Advanced Oxidative Coupling for High-Purity Aromatic Dimers and Commercial Scalability
Advanced Oxidative Coupling for High-Purity Aromatic Dimers and Commercial Scalability
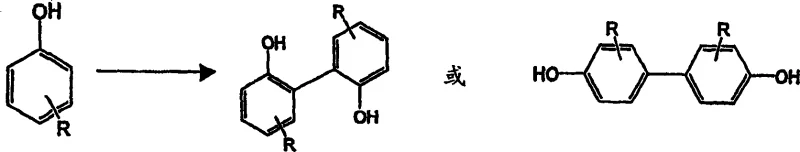
The chemical industry continuously seeks robust methodologies for constructing biaryl scaffolds, which serve as critical backbones for asymmetric catalysts, agrochemicals, and functional materials. Patent CN1463264A introduces a transformative approach to the synthesis of monohydroxyaromatic compound dimers, specifically addressing the limitations of traditional oxidative coupling protocols. This technology leverages a copper-catalyzed system within nitrogen-containing polar solvents to achieve high-yield dimerization of substrates ranging from simple phenols to complex naphthol derivatives. By eliminating the need for toxic non-polar solvents and cumbersome catalyst pre-activation steps, this method offers a streamlined pathway for producing high-value intermediates like 1,1'-bi-2-naphthol (BINOL) derivatives. For R&D directors and procurement specialists, understanding this mechanistic shift is crucial for optimizing supply chains and reducing the cost of goods sold for downstream applications in chiral synthesis and toner manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the dimerization of beta-naphthol and its derivatives has relied on protocols that introduce significant operational hazards and inefficiencies into the manufacturing process. Prior art, such as that disclosed in US Patent 3,278,610, typically necessitates the use of benzene or dichloromethane as reaction media. These solvents pose severe environmental and health risks, complicating waste disposal and increasing regulatory compliance costs for large-scale facilities. Furthermore, these non-polar solvents often fail to adequately dissolve functionalized substrates, particularly those bearing polar groups like carboxylic acids or amides, leading to heterogeneous reaction conditions that suppress yields and prolong reaction times. Another critical bottleneck in conventional wisdom involves the catalyst system; methods described in literature such as J. Org. Chem. 1999 require the预先 preparation of specific copper-amine complexes, such as copper(I) chloride-tetramethylethylenediamine adducts. This pre-complexation step adds a layer of procedural complexity, requiring additional reagents, time, and quality control measures before the actual coupling reaction can even commence, thereby inflating the overall production cost.
The Novel Approach
The methodology outlined in CN1463264A fundamentally reengineers the reaction environment to overcome these solubility and catalytic hurdles. By transitioning to nitrogen-containing polar solvents such as N,N-dimethylformamide (DMF), N,N-dimethylacetamide (DMAc), or N-methyl-2-pyrrolidone (NMP), the process ensures the complete dissolution of even recalcitrant substrates like 2-hydroxynaphthalene-3-carboxylic acid derivatives. This homogeneity is pivotal for maximizing molecular collisions and ensuring consistent reaction kinetics across the entire batch. Moreover, the novel approach simplifies the catalytic cycle by utilizing simple copper salts, preferably copper(I) chloride, directly without the need for separate ligand complexation. The solvent itself acts as a coordinating ligand, stabilizing the copper species and facilitating the formation of an active oxidative complex in the presence of oxygen. This elimination of the pre-complexation step not only reduces the number of unit operations but also minimizes the introduction of extraneous impurities, resulting in a cleaner crude product profile that requires less intensive purification downstream.
Mechanistic Insights into Copper-Catalyzed Oxidative Coupling
The efficacy of this dimerization process is rooted in the unique interplay between the copper salt, the nitrogenous solvent, and molecular oxygen. Mechanistically, the copper salt does not act in isolation; rather, it reacts with the hydroxyl group of the aromatic substrate and the lone pair electrons of the nitrogen-containing solvent to generate a transient, high-energy coordination complex. This complex possesses significant oxidative potential, enabling the abstraction of electrons from the aromatic ring to generate radical intermediates. These radicals subsequently undergo coupling at specific positions dictated by the electronic nature of the substrate—for phenols, this typically occurs at the ortho or para positions, while for beta-naphthols, coupling favors the 1-position to form 1,1'-binaphthyl structures. The presence of oxygen is critical not merely as an oxidant to drive the initial radical formation but also to regenerate the active copper species from its reduced state, creating a sustainable catalytic cycle that allows for the use of substoichiometric amounts of copper salt relative to the substrate.

Controlling the selectivity of the coupling is paramount for producing high-purity intermediates suitable for asymmetric catalysis. The reaction conditions described allow for precise modulation of the coupling mode. While the primary pathway leads to C-C single bond formation yielding biaryl diols, the system is also capable of facilitating further oxidation to form quinone methide-type dimers linked by double bonds, depending on the substrate structure and reaction duration. This versatility is managed by the stability of the intermediate radicals and the steric environment provided by substituents on the aromatic rings. For instance, bulky groups like tert-butyl can direct the coupling to specific open positions, preventing polymerization or over-oxidation. The use of polar solvents also helps stabilize charged transition states that might arise during the electron transfer processes, thereby suppressing side reactions such as ether formation or random polymerization. This level of mechanistic control ensures that the final product distribution is heavily skewed towards the desired dimer, simplifying the impurity profile and reducing the burden on downstream crystallization or chromatography steps.
How to Synthesize Aromatic Dimers Efficiently
Implementing this oxidative coupling strategy requires careful attention to solvent selection and oxygen mass transfer to ensure reproducibility at scale. The process begins by dissolving the monohydroxyaromatic starting material in a polar aprotic solvent like DMF at ambient temperature, ensuring a homogeneous solution before catalyst addition. Copper(I) chloride is then introduced, typically in an amount ranging from 0.5 to 10 mole percent relative to the substrate, although higher loadings can be used for difficult substrates. The reaction mixture is subsequently heated to a moderate temperature range of 60-80°C, which provides sufficient thermal energy to overcome the activation barrier for radical generation without promoting thermal decomposition of the sensitive biaryl products. Crucially, air or pure oxygen must be actively sparged or bubbled through the reaction mixture to maintain a constant supply of the terminal oxidant, which drives the catalytic turnover. Monitoring the reaction progress via HPLC allows for precise determination of the endpoint, typically achieved within 1 to 48 hours depending on the specific substrate reactivity.
- Dissolve or suspend the monohydroxyaromatic substrate in a nitrogen-containing polar solvent such as N,N-dimethylformamide at room temperature.
- Add a catalytic amount of copper salt, preferably copper(I) chloride, to the reaction mixture under stirring.
- Heat the mixture to 60-80°C while actively supplying air or oxygen to facilitate the oxidative coupling reaction until completion.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this copper-catalyzed oxidative coupling method offers substantial strategic advantages for procurement managers and supply chain leaders looking to optimize their sourcing of aromatic intermediates. The most immediate impact is seen in the reduction of raw material costs and processing complexity. By eliminating the requirement for specialized amine ligands like tetramethylethylenediamine and avoiding the separate synthesis of copper-ligand complexes, the bill of materials is significantly simplified. This streamlining translates directly into lower variable costs per kilogram of product, as fewer reagents need to be sourced, qualified, and inventoried. Furthermore, the substitution of hazardous solvents like benzene with industrially common polar solvents such as DMF reduces the regulatory burden associated with solvent handling, storage, and disposal, thereby lowering the total cost of ownership for the manufacturing facility.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the drastic simplification of the catalytic system and the high efficiency of the reaction medium. Traditional methods often suffer from low atom economy due to the need for stoichiometric oxidants or expensive ligand systems, whereas this method utilizes atmospheric oxygen as a free and abundant oxidant. The ability to use simple copper salts, which are commodity chemicals with stable pricing and widespread availability, shields the supply chain from volatility associated with specialty reagents. Additionally, the high solubility of substrates in the chosen solvents allows for higher concentration reactions, which increases the volumetric productivity of the reactors. This means that existing infrastructure can produce more output in less time, effectively spreading fixed capital costs over a larger volume of goods and driving down the unit cost of production significantly.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on universally available reagents and robust reaction conditions. Copper chloride and DMF are produced on a massive global scale for diverse industries, ensuring that supply disruptions are highly unlikely compared to niche ligands or custom-synthesized catalysts. The process tolerance for moisture and oxygen, inherent in an oxidative coupling that requires air, means that stringent anhydrous or inert atmosphere conditions are not necessary. This relaxation of environmental controls simplifies the engineering requirements for the production plant, allowing for operation in standard glass-lined or stainless steel reactors without the need for expensive nitrogen blanketing or glovebox techniques. Consequently, lead times for production scheduling can be shortened, and the risk of batch failure due to minor deviations in atmospheric conditions is virtually eliminated.
- Scalability and Environmental Compliance: Scaling this technology from laboratory to commercial production is facilitated by the straightforward workup procedure, which typically involves simple filtration and washing. The precipitation of the product upon addition of water or methanol allows for easy isolation of the solid dimer, avoiding energy-intensive distillation steps for solvent recovery in the initial isolation phase. From an environmental standpoint, the avoidance of chlorinated solvents like dichloromethane and carcinogenic solvents like benzene aligns perfectly with modern green chemistry principles and increasingly strict environmental regulations. This compliance reduces the risk of regulatory fines and facilitates easier permitting for capacity expansion. The high yield and purity achieved also mean less waste generation per unit of product, further enhancing the sustainability profile of the manufacturing process and appealing to environmentally conscious downstream customers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidative coupling technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational benefits for potential partners. Understanding these nuances is essential for evaluating the feasibility of integrating this method into existing production lines for fine chemical intermediates.
Q: What are the advantages of using nitrogen-containing polar solvents over benzene for dimerization?
A: Nitrogen-containing polar solvents like DMF or NMP significantly enhance the solubility of poorly soluble substrates such as hydroxynaphthoic acid derivatives, eliminating the need for toxic benzene and improving reaction homogeneity and yield.
Q: Is pre-complexation of the copper catalyst required in this method?
A: No, unlike prior art methods requiring the tedious preparation of copper-amine complexes, this process utilizes simple copper salts directly, which form the active catalytic species in situ with the solvent and oxygen.
Q: Can this method be scaled for industrial production of asymmetric catalyst precursors?
A: Yes, the method operates at moderate temperatures (60-80°C) using inexpensive reagents and simple workup procedures like filtration, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aromatic Dimer Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality aromatic dimers play in the synthesis of advanced pharmaceuticals and functional materials. Our technical team has extensively evaluated the copper-catalyzed oxidative coupling route described in CN1463264A and validated its potential for robust commercial manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale optimization to full-scale manufacturing is seamless and efficient. Our facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of binaphthol derivatives or phenolic dimers meets the exacting standards required for asymmetric catalysis and electronic applications. We are committed to delivering consistency and reliability in every shipment.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific supply chain needs. Our experts are ready to conduct a Customized Cost-Saving Analysis tailored to your target molecules, identifying opportunities to reduce expenses without compromising quality. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your projects. By partnering with us, you gain access to a secure, scalable, and cost-effective source of complex aromatic intermediates, empowering your R&D and production teams to focus on innovation while we handle the complexities of chemical manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
