Revolutionizing 3,3',4'-BTDA Production: A Safe, Scalable, and High-Yield Synthetic Route for Global Supply Chains
The chemical industry is currently witnessing a pivotal shift towards greener, more sustainable manufacturing processes, particularly for high-value intermediates like 3,3',4'-benzophenonetetracarboxylic dianhydride (3,3',4'-BTDA). As detailed in the recent patent CN115160271A, a groundbreaking preparation method has been disclosed that fundamentally alters the synthetic landscape for this critical polyimide precursor. This innovation moves away from hazardous reagents and stoichiometric oxidants, opting instead for a catalytic system driven by hydrogen peroxide and transition metals. For R&D Directors and Procurement Managers alike, this represents a dual opportunity: achieving higher purity specifications while simultaneously mitigating the supply chain risks associated with dangerous raw materials. The technology promises not only to enhance the safety profile of production facilities but also to deliver substantial economic benefits through improved yield and catalyst recyclability.
For global supply chain leaders, the implications of adopting this novel route are profound, especially when considering the stringent regulations surrounding chemical manufacturing in Europe and North America. By eliminating the need for bis(trichloromethyl) carbonate, a substance known for its high toxicity and handling complexity, manufacturers can significantly reduce their environmental liability and insurance costs. Furthermore, the switch to a catalytic oxidation system using hydrogen peroxide aligns perfectly with modern green chemistry principles, ensuring that the production of high-purity 3,3',4'-BTDA remains viable under increasingly strict environmental protocols. This patent serves as a blueprint for the next generation of industrial synthesis, offering a reliable 3,3',4'-BTDA supplier pathway that is both robust and commercially scalable.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
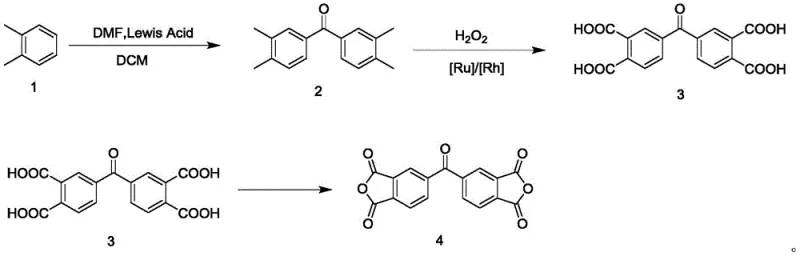
Historically, the industrial synthesis of 3,3',4'-benzophenonetetracarboxylic dianhydride has relied heavily on the use of bis(trichloromethyl) carbonate, commonly known as triphosgene, as a key carbonyl source. While effective in forming the benzophenone backbone, triphosgene is classified as a hazardous chemical that releases phosgene gas upon decomposition, posing severe safety risks to personnel and requiring expensive containment infrastructure. Following the acylation step, traditional methods typically employ potassium permanganate (KMnO4) to oxidize the methyl groups into carboxylic acids. This stoichiometric oxidation process is notoriously inefficient, generating vast quantities of manganese dioxide sludge that is difficult and costly to dispose of in an environmentally compliant manner. Consequently, the overall yield of these legacy processes often struggles to exceed 64%, leading to significant raw material waste and inflated production costs that erode profit margins.
The Novel Approach
In stark contrast, the methodology outlined in patent CN115160271A introduces a sophisticated three-step sequence that elegantly bypasses these historical bottlenecks. The process initiates with a safer Friedel-Crafts-type acylation using o-xylene and dichloromethane under Lewis acid catalysis, completely avoiding the use of toxic triphosgene. The subsequent oxidation step is the true technological leap, utilizing hydrogen peroxide as the terminal oxidant in the presence of a Ruthenium or Rhodium catalyst. This catalytic approach not only drives the reaction to near-completion with yields surpassing 87% but also ensures that the only byproduct is water, thereby eliminating heavy metal waste streams. This novel approach effectively realizes the purposes of large-scale production and cost reduction, making it an ideal candidate for cost reduction in polyimide precursor manufacturing where margin pressure is intense.
Mechanistic Insights into Lewis Acid Acylation and Ru-Catalyzed Oxidation
The core of this synthetic breakthrough lies in the precise control of reaction conditions during the initial acylation and the subsequent catalytic oxidation. In the first stage, o-xylene reacts with dichloromethane in the presence of a Lewis acid such as phosphorus oxychloride (POCl3) or thionyl chloride (SOCl2). This reaction proceeds through an electrophilic aromatic substitution mechanism where the Lewis acid activates the dichloromethane to form a reactive chloromethyl cation species. Maintaining the temperature between 0-5°C during the addition phase is critical to suppress poly-alkylation side reactions, ensuring high selectivity for the desired bis(3,4-dimethylphenyl)ketone intermediate. The careful modulation of the molar ratio between o-xylene and dichloromethane, typically kept between 1:0.6 and 1:0.8, further optimizes the conversion efficiency and minimizes the formation of oligomeric impurities that could complicate downstream purification.
Following the isolation of the ketone intermediate, the process employs a highly selective oxidation mechanism driven by hydrogen peroxide and a transition metal catalyst. Unlike the indiscriminate oxidation of permanganate, the Ruthenium or Rhodium catalyst facilitates the stepwise oxidation of the four methyl groups on the aromatic rings directly to carboxylic acids without degrading the sensitive benzophenone core. The reaction is conducted at moderate temperatures of 40-50°C, which provides sufficient energy for the catalytic cycle while preventing thermal decomposition of the hydrogen peroxide. Crucially, the catalyst can be filtered off while the solution is still hot, allowing for its recovery and reuse, which is a vital feature for reducing the cost of high-purity electronic chemical intermediates. This mechanistic elegance ensures that the final 3,3',4'-benzophenone tetracarboxylic acid is obtained with exceptional purity before the final thermal dehydration step.
How to Synthesize 3,3',4'-BTDA Efficiently
The practical implementation of this patented route requires strict adherence to the specified thermal profiles and separation techniques to maximize yield and purity. The process is designed to be robust enough for commercial scale-up of complex aromatic monomers, yet sensitive enough to demand precise operational control. Operators must ensure that the Lewis acid is added gradually to manage the exotherm during the acylation phase, and the subsequent oxidation must be monitored to prevent the accumulation of unreacted peroxide. For a detailed breakdown of the specific operational parameters, including stirring rates, exact addition times, and workup procedures, please refer to the standardized synthesis guide below.
- React o-xylene with dichloromethane using a Lewis acid catalyst (e.g., POCl3) at 0-5°C followed by room temperature to form bis(3,4-dimethylphenyl)ketone.
- Oxidize the ketone intermediate using hydrogen peroxide (H2O2) and a Ruthenium or Rhodium catalyst at 40-50°C to generate 3,3',4'-benzophenone tetracarboxylic acid.
- Dehydrate the tetracarboxylic acid in a high-temperature vacuum sintering furnace at 220-250°C to obtain the final 3,3',4'-benzophenonetetracarboxylic dianhydride.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this synthesis route offers compelling advantages that extend far beyond simple yield improvements. By transitioning to a process that utilizes commodity chemicals like o-xylene and dichloromethane alongside recyclable catalysts, manufacturers can decouple their production costs from the volatile pricing of specialized reagents like triphosgene. The elimination of hazardous waste streams also translates into significantly reduced disposal fees and lower regulatory compliance burdens, which are often hidden costs in traditional chemical manufacturing. For Supply Chain Heads, this means a more predictable cost structure and a reduced risk of production stoppages due to environmental inspections or raw material shortages.
- Cost Reduction in Manufacturing: The shift from stoichiometric oxidants to a catalytic system fundamentally changes the economics of production. By replacing expensive potassium permanganate with hydrogen peroxide and a recoverable Ruthenium catalyst, the process drastically lowers the variable cost per kilogram of product. Furthermore, the high yield of over 87% compared to the traditional 64% means that less raw material is required to produce the same amount of finished goods, leading to substantial cost savings in raw material procurement. The ability to recycle the precious metal catalyst further amortizes the cost of the catalytic system over multiple batches, enhancing the overall profitability of the manufacturing operation.
- Enhanced Supply Chain Reliability: Relying on hazardous reagents like triphosgene often necessitates complex logistics and specialized transportation, which can introduce delays and vulnerabilities into the supply chain. The new method utilizes widely available and easier-to-handle raw materials, thereby simplifying logistics and reducing the lead time for high-purity 3,3',4'-BTDA. Additionally, the improved safety profile of the process reduces the likelihood of unplanned shutdowns due to safety incidents, ensuring a more consistent and reliable supply for downstream customers in the polyimide and electronics industries. This stability is crucial for maintaining long-term contracts and meeting the just-in-time delivery requirements of major multinational corporations.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction conditions that are easily transferable from pilot plants to multi-ton reactors. The use of hydrogen peroxide as an oxidant generates water as the primary byproduct, which simplifies wastewater treatment and ensures compliance with stringent environmental regulations such as REACH and TSCA. This environmental friendliness not only future-proofs the manufacturing facility against tightening regulations but also enhances the brand value of the end-product as a 'green' chemical. The combination of high throughput and low environmental impact makes this route ideally suited for meeting the growing global demand for high-performance polymer precursors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering a transparent view of the technology's capabilities. Understanding these details is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: Why is the new H2O2/Ru oxidation method superior to traditional KMnO4 oxidation?
A: The traditional method using potassium permanganate (KMnO4) generates massive amounts of manganese dioxide sludge, creating severe environmental disposal issues and lowering yields to around 64%. The new method utilizes hydrogen peroxide as a green oxidant with a recyclable Ruthenium catalyst, achieving yields over 87% and significantly reducing hazardous waste.
Q: What are the safety advantages of avoiding bis(trichloromethyl) carbonate (triphosgene)?
A: Triphosgene is a highly toxic and hazardous reagent that poses significant risks during storage and handling, requiring specialized safety infrastructure. The novel process replaces this dangerous reagent with dichloromethane and Lewis acids, drastically improving the safety factor of the manufacturing plant and simplifying regulatory compliance.
Q: Can the Ruthenium catalyst be recovered in this process?
A: Yes, the process design explicitly includes a hot filtration step immediately after the oxidation reaction. This allows for the efficient recovery and potential reuse of the expensive Ruthenium or Rhodium catalyst, which is a critical factor in reducing the overall cost of goods sold (COGS) for large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3',4'-BTDA Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN115160271A can be fully realized in a practical industrial setting. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instruments to guarantee that every batch of 3,3',4'-BTDA meets the exacting standards required for high-performance polyimide applications. Our commitment to quality and consistency makes us a trusted ally for companies seeking to secure their supply of critical chemical intermediates.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can be tailored to your specific volume and purity requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic impact of switching to this greener, more efficient process. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and sustainability across your entire supply chain. Let us collaborate to bring this next-generation technology to life in your production facilities.
