Advanced Synthesis of 2-Ethyl-2-Methylpentanoic Acid for Pharmaceutical Quality Control
The rigorous control of related substances in Active Pharmaceutical Ingredients (APIs) is a cornerstone of modern pharmaceutical quality assurance, particularly for widely used antiepileptic drugs like Sodium Valproate. Patent CN116143586A introduces a groundbreaking preparation method for 2-ethyl-2-methylpentanoic acid, a critical impurity reference standard required for validating the purity of valproate bulk drugs. This innovation addresses long-standing challenges in the synthesis of complex branched fatty acids by replacing hazardous reduction steps and inefficient purification techniques with a streamlined, scalable protocol. By leveraging a cyanoacetate-based pathway, the technology ensures that manufacturers can produce high-purity reference materials essential for regulatory compliance and drug safety testing. The strategic shift away from traditional acetoacetate routes represents a significant evolution in fine chemical manufacturing, offering a robust solution for producing complex organic acids with superior structural fidelity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
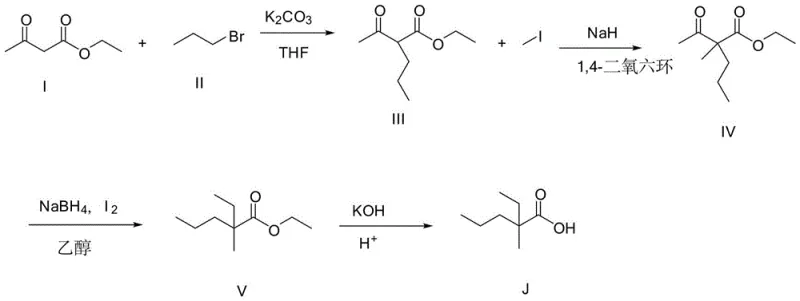
Traditional synthesis routes for 2-ethyl-2-methylpentanoic acid, such as those described in prior art patent CN112174799a, rely heavily on the alkylation of ethyl acetoacetate followed by reduction and hydrolysis. A major drawback of this conventional approach is the inevitable formation of dialkyl byproducts during the initial alkylation stage, which complicates downstream purification and drastically lowers the overall yield of the desired mono-alkylated intermediate. Furthermore, the reliance on sodium borohydride for the reduction step introduces significant safety hazards due to the potential for exothermic reactions and hydrogen gas evolution, requiring specialized containment and handling protocols. Perhaps most critically for industrial scalability, the final purification in these legacy methods often necessitates column chromatography, a technique that suffers from low equipment throughput, high solvent consumption, and difficulty in scaling to multi-kilogram batches. These factors collectively result in a process that is economically inefficient and operationally risky for large-scale commercial production.
The Novel Approach
In stark contrast, the novel methodology disclosed in CN116143586A utilizes a cyanoacetate precursor strategy that fundamentally redesigns the synthetic logic to enhance efficiency and safety. This innovative route employs sequential alkylation steps using methyl iodide and bromopropane, followed by a unique hydrolysis and decarboxylation sequence that avoids the use of dangerous reducing agents entirely. The process replaces the bottleneck of column chromatography with conventional extraction and rectification operations, which dramatically increases the output per unit volume of equipment and simplifies the isolation of high-purity intermediates. By integrating an alkali extraction step early in the synthesis, the method effectively removes unreacted starting materials before they can generate complex impurity profiles, ensuring a cleaner reaction stream. This approach not only mitigates safety risks associated with strong reducers but also aligns perfectly with green chemistry principles by reducing solvent waste and energy consumption.
Mechanistic Insights into Cyanoacetate Alkylation and Nitrous Acid Hydrolysis
The core of this synthetic breakthrough lies in the precise control of enolate chemistry during the alkylation of cyanoacetates, which serves as the foundation for building the quaternary carbon center. In the initial step, cyanoacetate is treated with a base such as sodium methoxide to generate a nucleophilic enolate, which then attacks bromopropane to form the mono-alkylated intermediate. The mechanistic elegance of this process is enhanced by a subsequent alkali wash using dilute sodium hydroxide, which selectively hydrolyzes and solubilizes any unreacted cyanoacetate (Compound A) into the aqueous phase, leaving the desired neutral ester (Compound B) in the organic layer. This selective extraction is crucial for preventing the carryover of starting materials that could lead to poly-alkylation side products in subsequent steps. The ability to purify the intermediate via simple phase separation rather than complex chromatography demonstrates a deep understanding of the physicochemical properties of the reaction components.
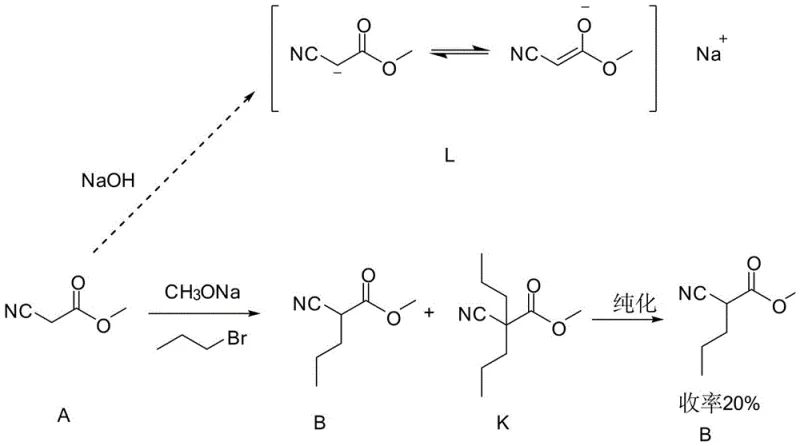
Further down the synthetic pathway, the conversion of the nitrile group to the carboxylic acid is achieved through a sophisticated hydrolysis protocol involving concentrated sulfuric acid and sodium nitrite. Unlike standard acidic hydrolysis which can be sluggish or require extreme temperatures, the presence of nitrous acid generated in situ facilitates the rapid and efficient conversion of the sterically hindered nitrile group. This step is critical for unlocking the final carboxylic acid functionality while maintaining the integrity of the sensitive branched alkyl chains. The reaction conditions are carefully optimized to proceed at moderate temperatures, minimizing the risk of thermal degradation or unwanted rearrangement of the carbon skeleton. This mechanistic pathway ensures that the final product retains the exact structural configuration required for its role as a reference standard, with impurity levels kept to an absolute minimum through the cumulative effect of the preceding purification stages.
How to Synthesize 2-Ethyl-2-Methylpentanoic Acid Efficiently
The synthesis of this valuable pharmaceutical intermediate requires a disciplined approach to reaction control and workup procedures to maximize yield and purity. The process begins with the careful alkylation of cyanoacetate, where temperature control and stoichiometry are paramount to preventing over-alkylation. Following the initial coupling, the reaction mixture undergoes a specialized extraction protocol to remove acidic impurities, followed by fractional distillation to isolate the key nitrile intermediate with high precision. Subsequent steps involve methylation, hydrolysis, and a thermal decarboxylation event that must be managed under reduced pressure to ensure clean conversion to the nitrile species. The final transformation involves a nitrous acid-mediated hydrolysis that converts the nitrile to the target acid, requiring precise pH control during the quench and extraction phases. For the complete standardized operating procedure and detailed reaction parameters, please refer to the guide below.
- Alkylation of cyanoacetate with bromopropane followed by base extraction to remove unreacted starting materials.
- Sequential methylation and hydrolysis to form the intermediate cyano-acid, followed by thermal decarboxylation.
- Final ethylation of the nitrile intermediate and hydrolysis using sulfuric acid and sodium nitrite to yield the target carboxylic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers transformative benefits in terms of cost structure and operational reliability. The elimination of column chromatography is a game-changer for manufacturing economics, as it removes a major bottleneck that typically limits batch size and extends production cycles significantly. By switching to distillation and extraction, the process allows for the use of standard reactor vessels and distillation columns, which are readily available in most chemical manufacturing facilities and do not require the specialized packing materials or vast quantities of elution solvents associated with chromatographic purification. This shift translates directly into a substantial reduction in operating expenses and a drastic simplification of the waste management profile, as the volume of organic solvent waste is minimized. Furthermore, the avoidance of hazardous reducing agents like sodium borohydride lowers the safety classification of the process, potentially reducing insurance premiums and simplifying regulatory compliance regarding hazardous material storage and handling.
- Cost Reduction in Manufacturing: The removal of chromatographic purification steps leads to a significant decrease in solvent consumption and labor costs associated with column packing and fraction collection. The use of commodity reagents such as bromopropane and cyanoacetate, combined with simple inorganic bases, ensures that raw material costs remain stable and predictable. Additionally, the higher throughput per unit volume of equipment means that the same facility can produce larger quantities of the intermediate in less time, effectively lowering the fixed cost allocation per kilogram of product. This economic efficiency makes the supply of high-purity reference standards more sustainable and affordable for downstream pharmaceutical applications.
- Enhanced Supply Chain Reliability: The reliance on widely available and stable chemical reagents reduces the risk of supply disruptions that can occur with specialized or hazardous catalysts. The robustness of the distillation-based purification ensures consistent product quality across different batches, which is critical for maintaining the trust of regulatory bodies and end-users. By simplifying the process flow, the manufacturer can respond more agilely to fluctuations in demand, scaling production up or down without the logistical complexities of managing large volumes of chromatography solvents. This stability is essential for long-term supply contracts where continuity of supply is as important as price.
- Scalability and Environmental Compliance: The transition from batch chromatography to continuous or semi-continuous distillation operations greatly enhances the scalability of the process from laboratory to commercial tonnage. This scalability is achieved without compromising the environmental footprint, as the reduction in solvent usage aligns with increasingly stringent global environmental regulations. The process generates less hazardous waste, simplifying disposal and treatment requirements, which further contributes to the overall sustainability of the manufacturing operation. This alignment with green chemistry principles positions the supplier favorably in markets where environmental stewardship is a key procurement criterion.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specialized pharmaceutical intermediate. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation to ensure accuracy and relevance. Understanding these details helps stakeholders make informed decisions regarding the integration of this material into their quality control workflows.
Q: Why is chromatography avoided in this new synthesis method?
A: Chromatography is avoided to significantly increase equipment throughput and reduce solvent consumption. The new method utilizes alkali extraction and rectification, which are more suitable for large-scale industrial production compared to column chromatography.
Q: What are the safety advantages of this route over prior art?
A: This route eliminates the use of strong reducing agents like sodium borohydride, which pose explosion and hydrogen gas risks. Instead, it uses safer reagents such as sodium hydride and sodium nitrite under controlled conditions.
Q: How is high purity achieved without complex purification?
A: High purity is achieved through a specific alkali extraction step that removes unreacted cyanoacetate early in the process, followed by precise fractional distillation which effectively separates the target intermediate from dialkyl byproducts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Ethyl-2-Methylpentanoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity impurity standards in the development and validation of safe pharmaceutical products. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistent quality. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of 2-ethyl-2-methylpentanoic acid meets the exacting standards required for analytical reference use. Our commitment to technical excellence allows us to deliver materials that support your regulatory filings and quality assurance protocols with confidence.
We invite you to contact our technical procurement team to discuss your specific requirements and explore how our advanced synthesis capabilities can support your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized manufacturing processes can reduce your overall procurement costs. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability to be your trusted partner in the supply of complex pharmaceutical intermediates.
