving from approximately 56% in conventional methods to over 73% with this novel strategy.
Q: Can the aniline protecting group be recovered during the synthesis?
A: Yes, the process is designed for sustainability. During the alkaline hydrolysis step in the second pot, the aniline protecting group is cleaved and can be recovered and recycled, significantly lowering raw material costs.
Q: What isomers of thioether dianhydride can be produced using this method?
A: By adjusting the ratio of 3-chlorophthalic anhydride to 4-chlorophthalic anhydride in the feedstock, manufacturers can selectively produce 3,3'-, 4,4'-, or mixed isomer thioether dianhydrides to suit specific polymer property requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thioether Dianhydride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team specializes in the scale-up of complex fine chemical intermediates, bridging the gap between patent concepts and industrial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies demonstrated in the lab are faithfully reproduced at the plant level. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs equipped with state-of-the-art analytical instrumentation, guaranteeing that every batch of thioether dianhydride meets the exacting standards required for high-performance polyimide applications.
We invite you to collaborate with us to unlock the full potential of this advanced synthesis route. Whether you are looking to optimize an existing supply chain or develop a new grade of polymer material, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our engineering excellence can drive value and efficiency in your operations.
