Advanced Synthesis of Quinoxalinedione Derivatives for CNS Drug Development and Commercial Scale-up
Advanced Synthesis of Quinoxalinedione Derivatives for CNS Drug Development and Commercial Scale-up
The pharmaceutical landscape for central nervous system (CNS) disorders is constantly evolving, driven by the need for highly selective receptor modulators that minimize off-target effects. A pivotal advancement in this domain is documented in Chinese Patent CN1053190C, which discloses a comprehensive series of quinoxalinedione derivatives exhibiting potent antagonistic activity at the AMPA receptor. These compounds, characterized by a distinct 1,2,3,4-tetrahydro-2,3-dioxoquinoxaline core, represent a critical class of pharmaceutical intermediates for developing treatments against hyperexcitability disorders such as epilepsy, stroke, and neurodegenerative conditions like Alzheimer's disease. The structural versatility of these molecules allows for extensive modification at the N-1 position and the benzene ring, enabling fine-tuning of pharmacokinetic profiles. As a leading entity in fine chemical manufacturing, understanding the synthetic nuances of these scaffolds is essential for ensuring a reliable supply chain for global drug developers.
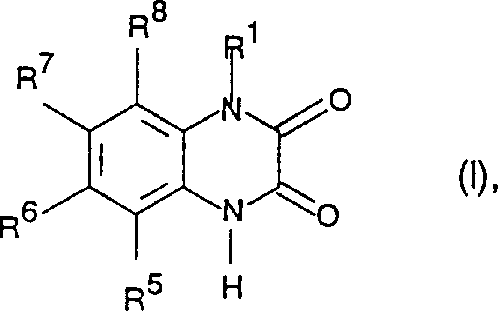
The core innovation lies in the robust construction of the quinoxaline dione ring system, which serves as a rigid bioisostere for glutamic acid. The patent details specific substituent patterns, particularly at the 6 and 7 positions, where electron-withdrawing groups like trifluoromethyl or halogens enhance receptor binding affinity. Furthermore, the inclusion of phosphonic acid or carboxylic acid moieties at the N-1 side chain significantly improves water solubility and bioavailability. For procurement and R&D teams, mastering the synthesis of these complex heterocycles is not merely a chemical challenge but a strategic imperative to secure cost reduction in pharmaceutical intermediate manufacturing. By leveraging the methodologies outlined in this intellectual property, manufacturers can bypass inefficient multi-step sequences and achieve higher overall yields through streamlined cyclization protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinoxaline-2,3-diones has been plagued by issues related to harsh reaction conditions and poor regioselectivity. Traditional approaches often rely on the direct condensation of o-phenylenediamines with oxalic acid under high thermal stress, which can lead to significant decomposition of sensitive functional groups, particularly when dealing with complex side chains containing esters or phosphonates. Moreover, conventional nitration or halogenation steps performed on the pre-formed quinoxaline core frequently result in mixtures of isomers that are notoriously difficult to separate, thereby inflating production costs and extending lead times. The use of strong mineral acids and elevated temperatures in older protocols also poses significant safety hazards and environmental compliance challenges, necessitating expensive waste treatment infrastructure. For large-scale operations, these inefficiencies translate into inconsistent batch quality and unreliable supply continuity, which are unacceptable for GMP-grade active pharmaceutical ingredient (API) production.
The Novel Approach
In contrast, the methodology presented in CN1053190C introduces a refined, two-step cyclization strategy that markedly improves process control and product purity. Instead of forcing the reaction with free oxalic acid, the novel approach employs activated oxalic acid derivatives, such as oxalyl chloride hemi-esters or reactive oxalylimidazoles, in the presence of organic bases like triethylamine. This allows the acylation to proceed smoothly at moderate temperatures, preserving the integrity of labile substituents on the aromatic ring. The subsequent ring closure is achieved under mild acidic conditions in alcoholic solvents, which facilitates the elimination of the leaving group without degrading the phosphonic acid ester side chains. This tactical shift from brute-force thermal cyclization to a controlled, stepwise acylation-cyclization sequence ensures that the final high-purity quinoxalinedione products are obtained with minimal impurity profiles. Such precision is vital for meeting the stringent regulatory standards required for CNS therapeutics.
Mechanistic Insights into Oxalyl-Mediated Cyclization
The mechanistic pathway for constructing the quinoxaline dione scaffold involves a nucleophilic attack by the amine nitrogen of the o-phenylenediamine precursor onto the electrophilic carbonyl carbon of the oxalyl derivative. In the first stage, the formation of an amide bond is facilitated by the base, which scavenges the generated hydrogen chloride, driving the equilibrium towards the acyclic bis-amide or mono-amide intermediate. This intermediate is crucial as it pre-organizes the molecule for the subsequent intramolecular cyclization. The presence of electron-withdrawing groups on the benzene ring, such as the trifluoromethyl or nitro groups described in the patent examples, enhances the nucleophilicity of the adjacent amino group through resonance and inductive effects, thereby accelerating the initial acylation step. Understanding this electronic interplay is key for R&D directors aiming to optimize reaction kinetics and minimize the formation of mono-acylated byproducts.
Following acylation, the cyclization step proceeds via an intramolecular nucleophilic attack of the second amine nitrogen onto the remaining carbonyl group of the oxalyl moiety. This step is typically catalyzed by acid in an alcoholic medium, which activates the carbonyl oxygen and promotes the departure of the alkoxy or chloro leaving group. The resulting tetrahedral intermediate collapses to form the stable six-membered dione ring, releasing the leaving group as a salt or alcohol. Crucially, the patent highlights that this cyclization can be performed in a one-pot manner or as a distinct second step, offering flexibility in process design. For commercial scale-up, controlling the pH and temperature during this phase is paramount to prevent hydrolysis of the sensitive phosphonate esters attached at the N-1 position. This mechanistic clarity allows process chemists to design robust control strategies that ensure consistent commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Quinoxalinedione Derivatives Efficiently
The practical execution of this synthesis route requires careful selection of solvents and reagents to maximize yield and safety. The process begins with the preparation of the requisite substituted o-phenylenediamine, often achieved via the reduction of a corresponding nitro-aniline precursor using catalytic hydrogenation or metal-acid reduction. Once the diamine is secured, it is reacted with an oxalylating agent in a dry, aprotic solvent such as tetrahydrofuran (THF) or dichloromethane. The detailed standardized synthesis steps below outline the precise operational parameters derived from the patent examples, ensuring reproducibility from gram to kilogram scales.
- Prepare the substituted o-phenylenediamine precursor (Formula II) via nucleophilic aromatic substitution or reduction of nitro-anilines.
- React the diamine with an activated oxalic acid derivative, such as oxalyl chloride hemi-ester, in a polar solvent like THF with a base.
- Effect ring closure by heating the intermediate in an alcoholic solvent with acid catalysis to yield the final quinoxalinedione core.
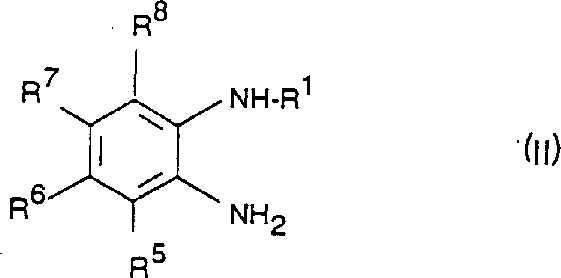
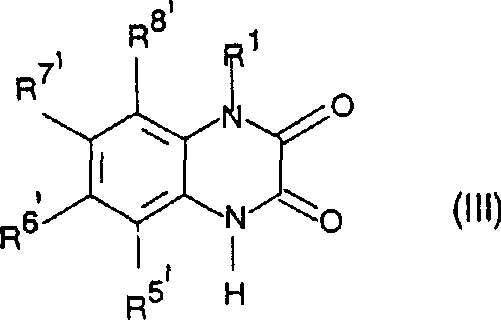
Commercial Advantages for Procurement and Supply Chain Teams
Adopting the synthetic strategies outlined in this patent offers substantial logistical and economic benefits for procurement managers and supply chain heads. By utilizing readily available starting materials such as fluoronitrobenzenes and simple alkyl amines, manufacturers can mitigate the risks associated with sourcing exotic or single-source reagents. The reliance on common industrial solvents like ethanol, THF, and ethyl acetate further simplifies the supply chain, as these commodities are globally accessible and cost-stable. Furthermore, the elimination of transition metal catalysts in the key cyclization step removes the need for expensive and time-consuming heavy metal scavenging processes, which are often a bottleneck in API manufacturing. This streamlining directly contributes to cost reduction in pharmaceutical intermediate manufacturing by lowering both material and processing expenses.
- Cost Reduction in Manufacturing: The process avoids the use of precious metal catalysts for the core ring formation, relying instead on inexpensive organic bases and oxalyl derivatives. This fundamental change in reagent profile drastically reduces the raw material bill of goods. Additionally, the high selectivity of the reaction minimizes the formation of difficult-to-remove isomers, reducing the burden on purification units and lowering solvent consumption during chromatography or recrystallization. The overall atom economy is improved by the efficient incorporation of the oxalyl unit, ensuring that waste generation is kept to a minimum while maximizing the output of valuable product per batch.
- Enhanced Supply Chain Reliability: The synthetic route is designed around robust chemical transformations that are less sensitive to minor fluctuations in temperature or moisture compared to older methods. This inherent stability translates to fewer failed batches and more predictable production schedules, which is critical for maintaining continuous supply to downstream drug formulators. The starting materials, primarily substituted anilines and phosphonates, are produced by multiple global suppliers, preventing supply bottlenecks. By diversifying the source of key inputs and simplifying the synthesis, companies can significantly reduce lead times and ensure a steady flow of materials for clinical and commercial needs.
- Scalability and Environmental Compliance: The reactions described operate under mild conditions that are easily transferable from laboratory glassware to large-scale stainless steel reactors. The absence of highly corrosive reagents or extreme pressures enhances operational safety and reduces the maintenance costs of manufacturing equipment. From an environmental perspective, the process generates fewer hazardous byproducts, and the solvents used are amenable to standard recovery and recycling protocols. This alignment with green chemistry principles facilitates easier regulatory approval and lowers the costs associated with waste disposal and environmental monitoring, making the entire operation more sustainable and economically viable in the long term.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these quinoxalinedione derivatives. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing production pipelines.
Q: What are the primary therapeutic applications of these quinoxalinedione derivatives?
A: These compounds exhibit high affinity for AMPA receptors, making them potent candidates for treating neurological disorders such as epilepsy, stroke, and neurodegenerative diseases like Alzheimer's.
Q: How does the patented cyclization method improve upon traditional synthesis routes?
A: The method utilizes mild conditions with oxalyl derivatives and avoids harsh oxidizing agents, resulting in higher purity profiles and simplified downstream processing for commercial manufacturing.
Q: Can the R1 phosphonic acid side chain be modified post-cyclization?
A: Yes, the patent describes versatile functionalization strategies, including ester hydrolysis and nucleophilic substitution, allowing for diverse structural modifications to optimize pharmacokinetic properties.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoxalinedione Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of CNS therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from clinical trials to market launch. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of quinoxalinedione derivative meets the highest international standards. Our commitment to technical excellence means we can navigate the complexities of phosphonate chemistry and heterocyclic cyclization with precision, delivering materials that accelerate your drug discovery timelines.
We invite you to collaborate with us to explore how our advanced manufacturing capabilities can support your specific requirements. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your project volume. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain efficiency and reduce overall production costs. Let us be your partner in bringing innovative neurological treatments to patients worldwide.
