Revolutionizing Polyimide Precursor Production: Advanced Synthesis of Diphenyl Sulfide Tetracarboxylic Acid
The landscape of high-performance polymer manufacturing is constantly evolving, driven by the demand for materials with superior thermal stability and mechanical strength. At the forefront of this innovation is the synthesis of diphenyl sulfide tetracarboxylic acid, a critical precursor for advanced polyimides used in electronics and aerospace applications. Patent CN1038676C introduces a groundbreaking methodology that fundamentally alters the traditional approach to constructing the diphenyl sulfide backbone. By replacing the conventional anhydrous sodium sulfide with a combination of elemental sulfur and anhydrous potassium carbonate, this technology addresses long-standing inefficiencies in raw material preparation and reaction control. For R&D directors and procurement specialists seeking a reliable electronic chemical supplier, understanding this shift is crucial for optimizing supply chains and reducing production costs. The implications of this patent extend beyond mere chemical curiosity; they represent a tangible pathway to more robust and economically viable manufacturing processes for complex organic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of diphenyl sulfide derivatives has relied heavily on anhydrous sodium sulfide as the primary sulfur source. However, the preparation of anhydrous sodium sulfide is fraught with logistical and technical challenges that can severely impact production efficiency. Typically, this reagent is derived from sodium sulfide nonahydrate through a rigorous vacuum dehydration process or via reaction with hydrogen sulfide and sodium ethylate. These preparatory steps are not only energy-intensive but also introduce significant variability in the quality of the sulfur source. Incomplete dehydration or exposure to air can lead to oxidation or residual moisture, which subsequently inhibits the nucleophilic substitution reaction, resulting in lower yields and inconsistent product quality. Furthermore, the handling of hydrogen sulfide poses serious safety and environmental concerns, complicating regulatory compliance and increasing operational overhead for manufacturing facilities. These cumulative factors create a bottleneck in the cost reduction in electronic chemical manufacturing, making the traditional route less attractive for large-scale commercial operations.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages the inherent stability and reactivity of elemental sulfur paired with anhydrous potassium carbonate. This strategic substitution eliminates the need for the precarious dehydration steps associated with sodium sulfide, thereby streamlining the entire upstream supply chain. Elemental sulfur is a stable, commercially abundant solid that does not require special atmospheric controls for storage, significantly enhancing supply chain reliability. When combined with potassium carbonate in a polar aprotic solvent, this system generates the necessary reactive sulfur species in situ under controlled thermal conditions. This method not only simplifies the operational workflow but also mitigates the risks associated with moisture sensitivity and oxidation. For supply chain heads, this translates to a more predictable procurement cycle and reduced dependency on specialized reagent suppliers. The ability to utilize standard, off-the-shelf chemicals while achieving high conversion rates represents a significant leap forward in process engineering, aligning perfectly with the goals of modern sustainable chemistry.
Mechanistic Insights into Sulfur-Mediated Nucleophilic Coupling
The core of this technological advancement lies in the mechanistic pathway of the sulfur-mediated coupling reaction. The process initiates with the activation of elemental sulfur by the base, anhydrous potassium carbonate, within the polar aprotic medium. This activation facilitates the formation of reactive polysulfide species or thiolate anions that act as potent nucleophiles. These species then attack the electrophilic carbon centers of the N-substituted halophthalimide, specifically targeting the carbon-halogen bond. The presence of the electron-withdrawing imide group enhances the electrophilicity of the aromatic ring, promoting efficient substitution even at moderate temperatures ranging from 130°C to 200°C. The reaction proceeds through a concerted mechanism that ensures the formation of the diphenyl sulfide linkage with high regioselectivity. Understanding this mechanism is vital for R&D teams aiming to optimize reaction parameters such as temperature profiles and stoichiometric ratios to maximize yield and minimize byproduct formation.
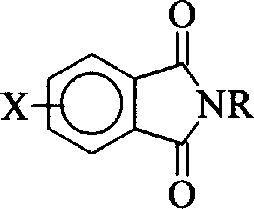
Controlling the impurity profile is another critical aspect of this synthesis, particularly for applications requiring high-purity OLED material or semiconductor grade polymers. The use of elemental sulfur minimizes the introduction of oxygen-containing impurities that often arise from the oxidation of sulfide salts. Additionally, the choice of solvent plays a pivotal role in solubilizing the intermediate species and preventing premature precipitation, which can trap impurities. The subsequent hydrolysis step, converting the tetraimide to the tetracarboxylic acid, is conducted under alkaline conditions that effectively cleave the imide rings without degrading the sensitive sulfide bridge. This two-stage process allows for intermediate purification, ensuring that the final product meets stringent purity specifications. For manufacturers, this level of control over the impurity spectrum is essential for maintaining the performance characteristics of the final polyimide films, such as dielectric constant and thermal expansion coefficient.
How to Synthesize Diphenyl Sulfide Tetracarboxylic Acid Efficiently
Implementing this synthesis route requires careful attention to reaction conditions and stoichiometry to ensure optimal outcomes. The process begins by charging a reactor with N-substituted halophthalimide, elemental sulfur, and anhydrous potassium carbonate in a molar ratio that favors complete conversion while minimizing excess reagent waste. The mixture is suspended in a polar aprotic solvent such as DMSO or NMP and heated under a nitrogen atmosphere to prevent oxidative degradation. Detailed standardized synthesis steps see the guide below.
- React N-substituted halophthalimide with elemental sulfur and anhydrous potassium carbonate in a polar aprotic solvent at 130-200°C under nitrogen.
- Precipitate the resulting N,N'-disubstituted diphenyl sulfide tetraimide by pouring the reaction mixture into water or ethanol.
- Hydrolyze the tetraimide intermediate using aqueous sodium hydroxide, followed by acidification to isolate the final tetracarboxylic acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis method offers profound advantages for procurement managers and supply chain strategists. The primary benefit stems from the drastic simplification of the raw material supply chain. By eliminating the need for custom-dehydrated sodium sulfide, companies can source elemental sulfur and potassium carbonate from a wide range of global suppliers, reducing geopolitical risk and price volatility. This diversification of the supply base enhances overall supply chain resilience, ensuring continuity of production even during market disruptions. Moreover, the stability of these reagents reduces storage costs and safety liabilities, contributing to a leaner and more efficient operational model. The ability to scale this process from laboratory benchtop to multi-ton production without significant re-engineering further underscores its commercial viability.
- Cost Reduction in Manufacturing: The elimination of the energy-intensive dehydration process for sodium sulfide directly translates to significant utility savings. Additionally, the higher stability of the reagents reduces waste generation and the need for expensive disposal protocols. The simplified workflow also lowers labor costs associated with complex reagent preparation and handling. By avoiding the use of hazardous gases like hydrogen sulfide, facilities can save on specialized ventilation and safety equipment maintenance. These cumulative efficiencies result in a substantially lower cost of goods sold, providing a competitive edge in the pricing of high-performance polymer precursors.
- Enhanced Supply Chain Reliability: Elemental sulfur and potassium carbonate are commodity chemicals with robust global production networks, ensuring consistent availability. Unlike specialized sulfide reagents that may have long lead times or limited suppliers, these inputs can be procured with short notice. This reliability allows for just-in-time inventory strategies, reducing capital tied up in raw material stockpiles. Furthermore, the reduced sensitivity to moisture means that transportation and storage requirements are less stringent, lowering logistics costs. For supply chain heads, this predictability is invaluable for maintaining steady production schedules and meeting customer delivery commitments without interruption.
- Scalability and Environmental Compliance: The process operates in standard polar aprotic solvents that are well-understood in industrial settings, facilitating easy scale-up from pilot plants to full commercial production. The absence of toxic gas evolution simplifies废气 treatment systems, ensuring compliance with increasingly strict environmental regulations. The solid nature of the sulfur reagent also minimizes spill risks compared to liquid sulfide solutions. Waste streams are easier to manage and treat, reducing the environmental footprint of the manufacturing facility. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate sustainability profile, appealing to environmentally conscious downstream customers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology. They are derived from the specific pain points identified in traditional manufacturing methods and the beneficial effects documented in the patent literature. These insights are intended to clarify the operational feasibility and strategic value of adopting this novel route for your production needs.
Q: What is the primary advantage of using elemental sulfur over anhydrous sodium sulfide?
A: Using elemental sulfur eliminates the complex and energy-intensive dehydration process required for sodium sulfide nonahydrate, significantly simplifying raw material preparation and improving reaction consistency.
Q: What solvents are compatible with this synthesis method?
A: The process is robust across various polar aprotic solvents including dimethyl sulfoxide (DMSO), dimethylformamide (DMF), N,N-dimethylacetamide, and N-methyl pyrrolidone, offering flexibility for scale-up.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the method utilizes standard industrial reagents and avoids moisture-sensitive intermediates, making it highly scalable and reliable for continuous commercial manufacturing of polyimide precursors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diphenyl Sulfide Tetracarboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality precursors play in the development of next-generation electronic materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from R&D to mass manufacturing is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of diphenyl sulfide tetracarboxylic acid meets the exacting standards required for polyimide synthesis. Our commitment to technical excellence allows us to support complex custom synthesis projects with confidence and precision.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your project goals. Let us help you engineer a more efficient and reliable production future.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
