Optimizing Reactive Dye Intermediate Production: A Technical Breakthrough in p-Aminophenyl-Beta-Ethoxyl Sulphone Sulphate Synthesis
Optimizing Reactive Dye Intermediate Production: A Technical Breakthrough in p-Aminophenyl-Beta-Ethoxyl Sulphone Sulphate Synthesis
The global demand for high-performance reactive dyes continues to drive innovation in the synthesis of key intermediates, specifically p-aminophenyl-beta-ethoxyl sulphone sulphate, a critical precursor for vinyl sulfone dyes. A pivotal advancement in this domain is detailed in patent CN103626684A, which discloses a novel preparation method that fundamentally restructures the synthetic pathway to enhance efficiency and sustainability. Unlike traditional routes that rely on hazardous chlorosulfonation and complex condensation steps, this technology utilizes a streamlined sequence starting from p-nitrochlorobenzene and mercaptoethanol. For R&D directors and procurement specialists seeking a reliable reactive dye intermediate supplier, understanding the mechanistic shifts in this patent is crucial for evaluating long-term supply stability and cost structures. The process not only addresses the chronic issues of low yield and high waste generation but also introduces a catalytic system that dramatically reduces the dependency on precious metal catalysts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of p-aminophenyl-beta-hydroxyethyl sulfone sulfuric ester has been plagued by severe operational and environmental inefficiencies inherent to the acetanilide-based route. The conventional process initiates with a sulfonation operation using excessive chlorosulfonic acid at cryogenic temperatures around -10°C, generating massive volumes of corrosive hydrogen chloride gas that require complex absorption systems to recover industrial hydrochloric acid. Following sulfonation, the reduction and condensation steps involve sodium pyrosulfite and ethylene oxide, necessitating strict pH control and generating significant amounts of acidic mother liquor and high-salt wastewater. The cumulative effect of these harsh conditions results in a relatively low overall yield of approximately 70%, coupled with a staggering waste profile that includes 8 to 10 tons of waste hydrochloric acid and 10 to 15 tons of waste sulfuric acid per ton of product. Furthermore, previous attempts to optimize this route, such as those cited in patent 200910064706.3, still suffered from excessive consumption of expensive palladium-carbon catalysts and hydrogen peroxide, limiting their economic viability for cost reduction in textile chemical manufacturing.
The Novel Approach
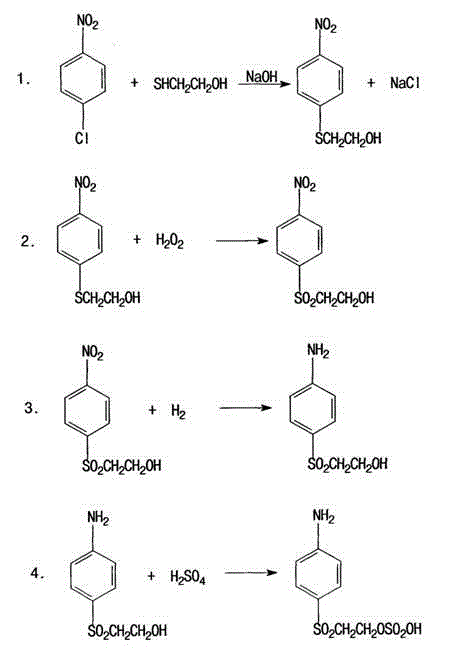
The methodology presented in CN103626684A represents a paradigm shift by adopting a nucleophilic aromatic substitution strategy that bypasses the need for chlorosulfonic acid entirely. This novel approach begins with the reaction of p-nitrochlorobenzene and mercaptoethanol in a dimethylformamide (DMF) solvent system, facilitated by sodium hydroxide, to directly form the thioether linkage. This is followed by a highly selective oxidation step using hydrogen peroxide and a tungstate catalyst to convert the sulfide to a sulfone, and finally, a catalytic hydrogenation to reduce the nitro group to an amine before esterification. By eliminating the generation of gaseous HCl and avoiding the use of ethylene oxide, the process inherently minimizes safety risks and waste treatment burdens. The strategic redesign of the synthetic route allows for a yield increase to approximately 92%, while simultaneously enabling the recycling of the palladium-carbon catalyst, thereby offering a compelling value proposition for commercial scale-up of complex dye intermediates.
Mechanistic Insights into Tungstate-Catalyzed Oxidation and Hydrogenation
The core chemical innovation lies in the precise control of the oxidation and reduction phases, which dictates the purity and impurity profile of the final high-purity vinyl sulfone precursor. In the oxidation step, the use of sodium tungstate as a catalyst activates hydrogen peroxide to selectively oxidize the sulfide bond to a sulfone without over-oxidizing the hydroxyl group or degrading the aromatic ring. The reaction is conducted at moderate temperatures between 80°C and 100°C, where the slow addition of hydrogen peroxide ensures that the exothermic reaction remains under control, preventing thermal runaway and the formation of sulfonic acid byproducts. Following oxidation, the crude material is washed with methanol and deionized water to remove residual catalyst and organic impurities, a purification step that is critical for the subsequent hydrogenation efficiency. The hydrogenation phase utilizes a palladium-carbon catalyst under hydrogen pressure of 1.5 to 2.5 MPa at temperatures ranging from 40°C to 110°C. Crucially, the patent specifies that the hydrogenation product does not require isolation from the aqueous solvent before esterification, which simplifies the workflow and reduces solvent loss.
Impurity control is further enhanced by the specific stoichiometry employed in the initial nucleophilic substitution. By maintaining a molar ratio of p-nitrochlorobenzene to mercaptoethanol between 1:1 and 1:1.2, the process minimizes the formation of disulfide byproducts that often complicate downstream purification. The filtration of sodium chloride byproduct at temperatures below 40°C ensures that the subsequent distillation of DMF is efficient, recovering the solvent for reuse and leaving a high-purity thioether intermediate. This rigorous attention to stoichiometric balance and temperature gradients throughout the four-step sequence ensures that the final esterification with sulfuric acid proceeds with minimal side reactions, resulting in a product with a consistent impurity spectrum that meets the stringent requirements of modern reactive dye formulations.
How to Synthesize p-Aminophenyl-Beta-Ethoxyl Sulphone Sulphate Efficiently
Implementing this synthesis route requires careful adherence to the specific reaction parameters outlined in the patent to maximize yield and catalyst longevity. The process is designed to be scalable, moving seamlessly from laboratory validation to industrial production with minimal modification to the core unit operations. Operators must focus on the precise timing of reagent addition, particularly during the exothermic oxidation and hydrogenation phases, to maintain safety and product quality. The following guide summarizes the critical operational stages derived from the patent examples, providing a roadmap for technical teams aiming to adopt this greener manufacturing protocol. For detailed standard operating procedures and safety data sheets, please refer to the standardized synthesis steps provided below.
- Perform nucleophilic substitution of p-nitrochlorobenzene with mercaptoethanol in DMF solvent using NaOH to form the thioether intermediate.
- Oxidize the thioether intermediate to the corresponding sulfone using hydrogen peroxide and a tungstate catalyst under controlled temperature.
- Execute catalytic hydrogenation using palladium-carbon to reduce the nitro group to an amine, followed by direct esterification with sulfuric acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this patented synthesis method offers tangible strategic advantages beyond mere technical superiority. The most significant impact is observed in the drastic reduction of raw material costs, primarily driven by the optimization of catalyst usage. In traditional processes, the palladium-carbon catalyst is often consumed in single-use batches, representing a substantial variable cost due to the high market price of palladium. This new technology reduces the catalyst dosage by approximately tenfold and enables its recovery and reuse for multiple cycles, effectively amortizing the cost of this precious metal over a much larger production volume. Additionally, the elimination of chlorosulfonic acid and the associated waste neutralization chemicals removes a major cost center related to hazardous material handling and disposal, leading to substantial cost savings in overall manufacturing overheads.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the significant decrease in catalyst consumption and the ability to recycle the palladium-carbon catalyst two to three times. Since palladium-carbon is a high-value input, reducing its usage intensity directly improves the gross margin of the final product. Furthermore, the higher reaction yield of up to 92% means that less raw material is required to produce the same amount of finished goods, enhancing material efficiency and reducing the cost per kilogram of the active intermediate.
- Enhanced Supply Chain Reliability: By simplifying the synthetic route and removing dependence on volatile reagents like ethylene oxide and chlorosulfonic acid, the supply chain becomes more robust and less susceptible to regulatory disruptions. The use of stable, commercially available starting materials such as p-nitrochlorobenzene and mercaptoethanol ensures consistent availability. Moreover, the reduction in waste generation simplifies logistics related to waste transport and treatment, minimizing the risk of production stoppages due to environmental compliance issues or waste storage capacity limits.
- Scalability and Environmental Compliance: The process is inherently designed for environmental sustainability, with wastewater output reduced to below 0.8 tons per ton of product, a figure significantly lower than the multi-ton waste profiles of legacy methods. This low waste footprint facilitates easier permitting for capacity expansion and aligns with the increasingly strict environmental regulations governing the chemical industry globally. The ability to operate with lower energy inputs for cooling and heating, combined with solvent recovery systems for DMF, further supports sustainable large-scale manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on performance metrics and operational feasibility. Understanding these details is essential for stakeholders evaluating the integration of this intermediate into their broader dye manufacturing supply chains.
Q: How does the new synthesis route improve catalyst efficiency compared to traditional methods?
A: The patented process significantly reduces the consumption of expensive palladium-carbon catalyst by approximately tenfold compared to prior art. Furthermore, the catalyst can be recovered and recycled 2 to 3 times without loss of activity, drastically lowering raw material costs.
Q: What are the environmental benefits of this preparation method?
A: This method eliminates the use of chlorosulfonic acid and ethylene oxide, thereby avoiding the generation of large quantities of waste hydrochloric acid and high-salt wastewater. The total wastewater output is reduced to below 0.8 tons per ton of product, ensuring better compliance with environmental regulations.
Q: What is the expected yield improvement with this technology?
A: While conventional methods typically achieve yields around 70% to 85%, this optimized process consistently achieves yields up to 92%. This improvement is attributed to the milder reaction conditions and the elimination of side reactions associated with harsh sulfonation agents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-Aminophenyl-Beta-Ethoxyl Sulphone Sulphate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced synthetic routes like CN103626684A requires a partner with deep technical expertise and robust manufacturing capabilities. As a specialized CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical application. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of p-aminophenyl-beta-ethoxyl sulphone sulphate meets the exacting standards required for high-quality reactive dye production. We are committed to delivering consistency and reliability, bridging the gap between innovative patent chemistry and commercial reality.
We invite global partners to collaborate with us to leverage these technological advancements for their specific market needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your current procurement volumes, demonstrating exactly how switching to this optimized intermediate can improve your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the dynamic textile chemical market.
