Advanced Synthesis of Diether Tetraacid Dianhydride Isomers for High-Performance Polyimide Production
Advanced Synthesis of Diether Tetraacid Dianhydride Isomers for High-Performance Polyimide Production
The chemical landscape for high-performance polymer precursors is undergoing a significant transformation driven by the innovations detailed in patent CN1850814A. This pivotal intellectual property introduces a robust and environmentally conscious methodology for preparing diether type tetraacid dianhydride isomers, which serve as critical monomers for advanced polyimide resins. Unlike traditional routes that rely on hazardous nitro-substituted starting materials, this novel approach utilizes N-substituted chlorophthalimide isomers derived from readily available and cost-effective chlorinated phthalic anhydrides. The technical breakthrough lies in the ability to synthesize these complex dianhydrides, including the previously elusive 3,4'-position isomer, through a streamlined one-pot reaction sequence. For R&D directors and procurement specialists in the electronics and aerospace sectors, this represents a paradigm shift towards more sustainable and economically viable supply chains for high-purity electronic chemicals and polymer additives without compromising on thermal or mechanical performance standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of diether type tetraacid dianhydrides has been plagued by significant technical and economic inefficiencies inherent to legacy processes. Conventional methods, as documented in various United States patents, predominantly rely on nitro-substituted phthalimides or nitro-substituted phthalonitriles as the primary electrophilic building blocks. These nitro-containing precursors are not only substantially more expensive due to complex synthesis requirements but also introduce severe environmental liabilities regarding toxic waste disposal and worker safety. Furthermore, reactions involving nitro groups often result in side reactions that generate deeply colored impurities, necessitating rigorous and costly purification steps to achieve the pale color required for optical grade polyimides. The multi-step nature of these traditional pathways, often involving separate salt formation and coupling stages, further exacerbates production costs and reduces overall atom economy, making the final dianhydride monomers less competitive in price-sensitive markets like consumer electronics and automotive coatings.
The Novel Approach
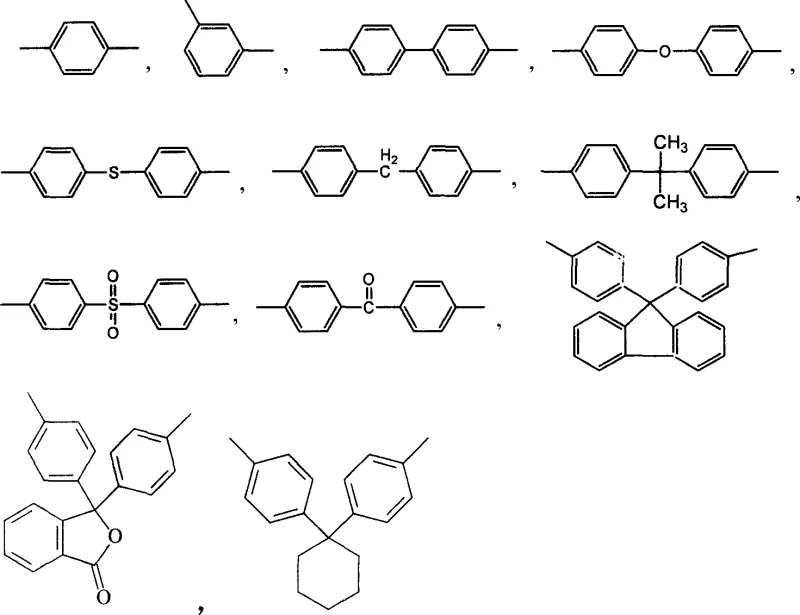
In stark contrast, the methodology disclosed in CN1850814A leverages the superior reactivity and availability of N-substituted chlorophthalimide isomers to overcome these longstanding barriers. By employing a direct nucleophilic substitution strategy, the process enables a one-pot reaction between the chlorophthalimide monomer, a diverse range of bisphenols, and anhydrous potassium carbonate within an aprotic polar solvent medium. This consolidation of reaction steps drastically simplifies the operational workflow, eliminating the need for intermediate isolation and reducing solvent consumption. The versatility of this approach is highlighted by the compatibility with various bisphenol structures, allowing for the fine-tuning of the final polymer's properties. As illustrated in the structural diversity of the bisphenol components below, the process accommodates different bridging groups which dictate the flexibility and thermal stability of the resulting polyimide backbone.
The implementation of this novel route results in a marked improvement in product quality, specifically yielding dianhydrides with significantly lighter color and higher purity profiles compared to nitro-based counterparts. The use of chlorine as a leaving group, activated by the adjacent imide carbonyls, facilitates efficient coupling under moderate thermal conditions ranging from 90°C to 180°C. Following the initial coupling, the subsequent hydrolysis, acidification, and dehydration steps are straightforward and scalable, utilizing common reagents like sodium hydroxide and acetic anhydride. This operational simplicity translates directly into reduced capital expenditure for manufacturing facilities and lower operating costs, positioning this technology as a preferred choice for the commercial scale-up of complex electronic chemical intermediates.
Mechanistic Insights into Nucleophilic Aromatic Substitution and Isomer Control
The core chemical transformation driving this synthesis is a nucleophilic aromatic substitution (SNAr) facilitated by the strong electron-withdrawing nature of the imide ring. In the presence of anhydrous potassium carbonate, the phenolic hydroxyl groups of the bisphenol are deprotonated to form reactive phenoxide anions in situ. These anions then attack the carbon atoms bearing the chlorine substituents on the N-substituted chlorophthalimide rings. The reaction kinetics are heavily influenced by the choice of aprotic polar solvent, such as DMSO or NMP, which effectively solvates the cationic potassium species while leaving the nucleophilic phenoxide anions relatively naked and highly reactive. This mechanistic pathway ensures high conversion rates even with sterically hindered bisphenols, such as bisphenol fluorene, demonstrating the robustness of the catalytic system. The precise control of reaction temperature and stoichiometry, typically maintaining a molar ratio of chlorophthalimide to bisphenol between 1.8:1 and 2.2:1, is critical to minimizing the formation of mono-substituted by-products and ensuring the formation of the desired diether tetraimide intermediate.
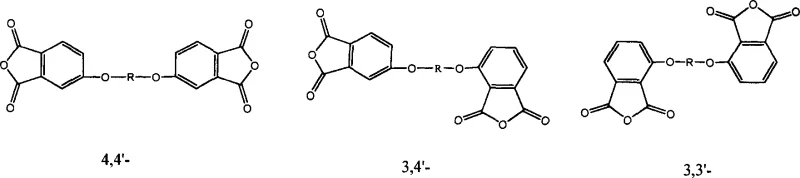
Following the formation of the tetraimide intermediate, the process proceeds through a hydrolysis step where the imide rings are opened using aqueous sodium hydroxide to form the water-soluble tetracarboxylic acid tetra-sodium salt. This step is crucial for purification, as many organic impurities remain insoluble or can be washed away before the final acidification. Upon acidification with hydrochloric acid, the tetracarboxylic acid precipitates as a white solid, which is then dehydrated using acetic anhydride to cyclize back into the thermodynamically stable dianhydride form. A unique aspect of this patent is the ability to control the isomeric composition of the final product. By mixing 3-chlorophthalimide and 4-chlorophthalimide isomers in specific ratios, manufacturers can access the 3,4'-isomer, which offers distinct packing and solubility characteristics compared to the symmetric 4,4' or 3,3' isomers. The structural relationships between these critical isomers are depicted below, highlighting the positional variance that defines their physical properties.
How to Synthesize Diether Tetraacid Dianhydride Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing the importance of anhydrous conditions during the initial coupling phase to prevent premature hydrolysis of the chlorophthalimide. Operators must carefully select the solvent system based on the solubility of the specific bisphenol being used, with DMSO and NMP showing excellent results across a wide range of substrates. The reaction mixture is typically heated under a nitrogen atmosphere to prevent oxidative degradation of the phenolic components, ensuring the final product maintains its high purity specifications. After the coupling is complete, the workup involves pouring the reaction mass into water or ethanol to precipitate the crude tetraimide, which is then washed thoroughly to remove inorganic salts. The subsequent hydrolysis and dehydration steps require precise pH control and temperature management to maximize yield and minimize thermal degradation of the sensitive anhydride functionality.
- React N-substituted chlorophthalimide isomers with bisphenol and anhydrous potassium carbonate in an aprotic polar solvent at 90-180°C to form the tetraimide intermediate.
- Hydrolyze the resulting N,N'-disubstituted bis-ether tetraimide using aqueous sodium hydroxide solution under reflux conditions to obtain the tetracarboxylic acid tetra-sodium salt.
- Acidify the salt solution to precipitate the tetracarboxylic acid, followed by dehydration using acetic anhydride to yield the final diether tetraacid dianhydride isomer.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this chlorophthalimide-based synthesis route offers compelling advantages in terms of cost structure and supply chain resilience. The primary driver for cost reduction is the substitution of expensive, specialty nitro-compounds with commodity-grade chlorinated phthalic anhydrides, which are produced in massive volumes for other industries and thus benefit from economies of scale. This raw material swap not only lowers the direct material cost but also mitigates the volatility associated with the supply of niche nitro-intermediates. Furthermore, the elimination of nitro-groups from the process stream removes the regulatory burden and disposal costs associated with nitro-containing hazardous waste, leading to substantial indirect savings in environmental compliance and waste treatment operations. The simplified one-pot nature of the reaction also reduces utility consumption and labor hours per batch, enhancing the overall manufacturing efficiency.
- Cost Reduction in Manufacturing: The transition to cheaper chlorophthalimide starting materials fundamentally alters the cost basis of dianhydride production. By avoiding the multi-step synthesis required for nitro-precursors and utilizing a one-pot coupling method, the process significantly reduces energy consumption and solvent usage. The high yields reported in the patent examples, often exceeding ninety percent, mean that less raw material is wasted, directly improving the gross margin for manufacturers. Additionally, the lighter color of the crude product reduces the need for expensive activated carbon treatments or recrystallization steps, further streamlining the purification budget and lowering the total cost of ownership for the final electronic chemical.
- Enhanced Supply Chain Reliability: Relying on widely available chlorinated feedstocks insulates the supply chain from the bottlenecks often seen with specialized nitro-chemicals. Since the key raw materials are commodity chemicals with multiple global suppliers, the risk of single-source dependency is drastically minimized. This diversification ensures consistent availability of precursors even during market fluctuations, guaranteeing uninterrupted production schedules for downstream polyimide manufacturers. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, providing a buffer against supply chain variability and ensuring consistent output quality for critical aerospace and electronics applications.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing standard reactor configurations and common solvents that are easily handled in large-scale chemical plants. The absence of nitro-groups simplifies the safety profile of the facility, reducing the need for specialized explosion-proof equipment and lowering insurance premiums. From an environmental standpoint, the aqueous workup and the ability to recycle solvents like DMSO or NMP align with modern green chemistry principles. The reduction in hazardous waste generation facilitates easier permitting and compliance with increasingly stringent environmental regulations, making this technology a future-proof investment for sustainable chemical manufacturing operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the feasibility of this route for your specific application needs. Understanding these nuances is essential for R&D teams planning pilot runs and procurement officers negotiating supply contracts for high-purity intermediates.
Q: What are the primary advantages of using chlorophthalimides over nitro-phthalimides in this synthesis?
A: Using N-substituted chlorophthalimides significantly reduces raw material costs and eliminates the environmental pollution associated with nitro-substituted precursors. Furthermore, this substitution avoids the formation of deeply colored by-products, resulting in a final dianhydride product with superior purity and lighter color, which is critical for optical applications in electronics.
Q: Can this process selectively produce the 3,4'-isomer of diether tetraacid dianhydride?
A: Yes, the patent explicitly details the first-time synthesis of the 3,4'-position isomer. By adjusting the ratio of 3-chlorophthalimide to 4-chlorophthalimide monomers in the reaction mixture, manufacturers can influence the isomeric distribution of the final product, allowing for tailored properties in the resulting polyimide resin.
Q: What solvents are compatible with this one-pot synthesis method?
A: The process utilizes common aprotic polar solvents such as dimethyl sulfoxide (DMSO), dimethylformamide (DMF), N-methyl pyrrolidone (NMP), or tetramethylene sulfone. These solvents facilitate the nucleophilic substitution reaction at temperatures ranging from 90°C to 180°C, ensuring high conversion rates and operational simplicity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diether Tetraacid Dianhydride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance monomers play in the development of next-generation polyimide materials for the electronics and aerospace industries. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative synthesis methods described in CN1850814A can be seamlessly transferred from the laboratory to full-scale manufacturing. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify isomer ratios and color values, thereby guaranteeing the performance consistency required by our global partners. Our infrastructure is designed to handle complex chemistries safely and efficiently, providing a secure foundation for your long-term supply needs.
We invite you to collaborate with us to leverage this advanced technology for your specific product lines. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this chlorophthalimide-based route for your operations. We encourage you to contact us today to discuss your requirements, obtain specific COA data for our available isomers, and receive comprehensive route feasibility assessments tailored to your production goals. Let us be your partner in driving innovation and efficiency in the high-performance polymer sector.
