Advanced Synthesis of BTMI-DMDPM for High-Performance Polymer Applications and Commercial Scale-Up
The landscape of high-performance polymer additives is constantly evolving, driven by the demand for materials that can withstand extreme thermal and mechanical stress. A pivotal advancement in this sector is detailed in patent CN100503569C, which outlines a robust method for preparing 4,4'-bis(4-carboxyphthalimide)-3,3'-dimethyldiphenylmethane, commonly known as BTMI-DMDPM. This compound serves as a critical monomer for polyamide-imide and polyester-imide resins, as well as a superior heat-resistant modifier for epoxy systems. The technical breakthrough lies in its ability to achieve exceptional purity levels exceeding 99% through a streamlined imidization process. For R&D directors and procurement specialists seeking a reliable polymer additive supplier, understanding the nuances of this synthesis route is essential for securing a competitive edge in material performance. The methodology described eliminates many of the traditional bottlenecks associated with phthalimide synthesis, offering a pathway that is not only chemically efficient but also environmentally considerate. By leveraging azeotropic dehydration techniques, the process ensures that the reaction equilibrium is driven towards completion without the need for excessive reagent excess or harsh conditions that typically degrade product quality. This patent represents a significant leap forward in the commercial scale-up of complex polymer additives, providing a foundation for manufacturing processes that are both economically viable and technically superior.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of bis-phthalimide derivatives has been plagued by several inherent challenges that hinder large-scale production and consistent quality control. Traditional routes often rely on high-temperature fusion methods or the use of corrosive catalysts that introduce metallic impurities into the final product, necessitating expensive and time-consuming purification steps. These conventional approaches frequently suffer from incomplete imidization, leading to a product spectrum contaminated with mono-imide intermediates and unreacted anhydrides, which can severely compromise the thermal stability of the resulting polymer matrix. Furthermore, many legacy processes operate under vacuum or high pressure, requiring specialized reactor vessels that increase capital expenditure and pose significant safety risks during operation. The inability to effectively remove water generated during the imidization reaction often results in hydrolysis of the anhydride starting material, drastically reducing overall yield and increasing the cost per kilogram of the active ingredient. For supply chain heads, these inefficiencies translate into unpredictable lead times and batch-to-batch variability, making it difficult to guarantee the stringent specifications required by downstream aerospace and electronics applications. The environmental footprint of these older methods is also substantial, often generating large volumes of hazardous waste that require complex treatment protocols before disposal.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data introduces a sophisticated solution that addresses these pain points through precise stoichiometric control and solvent engineering. By utilizing a molar ratio of 1.00:1.98 to 2.02 between 4,4'-diamino-3,3'-dimethyldiphenylmethane (MDT) and trimellitic anhydride (TMA), the process ensures that the reaction proceeds with minimal side products while maximizing the conversion rate. The introduction of a strong polar aprotic organic solvent, such as NMP or DMF, facilitates the formation of a homogeneous solution at room temperature, allowing for excellent mixing and heat transfer before the imidization even begins. The key innovation lies in the subsequent addition of an azeotropic dehydrating agent, which allows for the continuous removal of water generated during the reaction at reflux temperatures between 85°C and 180°C. This dynamic removal of byproduct water drives the equilibrium towards the desired imide product without requiring extreme temperatures that could cause decomposition. For a reliable polymer additive supplier, this method offers a distinct advantage in terms of process robustness, as it can be performed under atmospheric pressure using standard glass-lined or stainless-steel reactors. The result is a manufacturing protocol that is not only simpler to operate but also inherently safer and more scalable, directly addressing the needs of procurement managers looking for cost reduction in polymer additive manufacturing.
Mechanistic Insights into Azeotropic Dehydration Imidization
The core of this synthesis lies in the nucleophilic attack of the amine groups on the anhydride rings, followed by a dehydration cyclization to form the stable imide structure. In the initial phase, the MDT and TMA react in the polar aprotic solvent to form an amic acid intermediate. This step is exothermic and occurs readily at room temperature, creating a homogeneous solution that is crucial for uniform reaction kinetics. However, the conversion of the amic acid to the final imide is an equilibrium reaction that produces water as a byproduct. If this water is not removed, it can hydrolyze the anhydride or reverse the imidization, leading to low yields and impure products. The patent specifies the use of an azeotropic agent like toluene or xylene, which forms a low-boiling azeotrope with water. Upon heating the mixture to reflux, the water is co-distilled out of the reaction system, effectively shifting the chemical equilibrium towards the product side according to Le Chatelier's principle. This mechanism ensures that the cyclization proceeds to completion, resulting in the high purity levels observed in the experimental data. The choice of solvent system is critical; the polar aprotic solvent keeps the intermediates in solution, preventing premature precipitation which could trap impurities, while the azeotropic agent handles the water removal efficiently.
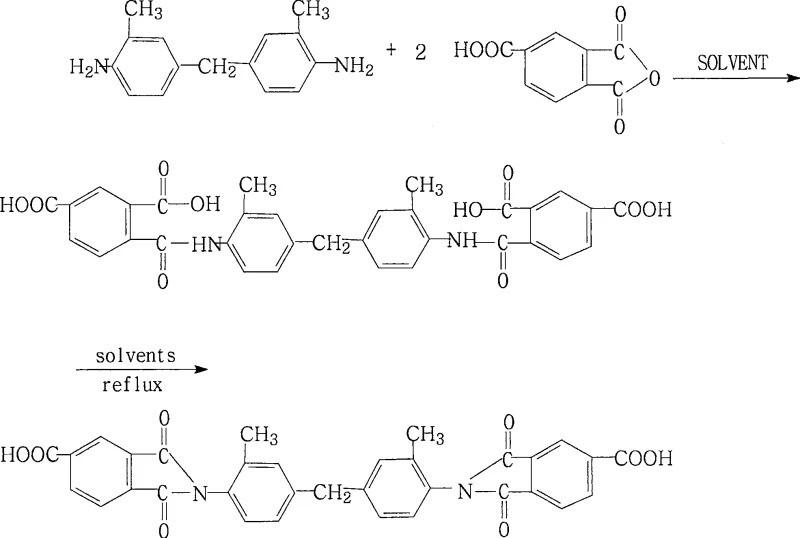
Controlling the impurity profile is another critical aspect of this mechanistic pathway, particularly for applications in electronic materials where ionic contamination must be minimized. The strict control of the molar ratio between the diamine and the anhydride is essential to prevent the formation of oligomers or unreacted starting materials that could act as plasticizers in the final resin. The patent data indicates that maintaining the TMA ratio between 1.98 and 2.02 relative to MDT provides a slight excess that ensures complete consumption of the diamine without leaving a significant surplus of anhydride that would require difficult removal steps. Furthermore, the post-reaction workup involves cooling the solution and adding water to precipitate the product. This step leverages the solubility differences between the high-molecular-weight imide product and the soluble impurities or residual solvents. The ability to wash the precipitate with cold solvent further enhances the purity by removing surface-adsorbed impurities. For R&D teams, understanding this mechanism allows for fine-tuning of the process parameters to optimize the particle size and morphology of the final powder, which can influence the processing characteristics of the compound in downstream compounding operations. The combination of precise stoichiometry and effective water removal creates a robust process window that is forgiving enough for industrial production yet precise enough for high-specification applications.
How to Synthesize 4,4'-bis(4-carboxyphthalimide)-3,3'-dimethyldiphenylmethane Efficiently
Implementing this synthesis route in a production environment requires careful attention to the sequence of addition and temperature control to ensure safety and reproducibility. The process begins with the dissolution of the diamine in the polar solvent, followed by the gradual addition of the anhydride to manage the exotherm. Once the amic acid stage is complete, the azeotropic agent is introduced, and the system is heated to reflux. The duration of the reflux, ranging from 2 to 12 hours depending on the specific solvent combination, is critical for achieving full conversion. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results.
- Mix MDT and TMA in a polar aprotic solvent at room temperature to form a homogeneous solution.
- Add an azeotropic dehydrating agent and heat to reflux between 85°C and 180°C for 2 to 12 hours.
- Recover solvents, cool the reaction, add water to precipitate the product, then filter and vacuum dry.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis method offers profound benefits that extend beyond mere chemical yield, directly impacting the bottom line and supply chain resilience for global buyers. The ability to operate under atmospheric pressure significantly reduces the capital investment required for reactor infrastructure, as there is no need for expensive high-pressure vessels or complex vacuum systems. This simplification of equipment translates into lower maintenance costs and reduced downtime, ensuring a more consistent supply of material to meet market demand. For procurement managers, the most compelling advantage lies in the solvent recovery system integrated into the process. The patent explicitly states that both the azeotropic dehydrating agent and the polar aprotic solvent can be recovered and reused repeatedly. This closed-loop solvent management drastically reduces the consumption of raw materials, leading to substantial cost savings in manufacturing without compromising on quality. By minimizing solvent waste, the process also lowers the environmental compliance burden, reducing the costs associated with waste disposal and regulatory reporting. These factors combine to create a highly competitive cost structure that allows suppliers to offer high-purity materials at more attractive price points.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the ability to recycle solvents multiple times creates a lean manufacturing model that significantly lowers the variable cost per unit. By avoiding the need for complex purification steps like column chromatography or extensive recrystallization, the process reduces labor and energy consumption. The high yield reported in the patent examples, often exceeding 90%, means that less raw material is wasted, further enhancing the economic efficiency of the production line. This economic efficiency is critical for maintaining margins in a competitive global market where price sensitivity is high.
- Enhanced Supply Chain Reliability: The simplicity of the reaction conditions, utilizing readily available commodity chemicals like toluene and NMP, ensures that the supply chain is not vulnerable to shortages of exotic reagents. The robustness of the process allows for flexible production scheduling, as the reaction time can be adjusted within a wide window (2 to 12 hours) without sacrificing product quality. This flexibility enables manufacturers to respond quickly to fluctuations in demand, reducing lead time for high-purity polymer additives. Furthermore, the stability of the intermediates and the final product ensures that inventory can be held safely without significant degradation, providing a buffer against supply disruptions.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by its suitability for industrial production mentioned in the patent. The absence of corrosive substances and the operation at atmospheric pressure make it easier to scale from pilot plant to full commercial capacity without encountering the engineering challenges typical of high-pressure chemistry. Additionally, the reduced generation of three wastes (waste gas, waste water, waste residue) aligns with increasingly stringent global environmental regulations. This compliance reduces the risk of production shutdowns due to environmental violations and enhances the brand reputation of the supplier as a responsible manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of BTMI-DMDPM, based on the specific details provided in the patent documentation. These answers are designed to clarify the capabilities of the manufacturing process and the quality of the resulting material for potential partners. Understanding these details is crucial for making informed sourcing decisions.
Q: What is the purity level achievable with this synthesis method?
A: According to patent CN100503569C, the described method consistently achieves a product purity of over 99%, making it suitable for high-performance resin applications.
Q: Can the solvents used in this process be recycled?
A: Yes, the process allows for the recovery and repeated use of both the azeotropic dehydrating agent and the polar aprotic solvent, significantly reducing waste and operational costs.
Q: Does this reaction require high-pressure equipment?
A: No, the reaction proceeds under atmospheric pressure, which simplifies equipment requirements and enhances operational safety for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-bis(4-carboxyphthalimide)-3,3'-dimethyldiphenylmethane Supplier
At NINGBO INNO PHARMCHEM, we recognize that the technical potential of a molecule is only realized through expert manufacturing and rigorous quality control. Our CDMO team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising chemistry described in patent CN100503569C can be translated into a reliable supply stream for your operations. We are committed to meeting stringent purity specifications through our rigorous QC labs, which utilize advanced analytical techniques to verify the identity and quality of every batch. Our facility is equipped to handle the specific solvent systems and thermal requirements of this imidization process, guaranteeing that the material you receive meets the high standards required for polyamide-imide and epoxy resin applications. We understand that consistency is key for your R&D and production teams, and we have optimized our processes to minimize batch-to-batch variation.
We invite you to collaborate with us to optimize your supply chain for this critical polymer additive. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that evaluates how switching to our manufacturing route can improve your overall material costs. We encourage you to contact us to request specific COA data and route feasibility assessments tailored to your project requirements. By partnering with us, you gain access to not just a product, but a comprehensive technical support system dedicated to your success in the high-performance materials market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
