Scalable Total Synthesis of Concentricolide: A Technical Breakthrough for API Manufacturing
Introduction to Novel Concentricolide Synthesis Technology
The pharmaceutical industry continuously seeks robust synthetic pathways for bioactive natural products, particularly those exhibiting potent antiviral properties. Patent CN103159776A introduces a groundbreaking universal and effective total synthesis method for Concentricolide and its analogues, addressing critical bottlenecks in previous manufacturing approaches. This technology leverages a strategic Michael-Aldol reaction sequence starting from 1,3-acetone dicarboxylate compounds and alkynals to construct the complex 6H-1,7-dioxa-as-indacen-8-one skeleton. Unlike earlier methods that relied on inefficient extraction from fungal fermentation or low-yielding Diels-Alder reactions, this approach offers a streamlined chemical route capable of producing both racemic and chiral forms of the target molecule. For R&D directors and procurement specialists, this represents a significant opportunity to secure a reliable pharmaceutical intermediate supplier source for high-purity anti-HIV candidates without the supply chain volatility associated with biological extraction.
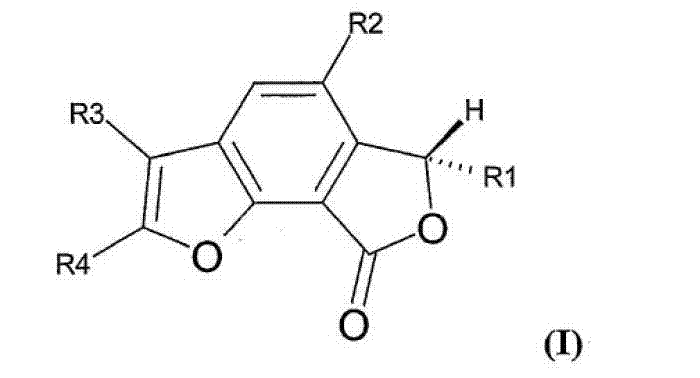
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of Concentricolide has been fraught with significant technical and logistical challenges that hinder commercial viability. Initial isolation methods depended entirely on the fermentation of the Ascomycete Daldinia concentrica, a process inherently limited by biological variability, seasonal factors, and extremely low natural abundance, resulting in insufficient quantities for extensive preclinical or clinical development. Furthermore, early synthetic attempts, such as the Diels-Alder reaction reported in 2009, suffered from extremely low yields and an inability to support large-scale preparation, rendering them economically unfeasible for industrial application. Subsequent methodologies, including those reported in 2011, often involved lengthy synthetic steps, harsh reaction conditions, and the utilization of toxic or explosive organic substances, which drastically increased operational costs and introduced severe safety hazards in a manufacturing environment. These legacy issues created a substantial barrier to entry for developing Concentricolide-based therapeutics, necessitating a more efficient and safer chemical strategy.
The Novel Approach
The methodology disclosed in CN103159776A fundamentally transforms the production landscape by establishing a concise, general, and safe synthetic route that bypasses the pitfalls of prior art. This novel approach initiates with a Michael-Aldol condensation to efficiently build the substituted phenyl core, followed by a series of controlled functional group transformations including phenolic protection, benzylic oxidation, and stereoselective reduction. Crucially, the construction of the benzofuranone lactone and the subsequent furan ring is achieved through Dieckmann condensation and Krapcho reactions under relatively mild alkaline conditions, avoiding the extreme temperatures and pressures of older methods. By utilizing commercially available starting materials like dimethyl 1,3-acetonedicarboxylate and various alkynals, this process ensures cost reduction in API manufacturing through simplified raw material sourcing and reduced waste generation. The ability to introduce chirality using specific catalytic reducing agents further enhances the value proposition, allowing for the production of specific enantiomers required for high-efficacy drug formulations.
Mechanistic Insights into Michael-Aldol Cyclization and Lactonization
The core of this synthetic innovation lies in the precise orchestration of the Michael-Aldol reaction and subsequent cyclization steps, which dictate the structural integrity and purity of the final product. The process begins with the deprotonation of the 1,3-propanedione carboxylate using strong bases such as sodium hydride or potassium carbonate, generating a nucleophilic enolate that attacks the electrophilic alkynal. This cascade reaction effectively constructs the tetrasubstituted phenolic compound with high regioselectivity, minimizing the formation of unwanted isomeric byproducts that often complicate downstream purification. Following phenolic protection and oxidation to the benzylic ketone, the stereochemistry is established during the reduction phase; the use of chiral reagents like (S)-Me-CBS-oxazaborolidine allows for the asymmetric induction necessary to create the specific R or S configuration at the chiral center, a critical parameter for biological activity. This level of control is essential for R&D teams aiming to optimize the structure-activity relationship (SAR) of Concentricolide analogues without the noise of racemic mixtures.
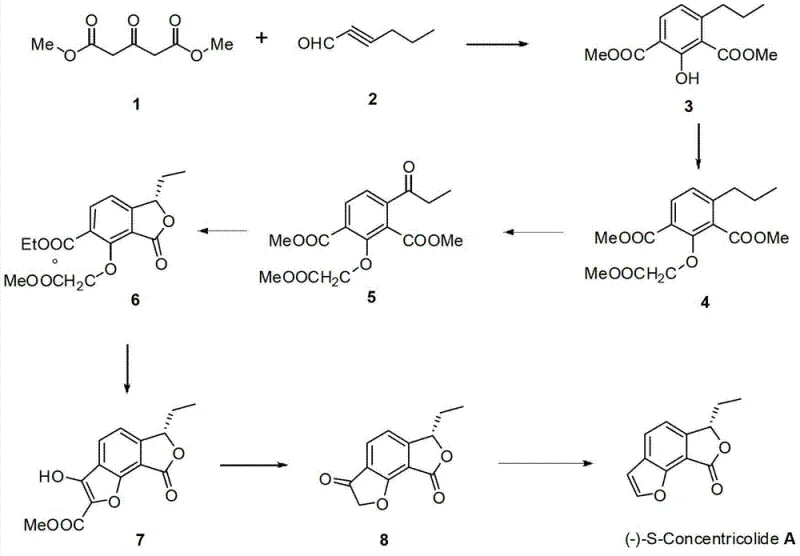
Furthermore, the impurity profile is tightly managed through the specific choice of oxidants and reducing agents in the later stages of the synthesis. The transition from the protected intermediate to the benzofuranone lactone involves a careful reduction step where reagents like sodium borohydride or lithium aluminum hydride are employed in controlled molar ratios to prevent over-reduction or side reactions on the ester groups. The final closure of the furan ring via reduction-dehydration is conducted under acidic conditions that promote elimination while preserving the sensitive lactone moiety. This mechanistic precision ensures that the final Concentricolide product meets stringent purity specifications, reducing the burden on downstream purification processes such as chromatography. For quality assurance teams, understanding these mechanistic nuances is vital for validating the consistency of the manufacturing process and ensuring that the impurity spectrum remains within acceptable limits for pharmaceutical grade materials.
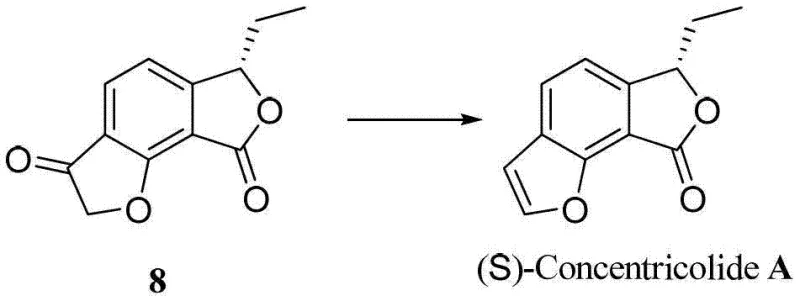
How to Synthesize Concentricolide Efficiently
Implementing this synthesis route requires adherence to specific reaction parameters to maximize yield and safety, particularly during the critical cyclization and reduction phases. The patent outlines a detailed protocol starting from the Michael-Aldol adduct, moving through protection and oxidation, and culminating in the dual ring closure. Operators must maintain strict temperature controls, ranging from 0°C during the initial enolate formation to 80°C for the Dieckmann condensation, to ensure optimal reaction kinetics. The use of anhydrous conditions and inert atmospheres during the reduction steps is paramount to prevent hydrolysis of sensitive intermediates. For a comprehensive understanding of the exact stoichiometry, solvent choices, and workup procedures required to replicate this high-efficiency pathway, please refer to the standardized synthesis guide below.
- Perform Michael-Aldol reaction between 1,3-propanedione carboxylate and alkynal compounds under basic conditions to construct the phenyl core.
- Protect the phenolic hydroxyl group via substitution, followed by benzylic oxidation to generate the ketone intermediate.
- Execute carbonyl reduction to form the benzofuranone lactone, followed by Dieckmann condensation and Krapcho reaction to finalize the furan ring.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, this patented methodology offers transformative benefits that directly address the volatility and cost issues associated with natural product sourcing. By shifting from a fermentation-dependent model to a fully synthetic chemical process, manufacturers can decouple production from biological variables, ensuring a consistent and predictable supply of Concentricolide intermediates regardless of seasonal or environmental factors. The reliance on commodity chemicals such as acetone dicarboxylates and simple alkynals means that raw material procurement is straightforward and less susceptible to market fluctuations compared to specialized biological extracts. This stability is crucial for long-term project planning and inventory management, allowing procurement managers to negotiate better terms with suppliers due to the standardized nature of the input materials. Additionally, the elimination of complex extraction and purification steps associated with fungal fermentation significantly reduces the overall processing time, thereby enhancing the agility of the supply chain in responding to market demands.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the elimination of expensive and hazardous reagents found in previous synthetic routes, alongside the use of cost-effective commodity starting materials. By avoiding the need for toxic or explosive organic substances, the facility saves substantially on safety infrastructure, waste disposal, and regulatory compliance costs, which collectively contribute to a lower cost of goods sold (COGS). The streamlined nature of the reaction sequence, which minimizes the number of isolation and purification steps, further reduces solvent consumption and labor hours, driving down operational expenditures. Moreover, the improved yields reported in the examples compared to historical Diels-Alder methods mean that less raw material is wasted per unit of product, maximizing resource efficiency and profitability.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures high supply chain reliability by mitigating the risks associated with single-source biological ingredients. Since the synthesis relies on well-established organic chemistry principles and widely available reagents, the risk of supply disruption due to crop failure or fermentation contamination is effectively nullified. This reliability allows for the establishment of multi-vendor sourcing strategies for raw materials, fostering a competitive procurement environment that can drive down prices. Furthermore, the scalability of the process from gram to kilogram scales without significant re-optimization means that production capacity can be ramped up quickly to meet sudden increases in demand, ensuring uninterrupted supply for downstream drug development programs.
- Scalability and Environmental Compliance: Environmental sustainability is increasingly a key metric for supply chain heads, and this method aligns well with green chemistry principles by reducing the use of hazardous substances and improving atom economy. The reaction conditions are generally mild, operating at temperatures that do not require excessive energy input for heating or cooling, which lowers the carbon footprint of the manufacturing process. The avoidance of heavy metal catalysts in favor of organic bases and borohydrides simplifies wastewater treatment and reduces the generation of toxic sludge, facilitating easier compliance with stringent environmental regulations. This eco-friendly profile not only reduces liability but also enhances the corporate social responsibility (CSR) standing of the manufacturing entity, making it a more attractive partner for global pharmaceutical companies with strict sustainability mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and experimental data. These insights are intended to clarify the feasibility of scaling this route and the specific advantages it holds over alternative methods. Understanding these details is essential for stakeholders evaluating the integration of Concentricolide into their development pipelines.
Q: What are the key advantages of this synthesis method over extraction?
A: Unlike extraction from fungal sources which is limited by seasonal availability and low yields, this total synthesis method utilizes readily available chemical raw materials, ensuring consistent supply continuity and higher purity specifications suitable for pharmaceutical applications.
Q: Can this route produce chiral Concentricolide?
A: Yes, the patent describes both racemic and asymmetric total synthesis. By employing chiral reducing agents such as (S)-Me-CBS-oxazaborolidine during the carbonyl reduction step, specific R or S configurations can be established at the chiral center.
Q: Is this process scalable for industrial production?
A: The methodology avoids harsh conditions and explosive reagents found in previous literature. It operates under mild temperatures (0°C to 90°C) and uses common solvents like THF and methanol, making it highly amenable to commercial scale-up in standard reactor systems.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Concentricolide Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of Concentricolide as a next-generation anti-HIV agent and are committed to supporting its development through advanced manufacturing capabilities. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to clinical supply is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Concentricolide or its analogues delivered meets the highest international standards for pharmaceutical intermediates. We understand the critical nature of timeline and quality in drug development, and our team is dedicated to providing the technical support necessary to optimize this synthesis route for your specific needs.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis platform to your project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of adopting this route for your specific volume needs. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete technical evidence. Let us collaborate to accelerate the development of life-saving therapies by securing a stable, high-quality supply of this vital natural product analogue.
