Optimizing Triazole Antifungal Intermediate Production for Global Pharmaceutical Supply Chains
Optimizing Triazole Antifungal Intermediate Production for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective routes for producing high-value active pharmaceutical ingredients (APIs) and their precursors. Patent CN1026788C discloses a sophisticated preparation method for a class of triazole antifungal agents characterized by the general Formula I. These compounds, specifically 2-aryl-3-(3-halopyridin-4-yl or 5-halopyrimidin-4-yl)-1-(1H-1,2,4-triazol-1-yl)alkane-2-ol derivatives, represent a critical segment in the antifungal market due to their potent activity against clinically significant fungi, including Aspergillus species. The invention provides a comprehensive chemical framework where R represents a phenyl group substituted with halogens or trifluoromethyl groups, R1 is a C1-4 alkyl group, and the heterocyclic core varies between pyridine and pyrimidine structures. This technical disclosure is pivotal for manufacturers aiming to secure a reliable pharmaceutical intermediate supplier capable of delivering complex chiral molecules with stringent purity specifications.
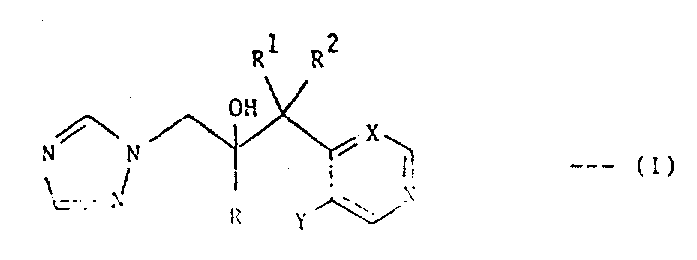
Beyond the structural definition, the patent emphasizes the unexpected pharmacokinetic properties of these compounds, particularly their long half-life (t1/2 value), which translates to superior therapeutic efficacy in treating systemic fungal infections. For procurement managers and supply chain heads, understanding the underlying chemistry is essential for evaluating the feasibility of commercial scale-up. The described methodology avoids reliance on scarce natural products or overly complex biocatalytic steps, instead utilizing well-established organometallic chemistry that can be adapted for large-scale production. By leveraging this intellectual property, chemical manufacturers can offer cost reduction in API manufacturing through streamlined synthetic routes that minimize the number of isolation steps and maximize overall yield efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis pathways for triazole antifungal intermediates often suffer from significant drawbacks related to regioselectivity and stereochemical control. Conventional alkylation methods may lead to mixtures of positional isomers when functionalizing the pyridine or pyrimidine rings, necessitating expensive and time-consuming purification processes such as preparative HPLC to separate the desired isomer from byproducts. Furthermore, older methods might rely on harsh reaction conditions that degrade sensitive functional groups, such as the triazole ring or halogen substituents, leading to reduced yields and increased waste generation. The lack of precise control over the formation of chiral centers at the C2 and C3 positions often results in racemic mixtures, requiring additional resolution steps that drastically increase the cost of goods sold (COGS) and extend the production lead time. These inefficiencies pose a substantial risk to supply chain continuity, especially when demand for high-purity antifungal agents surges during epidemic outbreaks.
The Novel Approach
The novel approach detailed in the patent overcomes these challenges by employing a directed ortho-metalation strategy followed by nucleophilic addition. This method utilizes strong, non-nucleophilic bases like lithium diisopropylamide (LDA) or sodium bis(trimethylsilyl)amide to selectively deprotonate the alkyl-substituted heterocycle at the position adjacent to the ring nitrogen. This generates a highly reactive organometallic intermediate that reacts cleanly with the triazole-substituted ketone. The reaction is conducted at low temperatures, typically between -80°C and -50°C, which suppresses side reactions and ensures high regioselectivity. This precision allows for the direct construction of the carbon-carbon bond connecting the heterocyclic core to the chiral alcohol backbone without the need for protecting groups or multiple activation steps. Consequently, this route offers a more direct path to the target molecule, significantly simplifying the process flow and enhancing the overall economic viability of the manufacturing process.
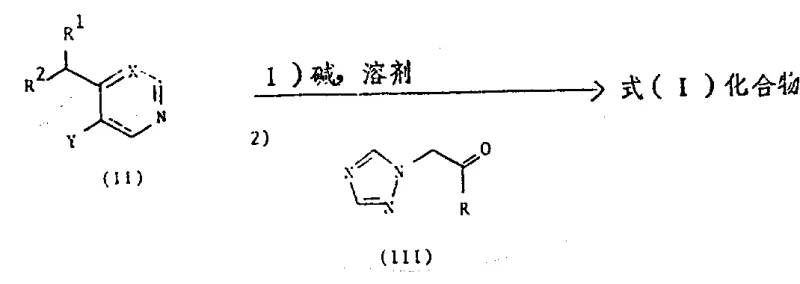
Mechanistic Insights into Organolithium-Mediated Coupling
The core of this synthetic strategy lies in the generation and utilization of the organolithium intermediate derived from the 4-ethyl-pyridine or pyrimidine precursor. When treated with a stoichiometric amount of a strong base like LDA in an aprotic solvent such as tetrahydrofuran (THF) or toluene, the acidic proton at the alpha-position of the ethyl group is abstracted. This deprotonation occurs rapidly at cryogenic temperatures (-70°C) to form a stabilized carbanion species. The stability of this intermediate is crucial; it must persist long enough to react with the electrophile but not decompose via elimination or other degradation pathways. The subsequent addition of the ketone, specifically a 1-(substituted phenyl)-2-(1H-1,2,4-triazol-1-yl)ethanone, triggers a nucleophilic attack on the carbonyl carbon. This step forms the new carbon-carbon bond and generates the alkoxide intermediate, which upon aqueous workup yields the target tertiary alcohol. The mechanism ensures that the stereochemistry at the newly formed centers is dictated by the approach of the nucleophile, allowing for the potential formation of specific diastereomers depending on the reaction conditions and the nature of the substituents.
Impurity control is inherently built into this mechanism through the choice of reagents and temperature profiles. By maintaining the reaction under an inert atmosphere (nitrogen or argon) and strictly controlling the temperature, the formation of homocoupling byproducts or protonated starting materials is minimized. The use of specific bases like LDA ensures that deprotonation occurs exclusively at the desired site, preventing random functionalization of the aromatic rings. Furthermore, the patent describes alternative routes involving epoxide intermediates (Formula IV), which can be opened by triazole salts to achieve the same structural motif. This mechanistic flexibility allows manufacturers to choose the most cost-effective pathway based on raw material availability. For instance, if the ketone is expensive or unstable, the epoxide route offers a viable alternative that still delivers the high-purity triazole antifungal intermediates required for downstream API synthesis.
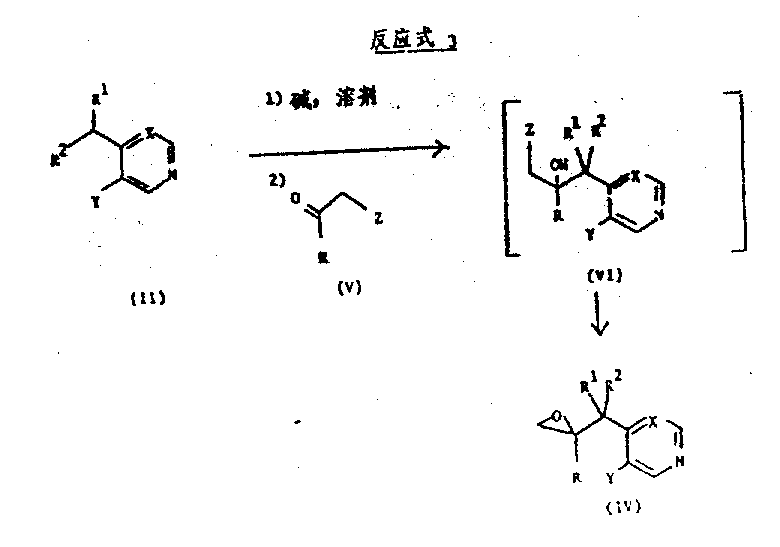
How to Synthesize Triazole Antifungal Intermediates Efficiently
Implementing this synthesis requires careful attention to reaction parameters to ensure reproducibility and safety on a commercial scale. The process begins with the preparation of the organometallic reagent, followed by the controlled addition of the electrophile. Detailed operational guidelines are essential for maintaining the integrity of the sensitive intermediates involved. The following section outlines the standardized synthesis steps derived from the patent examples, providing a clear roadmap for technical teams to follow during process development and scale-up activities.
- Deprotonate the heterocyclic starting material (Formula II) using a strong base like lithium diisopropylamide (LDA) or sodium bis(trimethylsilyl)amide at temperatures between -80°C and -50°C under inert atmosphere.
- React the resulting organometallic intermediate in situ with the corresponding triazole-substituted ketone (Formula III) to form the carbon-carbon bond.
- Quench the reaction mixture with water or dilute acid, extract with organic solvents like ethyl acetate, and purify the crude product via silica gel chromatography or recrystallization to obtain the target alcohol derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the significant simplification of the manufacturing process, which directly correlates to reduced operational costs and improved supply security. By utilizing a convergent synthesis strategy where two complex fragments are joined in a single key step, the total number of unit operations is minimized. This reduction in process complexity lowers the capital expenditure required for plant equipment and decreases the labor hours needed per batch, resulting in substantial cost savings in pharmaceutical intermediate manufacturing. Additionally, the high selectivity of the reaction reduces the burden on purification systems, meaning less solvent consumption and lower waste disposal costs, aligning with modern environmental compliance standards.
- Cost Reduction in Manufacturing: The elimination of multiple protection and deprotection steps, which are common in less efficient synthetic routes, leads to a drastic reduction in raw material consumption and processing time. The use of readily available starting materials, such as substituted ethyl pyridines and triazole ketones, ensures that the supply chain is not vulnerable to bottlenecks associated with exotic reagents. Furthermore, the ability to recycle solvents like THF and ethyl acetate, which are used in the extraction and purification stages, contributes to a leaner cost structure. The high yield reported in the examples suggests that the process is robust, minimizing the loss of valuable intermediates and maximizing the output per batch, which is critical for maintaining competitive pricing in the global market.
- Enhanced Supply Chain Reliability: The versatility of the chemistry allows for the production of a wide range of analogues by simply varying the substituents on the phenyl ring or the heterocyclic core. This flexibility means that a single manufacturing line can be adapted to produce different intermediates for various antifungal APIs, reducing the need for dedicated infrastructure for each product. The robustness of the organolithium chemistry, when properly managed, ensures consistent quality and batch-to-batch reproducibility, which is paramount for meeting the rigorous quality agreements of multinational pharmaceutical clients. By securing a supply of these key intermediates through a proven and scalable method, companies can mitigate the risk of production delays caused by supplier failures or quality deviations.
- Scalability and Environmental Compliance: The reaction conditions, while requiring low temperatures, are compatible with standard industrial cooling systems and do not necessitate cryogenic fluids that are difficult to handle on a multi-ton scale. The workup procedures involve standard liquid-liquid extractions and crystallizations, which are easily scalable from kilogram to metric ton quantities. Moreover, the process avoids the use of heavy metal catalysts, which often require complex removal steps to meet residual metal specifications in the final drug product. This absence of heavy metals simplifies the purification train and reduces the environmental footprint of the manufacturing process, making it easier to obtain regulatory approval and maintain sustainable operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these triazole antifungal intermediates. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals seeking to implement this technology.
Q: What are the critical reaction conditions for the lithiation step in this process?
A: The lithiation step requires strict temperature control, typically between -80°C and -50°C, preferably around -70°C, under an inert atmosphere such as nitrogen or argon. Suitable bases include lithium diisopropylamide (LDA) or sodium bis(trimethylsilyl)amide to ensure regioselective deprotonation without side reactions.
Q: How does this synthesis method address stereochemical purity concerns?
A: The process generates compounds with chiral centers, existing as enantiomers or diastereomers. The patent describes methods for resolution, such as fractional crystallization of diastereomeric salts formed with optically active acids like 10-camphorsulfonic acid, or separation via HPLC using chiral supports, ensuring high optical purity for the final API.
Q: Can this route be adapted for different halogen substituents on the phenyl ring?
A: Yes, the synthetic route is highly versatile regarding the 'R' group. It accommodates phenyl rings substituted with one to three halogens (F, Cl, Br, I) or trifluoromethyl groups. Preferred embodiments utilize 2-fluorophenyl, 2,4-difluorophenyl, or 2-chlorophenyl groups, demonstrating broad substrate tolerance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triazole Antifungal Intermediates Supplier
At NINGBO INNO PHARMCHEM, we understand the critical role that high-quality intermediates play in the successful development and commercialization of life-saving antifungal medications. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of specific stereoisomers or large-scale production of racemic mixtures, our flexible manufacturing capabilities allow us to tailor our services to your specific project requirements, guaranteeing a seamless integration into your supply chain.
We invite you to contact our technical procurement team to discuss how our advanced synthesis capabilities can support your upcoming projects. By partnering with us, you gain access to a Customized Cost-Saving Analysis that identifies opportunities to optimize your current sourcing strategy. We encourage you to request specific COA data and route feasibility assessments for the triazole antifungal intermediates discussed in this report. Let us demonstrate our commitment to quality and efficiency, helping you bring effective antifungal therapies to patients faster and more economically.
