Advanced Synthesis of Fluorine-Containing Isopropenyl Zinc Reagents for Pharmaceutical Manufacturing
Advanced Synthesis of Fluorine-Containing Isopropenyl Zinc Reagents for Pharmaceutical Manufacturing
The pharmaceutical and agrochemical industries are increasingly reliant on fluorinated organic compounds due to their unique metabolic stability and bioavailability profiles. A pivotal advancement in this domain is documented in patent CN1027539C, which details a robust preparation method for fluorine-containing isopropenyl zinc reagents and their derivatives. This technology introduces a metallized fluoro-building block that overcomes historical limitations in introducing perfluoroalkyl groups into organic molecules. By utilizing activated zinc species in conjunction with nitrogen-containing organic ligands, manufacturers can now access stable organozinc intermediates that serve as precursors for high-value perfluoroalkyl-substituted styrenes and conjugated dienes. This innovation represents a significant leap forward for reliable API intermediate suppliers seeking to streamline the synthesis of complex fluorinated architectures without resorting to hazardous or energy-intensive processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of fluorinated organic building blocks relied heavily on methods such as metal-catalyzed hydroformylation of trifluoropropene. While effective in theory, these conventional approaches often necessitate specialized reaction units capable of withstanding high pressures and specific gas handling requirements, creating substantial barriers to entry for many fine chemical manufacturers. Furthermore, alternative routes involving bromination and dehydrobromination to generate starting materials like 2-bromo-trifluoropropene often suffer from limited scalability and inconsistent yields when attempting to introduce the fluorinated moiety into complex molecular scaffolds. The lack of versatile, stable metallated reagents has frequently forced R&D teams to compromise on route efficiency, leading to multi-step sequences that inflate costs and extend lead times for high-purity pharmaceutical intermediates.
The Novel Approach
The methodology outlined in the patent data presents a transformative solution by leveraging activated zinc metal to directly metallize fluorine-containing allylic halides. This novel approach eliminates the need for extreme pressure equipment, allowing reactions to proceed in standard polar solvents like tetrahydrofuran (THF) or dimethoxyethane (DME) under mild thermal conditions ranging from 40°C to 80°C. By forming a stable carbon-zinc bond, the resulting reagent acts as a nucleophilic partner that can smoothly engage in cross-coupling reactions with aryl and alkenyl halides. This shift from gas-phase hydroformylation to solution-phase organometallic chemistry drastically simplifies the operational complexity, offering a pathway for cost reduction in electronic chemical manufacturing and pharmaceutical production alike by utilizing widely available reactor infrastructure.
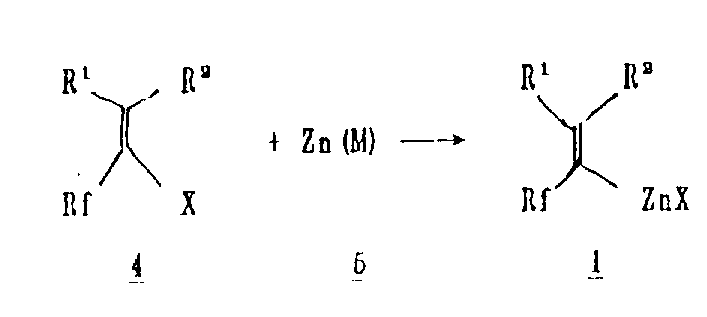
Mechanistic Insights into Zinc-Mediated Fluorination and Cross-Coupling
The core of this technology lies in the generation of the fluorine-containing pseudoallyl zinc species, represented generally by Formula 1 in the patent documentation. The mechanism initiates with the activation of zinc metal, often enhanced by additives such as silver, copper, or mercury, or surface activators like iodoethane. This activated zinc inserts into the carbon-halogen bond of the fluorinated precursor, such as 2-bromo-trifluoropropene, to form the organozinc intermediate. Crucially, the presence of electron-withdrawing fluorine atoms adjacent to the double bond could typically destabilize such species; however, the specific electronic environment created by the isopropenyl structure allows for sufficient stability to permit isolation or in-situ usage. The ability to tune the steric and electronic properties by varying the Rf group from trifluoromethyl to longer perfluoroalkyl chains provides chemists with a versatile toolkit for molecular design.
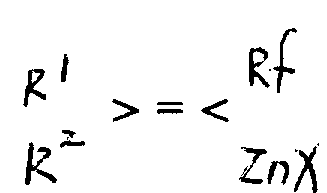
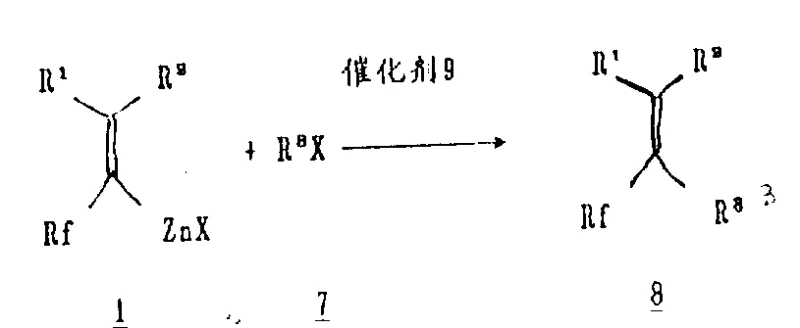
To further enhance stability and handling, the process allows for the complexation of the zinc reagent with nitrogenous organic ligands, as depicted in Formulas 2 and 3. Ligands such as tetramethylethylenediamine (TMEDA) or bipyridine coordinate with the zinc atom through lone pair electrons, forming chelated complexes that exhibit remarkable thermostability. These complexes can remain intact at room temperature for months or withstand reflux conditions without decomposition, a feature that is critical for supply chain continuity and batch-to-batch consistency. When subjected to transition metal catalysis, typically using palladium or nickel complexes like tetrakis(triphenylphosphine)palladium, these zinc reagents undergo transmetallation. This step transfers the fluorinated alkyl group to the palladium center, followed by reductive elimination with the aryl or alkenyl halide partner to forge the new carbon-carbon bond with high stereospecificity, preserving the geometry of the double bond in the final product.
How to Synthesize Fluorine-Containing Isopropenyl Zinc Reagents Efficiently
The synthesis of these valuable building blocks is designed to be operationally simple, requiring standard laboratory or plant equipment rather than specialized high-pressure vessels. The process begins with the activation of zinc dust, followed by the controlled addition of the fluorinated halide in a polar aprotic solvent. The reaction temperature is carefully maintained between 40°C and 80°C to optimize conversion while minimizing side reactions. For applications requiring extended storage or transport of the reagent, the addition of a nitrogenous ligand is recommended to form the stable complex. Detailed standardized synthetic steps see the guide below for specific molar ratios and workup procedures that ensure maximum yield and purity.
- Activate zinc metal (Zn(M)) using agents like iodoethane or trimethylchlorosilane to enhance reactivity.
- React fluorine-containing allylic halides with activated zinc in polar solvents like THF or DME at 40-80°C.
- Optionally add nitrogenous ligands such as TMEDA to form stable complexes before proceeding to palladium-catalyzed coupling.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this zinc-mediated fluorination technology offers substantial strategic benefits regarding raw material sourcing and operational expenditure. The primary starting materials, such as trifluoropropene derivatives, are industrially available organofluorine compounds, reducing dependency on exotic or custom-synthesized precursors. By shifting away from high-pressure hydroformylation processes, facilities can utilize existing reaction vessels, thereby avoiding significant capital expenditure on new infrastructure. This compatibility with standard equipment translates directly into lower barrier-to-entry costs and faster implementation timelines for scaling up production of complex fluorinated intermediates.
- Cost Reduction in Manufacturing: The elimination of specialized high-pressure reaction units significantly lowers the capital intensity of the production process. Furthermore, the use of activated zinc, which is relatively inexpensive compared to other organometallic reagents like organoboron or organostannane species, contributes to a leaner bill of materials. The high conversion rates and the ability to recycle solvent systems further drive down the unit cost of goods sold, making the commercial scale-up of complex polymer additives and pharmaceutical intermediates more economically viable.
- Enhanced Supply Chain Reliability: The formation of stable zinc-ligand complexes addresses a critical pain point in the supply of reactive intermediates. Unlike many organometallic species that degrade rapidly upon exposure to air or moisture, these complexes demonstrate robust thermostability, allowing for safer storage and transportation. This stability reduces the risk of batch failure due to reagent degradation, ensuring a consistent supply of high-quality raw materials for downstream synthesis. Consequently, manufacturers can maintain tighter inventory control and reduce the safety stock levels required to buffer against supply disruptions.
- Scalability and Environmental Compliance: The reaction conditions are inherently safer, operating at atmospheric pressure and moderate temperatures, which simplifies hazard management and waste treatment protocols. The process avoids the generation of toxic tin byproducts often associated with Stille couplings, aligning better with modern green chemistry principles and environmental regulations. This cleaner profile facilitates easier regulatory approval for new drug applications and reduces the environmental footprint of the manufacturing site, supporting long-term sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the application and scope of this fluorination technology. These insights are derived directly from the experimental data and embodiments provided in the patent literature, offering clarity on reaction scope, catalyst selection, and product outcomes. Understanding these parameters is essential for process chemists evaluating the feasibility of integrating this route into their existing manufacturing workflows.
Q: What are the stability characteristics of the fluorine-containing pseudoallyl zinc complexes?
A: The complexes formed with nitrogenous ligands like TMEDA exhibit excellent thermostability, remaining intact at room temperature for months or under reflux at 80°C without decomposition, ensuring consistent reactive behavior.
Q: Can this method be used for stereoselective synthesis of diene compounds?
A: Yes, the reagent reacts stereospecifically with alkenyl halides under palladium or nickel catalysis to produce single E or Z-1,3-diene coupled products with high stereochemical fidelity.
Q: What types of catalysts are compatible with this cross-coupling reaction?
A: The process utilizes standard organic transition metal catalysts, specifically organic palladium compounds like tetrakis(triphenylphosphine)palladium or organic nickel compounds, facilitating mild reaction conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorine-Containing Isopropenyl Zinc Reagent Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced fluorination technologies play in the development of next-generation therapeutics and high-performance materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of fluorine-containing building blocks meets the exacting standards required by global pharmaceutical and agrochemical clients.
We invite you to collaborate with us to leverage this innovative zinc-mediated synthesis for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in organometallic chemistry can accelerate your development timeline while optimizing your overall production costs.
