Advanced Synthesis of N,N'-Bis(4-Ethoxycarbonylphenyl)-N'-Benzylformamidine for Industrial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes that balance high purity with economic efficiency, particularly for functional additives like UV absorbers. Patent CN101481331B introduces a refined methodology for the preparation of N,N'-bis(4-ethoxycarbonylphenyl)-N'-benzylformamidine, a critical intermediate widely utilized in plastics, resins, and cosmetics to prevent photodegradation. Unlike earlier iterations that suffered from cumbersome multi-step sequences or reliance on expensive reagents, this invention streamlines the production into three distinct phases: formylation, N-alkylation, and condensation. By shifting the alkylation environment to an aprotic dipolar solvent system and substituting costly benzyl bromide with benzyl chloride, the process achieves a total yield of approximately 55% with product purity exceeding 99%. This technical advancement represents a significant leap forward for manufacturers aiming to secure a reliable UV absorber intermediate supplier capable of delivering consistent quality at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of formamidine derivatives has been plagued by inefficiencies that drive up operational expenditures and complicate supply chain logistics. Early methods, such as those described in US4,021,471, relied on a lengthy sequence involving aniline monoacetylation, amide alkylation, and subsequent hydrolysis just to prepare the necessary N-alkylaniline starting material. This extended synthetic route not only increased the consumption of solvents and reagents but also introduced multiple points where yield loss could occur, ultimately inflating the cost per kilogram of the final product. Furthermore, more recent attempts like CN1956947A, while shortening the route, still depended on a biphasic water/toluene system requiring tert-butyl ammonium bromide as a phase transfer catalyst. This approach necessitated the use of expensive benzyl bromide and resulted in a disappointing total yield of merely 37.8%, rendering it economically unviable for cost reduction in fine chemical manufacturing where margin compression is a constant pressure.
The Novel Approach
The methodology outlined in CN101481331B fundamentally re-engineers the alkylation step to overcome these historical bottlenecks, offering a pathway that is both chemically elegant and commercially viable. By transitioning the N-alkylation reaction from a heterogeneous two-phase system to a homogeneous aprotic dipolar solvent environment, the inventors have eliminated the need for phase transfer catalysts entirely. This shift allows for the use of inexpensive inorganic weak bases like potassium carbonate alongside the much cheaper benzyl chloride, replacing the costly benzyl bromide without sacrificing reactivity. The result is a dramatic improvement in the yield of the alkylation intermediate, which can reach as high as 99%, thereby significantly boosting the overall throughput of the synthesis. This streamlined approach not only simplifies the post-treatment process to basic solid-liquid separation but also ensures that the commercial scale-up of complex polymer additives and UV stabilizers becomes far more predictable and profitable.
Mechanistic Insights into N-Alkylation and Condensation Dynamics
The core innovation of this synthesis lies in the mechanistic optimization of the N-alkylation step, where the choice of solvent plays a pivotal role in determining reaction kinetics and selectivity. In the conventional biphasic systems, the reaction rate is often limited by the mass transfer of ions across the interface between the aqueous and organic layers, necessitating aggressive stirring and phase transfer agents to facilitate contact. In contrast, the use of aprotic dipolar solvents such as N,N-dimethylformamide (DMF) or dimethyl sulfoxide (DMSO) creates a homogeneous medium where the nucleophilicity of the amide nitrogen is significantly enhanced due to the lack of hydrogen bonding solvation. This environment allows the inorganic weak base to effectively deprotonate the formamide nitrogen, generating a highly reactive nucleophile that readily attacks the benzyl chloride. The reaction proceeds smoothly at moderate temperatures of 50-70°C over 20-25 hours, minimizing side reactions and ensuring that the formation of the N-benzyl-4-formamidobenzoate intermediate occurs with exceptional fidelity and minimal byproduct generation.
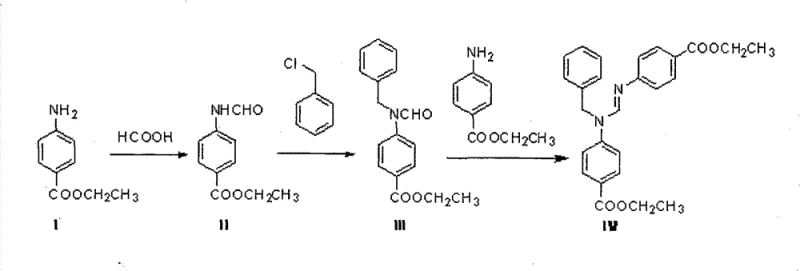
Following the successful alkylation, the final condensation step employs phosphorus oxychloride (POCl3) as a potent dehydrating agent within a halogenated hydrocarbon solvent like chloroform. This step involves the reaction of the N-benzyl intermediate with a second equivalent of ethyl p-aminobenzoate to form the final formamidine linkage. The mechanism likely involves the activation of the formyl group by POCl3 to form an imidoyl chloride species, which is then attacked by the amine nucleophile. The subsequent elimination of HCl drives the equilibrium towards the product, and the use of a halogenated solvent ensures that all reactants remain in solution during the reflux period of 15-17 hours. Crucially, the workup involves a simple neutralization with sodium hydroxide solution, which hydrolyzes any excess activating agent and facilitates the precipitation of the crude product. This mechanistic clarity allows for rigorous impurity control, ensuring that the final recrystallized solid meets the stringent purity specifications required for high-purity OLED material or pharmaceutical additive applications.
How to Synthesize N,N'-Bis(4-Ethoxycarbonylphenyl)-N'-Benzylformamidine Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize the benefits of the novel solvent system and reagent choices. The process begins with the formylation of ethyl p-aminobenzoate in toluene, followed by the critical alkylation in DMF, and concludes with the condensation in chloroform. Each step is designed to minimize purification overhead, allowing intermediates to be carried forward with simple washing rather than extensive chromatography. For R&D teams looking to replicate or adapt this chemistry, understanding the specific molar ratios and temperature profiles is essential to achieving the reported 55% total yield. The detailed standardized synthesis steps see the guide below.
- Perform formylation of ethyl p-aminobenzoate with formic acid in toluene at 90-120°C to obtain ethyl 4-formamidobenzoate.
- Conduct N-alkylation using benzyl chloride and inorganic weak base in an aprotic dipolar solvent like DMF at 50-70°C.
- Execute condensation with ethyl p-aminobenzoate in halogenated hydrocarbon using a dehydrating agent, followed by recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process translates directly into tangible operational improvements and risk mitigation strategies. The shift away from expensive benzyl bromide to commodity-grade benzyl chloride removes a significant variable cost driver, while the elimination of phase transfer catalysts simplifies the bill of materials. Furthermore, the high yield of the alkylation step reduces the volume of raw materials required per unit of output, effectively lowering the cost of goods sold without compromising on quality. This efficiency is compounded by the simplified workup procedures, which reduce solvent consumption and waste disposal costs, aligning with modern environmental compliance standards. Consequently, this route offers a compelling value proposition for reducing lead time for high-purity UV absorber intermediates by ensuring a more stable and predictable production schedule.
- Cost Reduction in Manufacturing: The substitution of benzyl bromide with benzyl chloride represents a direct raw material cost saving, as benzyl chloride is significantly less expensive and more readily available in the global chemical market. Additionally, the removal of the phase transfer catalyst eliminates another line item from the production cost structure, while the high yield of the alkylation step minimizes the loss of valuable intermediates. These factors combine to create a leaner manufacturing process that maximizes output from every kilogram of input, driving substantial cost savings in the overall production budget.
- Enhanced Supply Chain Reliability: By relying on common, non-proprietary reagents such as potassium carbonate and DMF, the supply chain becomes more resilient to disruptions that might affect specialized catalysts or exotic halides. The robustness of the reaction conditions, which tolerate slight variations in temperature and stoichiometry without significant yield penalties, further ensures consistent batch-to-batch performance. This reliability is crucial for maintaining continuous supply lines to downstream customers in the plastics and cosmetic industries, where interruptions can be costly.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on standard unit operations like reflux, filtration, and recrystallization, which are easily replicated in large-scale reactors. The simplified post-treatment, which avoids complex extractions or chromatographic separations, reduces the generation of hazardous waste streams and lowers the burden on wastewater treatment facilities. This alignment with green chemistry principles not only facilitates regulatory approval but also enhances the sustainability profile of the final product, making it more attractive to environmentally conscious end-users.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific improvements highlighted in the patent data, focusing on yield optimization, reagent selection, and process scalability. Understanding these details is vital for technical teams evaluating the feasibility of adopting this method for their own production lines.
Q: Why is the new N-alkylation method superior to traditional phase transfer catalysis?
A: The new method utilizes an aprotic dipolar solvent system which eliminates the need for expensive phase transfer catalysts and achieves significantly higher yields compared to the traditional water/toluene two-phase system.
Q: What are the cost advantages of using benzyl chloride over benzyl bromide?
A: Benzyl chloride is substantially cheaper than benzyl bromide while maintaining high reactivity under the optimized conditions described in the patent, leading to direct raw material cost reductions.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process features simple workup procedures such as solid-liquid separation and recrystallization, avoiding complex purification steps, which makes it highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N,N'-Bis(4-Ethoxycarbonylphenyl)-N'-Benzylformamidine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of processes like CN101481331B are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of N,N'-bis(4-ethoxycarbonylphenyl)-N'-benzylformamidine meets the exacting standards required for high-performance UV protection applications. Our commitment to quality assurance means that you can rely on us for consistent supply without the variability often associated with smaller-scale producers.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific application needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic impact of switching to this more efficient manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring that your supply chain is built on a foundation of scientific excellence and commercial viability.
