Advanced Synthesis of 2,3,3',4'-Diphenyl Ether Tetracarboxylic Acid for High-Performance Polyimide Production
Introduction to Advanced Polyimide Monomer Synthesis
The global demand for high-performance polyimides in aerospace, microelectronics, and liquid-crystal display sectors has necessitated the development of more efficient synthetic routes for their key monomers. Patent CN102557925A introduces a groundbreaking method for synthesizing 2,3,3',4'-diphenyl ether tetracarboxylic acid, a critical precursor for 3,4'-ODPA, which imparts superior thermal stability and solubility to final polymer products. Unlike traditional methods that struggle with low yields and severe environmental pollution, this innovation utilizes a controlled dilute nitric acid oxidation system. By implementing a sophisticated two-stage heating protocol, the process effectively mitigates the risk of ether bond cleavage and unwanted nitration, ensuring the production of high-purity intermediates essential for next-generation electronic materials. This technical advancement represents a pivotal shift towards greener and more economically viable manufacturing practices in the specialty chemicals industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the oxidation of methyl groups to carboxyl groups in diphenyl ether derivatives has relied heavily on potassium permanganate or high-temperature air oxidation, both of which present substantial industrial drawbacks. The potassium permanganate method, often conducted in pyridine solutions, generates massive quantities of manganese dioxide sludge, creating a severe burden on waste treatment facilities and drastically inflating operational costs due to heavy metal disposal requirements. Alternatively, gas-phase air oxidation techniques typically demand extreme temperatures ranging from 300°C to 450°C and require complex catalyst systems like cobalt-manganese-bromine, which pose significant safety risks and operational difficulties in maintaining consistent selectivity. These conventional pathways frequently suffer from low conversion rates and the formation of intractable byproducts, making them increasingly unsuitable for the rigorous purity standards demanded by modern high-tech applications such as flexible circuit boards and advanced composite materials.
The Novel Approach
In stark contrast to these legacy technologies, the novel approach detailed in the patent employs dilute nitric acid as a benign yet potent oxidant within a closed pressure system, fundamentally altering the economic and environmental landscape of production. This method eliminates the need for expensive transition metal catalysts and avoids the generation of solid heavy metal waste, thereby simplifying the downstream purification process to mere crystallization and washing. The strategic use of a two-stage temperature profile allows for precise control over the reaction kinetics, ensuring that the oxidation proceeds smoothly without degrading the sensitive ether linkage that is crucial for the polymer's final mechanical properties. By operating at moderate temperatures between 120°C and 220°C, the process achieves a remarkable balance between reaction rate and selectivity, resulting in a product with purity levels exceeding 99% and yields consistently between 75% and 90%. This streamlined workflow not only reduces raw material consumption but also significantly shortens the overall production cycle time.
Mechanistic Insights into Dilute Nitric Acid Oxidation
The core of this synthesis lies in the meticulous control of oxidative conditions to favor the conversion of methyl groups into carboxylic acids while preserving the structural integrity of the diphenyl ether backbone. The reaction mechanism involves the generation of reactive nitrogen species from the dilute nitric acid under elevated pressure, which selectively attack the benzylic positions of the methyl substituents. The initial heating phase at 120°C to 150°C serves as a critical induction period where the oxidation begins gently, preventing the violent exothermic reactions often associated with concentrated nitric acid that could lead to ring nitration. As the reaction progresses, the temperature is ramped up to 180°C to 220°C, providing the necessary activation energy to drive the oxidation of the intermediate aldehyde or alcohol species fully to the carboxylic acid state. This staged thermal input is essential for managing the internal pressure, which is maintained between 1MPa and 3MPa to keep the nitric acid in a highly reactive liquid phase without causing decomposition of the solvent or the substrate.
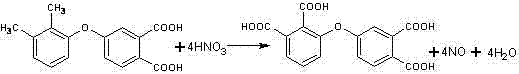
Furthermore, the choice of dilute nitric acid concentration, specifically between 10% and 40%, plays a pivotal role in suppressing side reactions that typically plague aromatic oxidations. High concentrations of nitric acid are notorious for electrophilic aromatic substitution, which would introduce nitro groups onto the benzene rings, creating difficult-to-remove impurities that degrade the optical and electrical properties of the final polyimide. The optimized molar ratio of substrate to nitric acid, ranging from 1:4 to 1:10, ensures an excess of oxidant is available to drive the reaction to completion without creating an overly aggressive acidic environment. Post-reaction, the product is isolated through a controlled cooling crystallization process, often utilizing frozen water to maximize recovery rates. This physical separation method is highly effective because the target tetracarboxylic acid has distinct solubility characteristics compared to any minor unreacted starting materials or mono-oxidized intermediates, facilitating the achievement of the reported >99% purity without the need for complex chromatographic purification steps.
How to Synthesize 2,3,3',4'-Diphenyl Ether Tetracarboxylic Acid Efficiently
Implementing this synthesis route requires precise adherence to the thermal and pressure parameters defined in the patent to ensure reproducibility and safety on a commercial scale. The process begins with the charging of dimethyl-phenyl ether diacid and dilute nitric acid into a corrosion-resistant autoclave, followed by a carefully monitored heating ramp that transitions through two distinct thermal plateaus. Operators must maintain strict control over the internal pressure during the high-temperature phase to prevent safety incidents while ensuring the reaction mixture remains homogeneous. Detailed standard operating procedures regarding the cooling and crystallization phases are critical for maximizing yield, as rapid temperature drops can lead to the occlusion of impurities within the crystal lattice. For a comprehensive guide on the exact equipment specifications and safety protocols required for this transformation, please refer to the standardized synthesis steps outlined below.
- Mix dimethyl-phenyl ether diacid with 10-40% dilute nitric acid in a closed vessel and heat to 120-150°C for 1-2 hours.
- Increase temperature to 180-220°C while maintaining pressure at 1-3MPa for an additional 0.5-2 hours to complete oxidation.
- Cool the reaction mixture to below 80°C, release pressure, and crystallize the product using frozen water for 6-12 hours.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this dilute nitric acid oxidation technology translates directly into tangible operational efficiencies and risk mitigation strategies. The elimination of potassium permanganate and pyridine removes the logistical complexities associated with sourcing and handling hazardous heavy metal oxidants and toxic solvents, thereby reducing regulatory compliance burdens and insurance costs. Furthermore, the simplified workup procedure, which relies on crystallization rather than complex extraction or distillation, significantly lowers utility consumption and labor hours per batch, driving down the overall cost of goods sold. The high selectivity of the reaction minimizes the formation of tars and sludges, extending the lifespan of reactor vessels and reducing downtime for maintenance and cleaning. These factors collectively contribute to a more resilient supply chain capable of meeting the fluctuating demands of the electronics and aerospace industries without compromising on delivery schedules or product quality.
- Cost Reduction in Manufacturing: The shift away from expensive transition metal catalysts and the reduction in waste treatment volumes create a substantially leaner cost structure for producing high-purity polyimide intermediates. By avoiding the generation of heavy metal sludge, manufacturers can bypass costly disposal fees and environmental remediation processes, leading to significant long-term savings. Additionally, the high yield of the process means that less raw material is wasted, optimizing the atom economy and further enhancing the financial viability of large-scale production runs. The use of dilute nitric acid, a commodity chemical, ensures stable pricing and easy availability, shielding the supply chain from the volatility often seen with specialized reagents.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent output quality, which is critical for maintaining long-term contracts with downstream polymer manufacturers who require strict specification adherence. The simplicity of the raw material profile reduces the risk of supply disruptions caused by the scarcity of niche catalysts or solvents, allowing for more agile inventory management. Moreover, the shorter reaction times and streamlined isolation steps enable faster turnover of production batches, effectively increasing the throughput capacity of existing manufacturing facilities without the need for capital-intensive expansion. This agility allows suppliers to respond more rapidly to market spikes in demand for advanced electronic materials.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on standard pressure vessel technology and common industrial acids, making it suitable for expansion from pilot plants to multi-ton annual production capacities. The reduced environmental footprint, characterized by lower emissions and minimal solid waste, aligns perfectly with increasingly stringent global environmental regulations and corporate sustainability goals. This compliance advantage not only future-proofs the manufacturing operation against regulatory changes but also enhances the brand reputation of the supplier among eco-conscious clients in the high-tech sector. The ability to produce such a critical intermediate with such a clean profile is a distinct competitive advantage in the green chemistry landscape.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of 2,3,3',4'-diphenyl ether tetracarboxylic acid, derived directly from the proprietary data within the patent documentation. Understanding these nuances is vital for R&D teams evaluating this route for potential technology transfer or for procurement specialists assessing the quality implications of this manufacturing method. The answers provided reflect the specific advantages of the two-stage heating and dilute acid protocol over legacy oxidation techniques, highlighting the balance between performance and process safety.
Q: Why is dilute nitric acid preferred over potassium permanganate for this oxidation?
A: Potassium permanganate oxidation generates significant manganese dioxide sludge and requires pyridine, leading to high waste treatment costs and environmental pollution. Dilute nitric acid offers a cleaner profile with simpler post-treatment.
Q: How does the two-stage heating process improve product purity?
A: The initial lower temperature stage (120-150°C) initiates controlled oxidation without damaging the ether bond, while the second higher temperature stage (180-220°C) ensures complete conversion of methyl groups to carboxyls, minimizing nitrated byproducts.
Q: What is the typical yield and purity achievable with this method?
A: According to patent data, this method achieves a product purity exceeding 99% with yields ranging from 75% to 90%, significantly outperforming traditional high-temperature air oxidation methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3,3',4'-Diphenyl Ether Tetracarboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity monomers play in the performance of advanced polyimide materials used in cutting-edge electronics and aerospace applications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our expertise in handling complex oxidation chemistries allows us to optimize the dilute nitric acid process for maximum yield and minimal environmental impact, providing our partners with a sustainable and reliable source of this essential intermediate.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific polyimide development projects. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our optimized manufacturing process can reduce your overall material costs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us support your supply chain with the quality and consistency you demand for your high-performance material applications.
