Scalable Synthesis of Fluorinated Polyimide Monomers for Advanced Material Applications
Scalable Synthesis of Fluorinated Polyimide Monomers for Advanced Material Applications
The development of high-performance polymers relies heavily on the availability of specialized monomers that can impart unique thermal and mechanical properties to the final resin. Patent CN101230031B introduces a robust and industrially viable preparation method for 2,2-bis[3-(4-carboxyphthalimido)-4-hydroxyphenyl]hexafluoropropane, a critical building block for advanced polyimide and polyester-imide resins. This specific fluorinated monomer is essential for creating materials with exceptional heat resistance and dielectric properties, which are increasingly demanded in the electronics and aerospace sectors. The patented process distinguishes itself by combining simplicity with high efficiency, utilizing a one-pot reaction strategy that minimizes unit operations and maximizes throughput. By addressing the historical challenges associated with synthesizing complex fluorinated imides, this technology offers a reliable pathway for manufacturers seeking to secure a stable supply of high-purity electronic chemical intermediates.
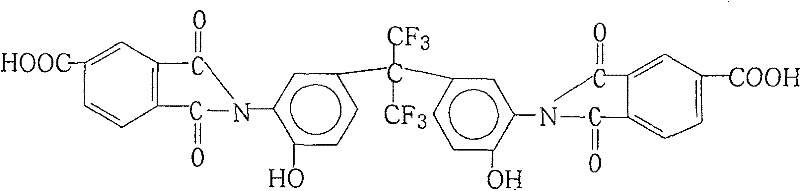
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of complex imide monomers often involves multi-step procedures that require the isolation and purification of intermediate amic acids before cyclization can occur. These conventional routes are frequently plagued by low overall yields due to material losses during multiple filtration and drying stages, which significantly inflates the cost of goods sold. Furthermore, older methods may rely on harsh chemical dehydrating agents that generate substantial amounts of hazardous waste, creating severe environmental compliance burdens for production facilities. The need for stringent temperature controls and extended reaction times in traditional processes also limits the scalability, making it difficult to transition from laboratory benchtop quantities to commercial metric ton production without encountering heat transfer issues. Consequently, supply chains for these specialized intermediates have historically been fragile, with long lead times and inconsistent quality batches that disrupt downstream polymer manufacturing schedules.
The Novel Approach
The methodology described in the patent revolutionizes this landscape by employing a direct, one-pot imidization technique that seamlessly transitions from amidation to cyclodehydration within a single reactor vessel. By carefully selecting a system of strong polar aprotic solvents combined with specific azeotropic dehydrating agents, the process drives the equilibrium towards the desired imide product without the need for intermediate isolation. This approach not only streamlines the operational workflow but also allows for the efficient recovery and recycling of expensive solvents, thereby drastically reducing the environmental footprint and raw material costs. The ability to conduct the reaction under relatively mild initial conditions followed by controlled reflux ensures that the delicate fluorinated structures remain intact, preventing degradation and ensuring high structural fidelity. This innovation represents a significant leap forward in cost reduction in electronic chemical manufacturing, providing a sustainable and economically attractive solution for large-scale production.
Mechanistic Insights into Azeotropic Dehydration Imidization
The core of this synthesis lies in the precise control of the reaction environment to facilitate the conversion of amino and anhydride groups into stable imide rings. Initially, the diamine BAHPFP and trimellitic anhydride (TMA) react at room temperature in a polar aprotic solvent to form a soluble poly(amic acid) intermediate, ensuring a homogeneous phase before the energy-intensive cyclization step begins. 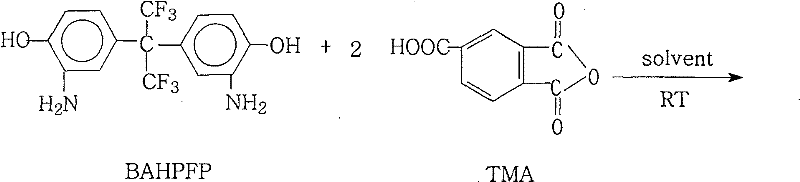 Once the homogeneous solution is established, the addition of an azeotropic agent such as xylene or toluene allows for the continuous removal of water generated during the ring-closing reaction. As the temperature is raised to between 85°C and 210°C, the azeotropic mixture boils, carrying away water vapor and shifting the chemical equilibrium decisively towards the formation of the cyclic imide structure. This mechanism effectively bypasses the kinetic barriers often associated with thermal imidization in solid state or viscous media, resulting in a cleaner reaction profile with fewer side products. The use of an inert nitrogen atmosphere throughout the process further protects the reactive species from oxidative degradation, ensuring that the final product maintains its intended chemical integrity and color characteristics.
Once the homogeneous solution is established, the addition of an azeotropic agent such as xylene or toluene allows for the continuous removal of water generated during the ring-closing reaction. As the temperature is raised to between 85°C and 210°C, the azeotropic mixture boils, carrying away water vapor and shifting the chemical equilibrium decisively towards the formation of the cyclic imide structure. This mechanism effectively bypasses the kinetic barriers often associated with thermal imidization in solid state or viscous media, resulting in a cleaner reaction profile with fewer side products. The use of an inert nitrogen atmosphere throughout the process further protects the reactive species from oxidative degradation, ensuring that the final product maintains its intended chemical integrity and color characteristics.
Impurity control is inherently built into this mechanistic design through the solubility differences between the product and the reaction byproducts. As the imidization proceeds to completion, the resulting 2,2-bis[3-(4-carboxyphthalimido)-4-hydroxyphenyl]hexafluoropropane exhibits specific solubility characteristics that allow it to be precipitated cleanly upon the addition of water to the cooled reaction mixture. This precipitation step acts as a crude purification, separating the target molecule from residual solvents and unreacted starting materials which remain in the aqueous-organic phase. Subsequent washing with cold polar aprotic solvents removes any adhering impurities without dissolving the product cake, leading to a final material with purity levels consistently exceeding 99%. This high degree of purity is critical for downstream applications where trace impurities could act as defect sites in polymer films or compromise the dielectric strength of the final electronic material.
How to Synthesize 2,2-Bis[3-(4-Carboxyphthalimido)-4-Hydroxyphenyl]Hexafluoropropane Efficiently
Implementing this synthesis route requires careful attention to solvent ratios and temperature profiles to maximize yield and minimize cycle time. The process begins with the dissolution of the fluorinated diamine and the anhydride in a solvent like N-methyl-2-pyrrolidone (NMP) under nitrogen, followed by the strategic addition of a dehydrating agent to initiate the reflux. Detailed standard operating procedures regarding specific molar ratios, heating ramps, and workup protocols are essential for reproducibility and safety.
- Dissolve BAHPFP and TMA in a polar aprotic solvent like NMP at room temperature under inert atmosphere to form a homogeneous amic acid solution.
- Add an azeotropic dehydrating agent such as xylene or toluene and heat the mixture to reflux between 85°C and 210°C.
- Maintain reflux for 2 to 18 hours to complete imidization, then recover solvents, cool, precipitate with water, and dry the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process translates directly into tangible economic and operational benefits that strengthen the overall value proposition of the supply base. The elimination of intermediate isolation steps reduces the total processing time and labor requirements, leading to a more streamlined production schedule that can respond faster to market demand fluctuations. Moreover, the capability to recycle solvents repeatedly without significant loss of performance creates a closed-loop system that insulates the manufacturing cost from volatile raw material price swings. This stability is crucial for long-term contracting and budget forecasting, allowing partners to lock in favorable pricing structures that are not susceptible to short-term market volatility. The simplified equipment requirements, operating at atmospheric pressure without the need for exotic corrosion-resistant alloys, further lower the capital expenditure barrier for scaling up production capacity.
- Cost Reduction in Manufacturing: The integrated solvent recovery system allows for the reclamation of both the polar aprotic solvent and the azeotropic agent, significantly lowering the variable cost per kilogram of product. By avoiding the purchase of fresh solvents for every batch and reducing waste disposal fees, the overall production economics are vastly improved compared to traditional multi-step syntheses. This efficiency gain enables the supplier to offer competitive pricing models that reflect the true optimization of the chemical process rather than passing on inefficiency costs to the buyer.
- Enhanced Supply Chain Reliability: The robustness of the one-pot reaction reduces the risk of batch failures and off-spec material, ensuring a consistent flow of high-quality intermediates to downstream customers. Since the process does not rely on hard-to-source reagents or complex catalytic systems, the supply chain is less vulnerable to disruptions caused by raw material shortages. This reliability fosters stronger partnerships and allows manufacturers to maintain leaner inventory levels without fearing stockouts of critical monomer supplies.
- Scalability and Environmental Compliance: The absence of corrosive byproducts and the operation under normal pressure simplify the engineering requirements for large-scale reactors, facilitating a smoother transition from pilot plant to full commercial production. Additionally, the reduced generation of three wastes (waste water, gas, and solids) aligns with increasingly stringent global environmental regulations, minimizing the risk of regulatory shutdowns or fines. This environmental stewardship enhances the corporate social responsibility profile of the supply chain, appealing to end-users who prioritize sustainable sourcing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this fluorinated monomer. These answers are derived directly from the experimental data and process descriptions found in the underlying patent documentation to ensure accuracy and relevance. Understanding these details helps stakeholders make informed decisions about integrating this material into their specific product formulations.
Q: What are the key advantages of this synthesis method over traditional routes?
A: This method utilizes a one-pot procedure that eliminates the need for isolating intermediate amic acids, significantly simplifying the workflow and reducing solvent consumption while achieving yields exceeding 93%.
Q: Can the solvents used in this process be recycled?
A: Yes, the process explicitly includes a step for recovering both the azeotropic dehydrating agent and the polar aprotic solvent, allowing for repeated reuse which drastically lowers raw material costs and environmental impact.
Q: What is the typical purity level achievable with this protocol?
A: Experimental data from the patent indicates that the final product consistently achieves purity levels above 99.0%, making it suitable for high-performance polymer applications requiring stringent quality control.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,2-Bis[3-(4-Carboxyphthalimido)-4-Hydroxyphenyl]Hexafluoropropane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance monomers play in the development of next-generation polyimide materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We operate stringent purity specifications and utilize rigorous QC labs to verify that every batch of 2,2-bis[3-(4-carboxyphthalimido)-4-hydroxyphenyl]hexafluoropropane meets the exacting standards required for electronic and aerospace applications. Our commitment to quality assurance means that you receive a product that performs predictably in your polymerization processes, reducing your own production risks.
We invite you to contact our technical procurement team to discuss how our optimized synthesis route can benefit your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our efficient manufacturing process translates into better value for your organization. We are ready to provide specific COA data and route feasibility assessments to support your R&D and sourcing strategies, ensuring a seamless integration of our materials into your supply chain.
