Advanced Synthesis of Rigid Aromatic Diamine Monomers for High-Performance Polyaramid Manufacturing
The landscape of high-performance polymer manufacturing is undergoing a significant transformation driven by the demand for materials that balance thermal stability with processability. Patent CN103497110A introduces a breakthrough in this domain by detailing the synthesis of a novel rigid aromatic diamine monomer, specifically 9,9-bis(4-amino-3-isopropylphenyl)fluorene. This compound represents a critical advancement for the production of soluble polyarylamides, addressing the longstanding challenge of processing rigid polymer chains that traditionally require harsh solvents like concentrated sulfuric acid. By incorporating a bulky fluorenyl structure combined with dual isopropyl substitutions, this monomer enables the creation of polymers with exceptional solubility and film-forming capabilities, making it an ideal candidate for advanced applications such as gas separation membranes and high-temperature dielectric materials. For R&D directors and procurement specialists seeking a reliable polyaramid intermediate supplier, understanding the mechanistic elegance and commercial viability of this synthesis route is paramount for securing a competitive edge in the specialty chemicals market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional polyarylamides, while renowned for their mechanical strength and thermal resistance, suffer from severe processing limitations due to their rigid molecular backbones and strong intermolecular interactions. These structural characteristics lead to high melting temperatures and poor solubility in common organic solvents, often restricting their dissolution to aggressive media like concentrated sulfuric acid, which complicates downstream processing and increases environmental hazards. Furthermore, the synthesis of modified monomers to overcome these issues has historically involved multi-step pathways with low atom economy, requiring expensive catalysts or difficult purification protocols that drive up the overall cost of goods. The inability to easily cast films or spin fibers from solution without specialized equipment has hindered the broader adoption of these high-performance materials in emerging fields like flexible electronics and advanced filtration systems, creating a bottleneck for supply chain managers looking to scale production efficiently.
The Novel Approach
The innovative strategy outlined in the patent leverages a direct condensation reaction between 2-isopropylaniline and 9-fluorenone under acidic catalysis to produce the target diamine in a single, high-yielding step. This approach fundamentally alters the polymer architecture by introducing large, non-coplanar fluorene units that act as molecular spacers, effectively disrupting chain packing and enhancing free volume within the resulting polymer matrix. 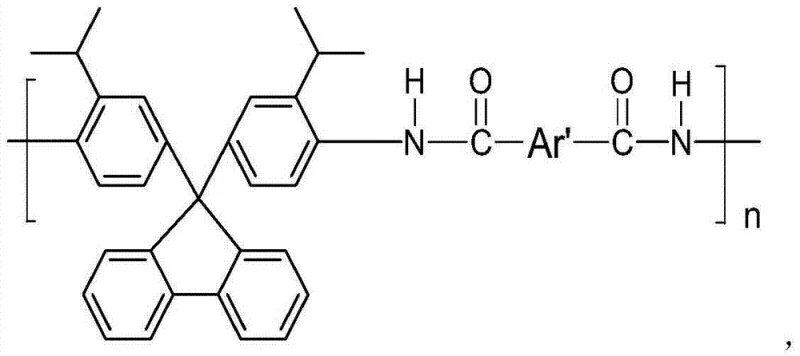 As illustrated in the polymer structure, the integration of these rigid yet bulky groups allows the final polyarylamide to retain thermal robustness while gaining significant solubility in polar aprotic solvents. This structural modification not only simplifies the molding and processing of the final material but also opens new avenues for application in seawater desalination and gas separation technologies, offering a distinct value proposition for manufacturers aiming to diversify their product portfolios with next-generation functional materials.
As illustrated in the polymer structure, the integration of these rigid yet bulky groups allows the final polyarylamide to retain thermal robustness while gaining significant solubility in polar aprotic solvents. This structural modification not only simplifies the molding and processing of the final material but also opens new avenues for application in seawater desalination and gas separation technologies, offering a distinct value proposition for manufacturers aiming to diversify their product portfolios with next-generation functional materials.
Mechanistic Insights into Acid-Catalyzed Condensation
The core of this synthesis lies in the efficient acid-catalyzed condensation mechanism, where trifluoromethanesulfonic acid (TfOH) serves as a potent promoter for the nucleophilic attack of the amine on the ketone carbonyl group. Under nitrogen protection, the reaction proceeds through the formation of a carbinolamine intermediate, which subsequently dehydrates to form the stable carbon-nitrogen bonds linking the fluorene core to the aniline derivatives. The use of TfOH is particularly strategic due to its superacidic nature, which activates the carbonyl carbon of 9-fluorenone more effectively than conventional mineral acids, thereby driving the equilibrium towards product formation even at moderate reflux temperatures. This catalytic efficiency minimizes side reactions and ensures a clean conversion profile, which is critical for maintaining the high purity required for subsequent polymerization steps where stoichiometric imbalance could severely impact molecular weight and material properties.
Impurity control is meticulously managed through a robust workup procedure that exploits the differential solubility of the product and byproducts. Following the reaction, the mixture is neutralized with aqueous alkali solutions such as sodium hydroxide or potassium carbonate, which quenches the acid catalyst and facilitates the precipitation of the crude diamine. The subsequent recrystallization from solvents like toluene or ethanol removes unreacted starting materials and oligomeric impurities, yielding a white solid powder with a sharp melting point range of 227-228°C. This rigorous purification protocol ensures that the monomer meets stringent quality specifications, eliminating potential defects in the final polyaramid that could compromise membrane integrity or mechanical performance, thus providing supply chain heads with confidence in the consistency and reliability of the raw material supply.
How to Synthesize 9,9-Bis(4-amino-3-isopropylphenyl)fluorene Efficiently
The synthesis protocol described offers a streamlined pathway for producing this high-value monomer, utilizing readily available feedstocks and standard reactor configurations that are easily adaptable to commercial scale. The process begins with the precise metering of 2-isopropylaniline and 9-fluorenone into a reactor under an inert atmosphere, followed by the controlled addition of the acid catalyst to initiate the exothermic condensation. Maintaining the reaction at reflux for a defined period ensures complete conversion, after which the product is isolated through a simple phase separation and crystallization sequence that avoids the need for complex chromatographic purification. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps provided below.
- Under nitrogen protection, mix 2-isopropylaniline and 9-fluorenone (molar ratio 3: 1 to 1.4) and add trifluoromethanesulfonic acid catalyst.
- Heat the mixture to reflux conditions and maintain stirring for 5 to 10 hours to ensure complete reaction.
- Neutralize the product with alkali, precipitate in ethanol/water, filter, and recrystallize using toluene or ethanol to obtain the pure monomer.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis route offers substantial advantages by leveraging low-cost commodity chemicals and eliminating the need for precious metal catalysts that often plague fine chemical manufacturing. The reliance on 2-isopropylaniline and 9-fluorenone as starting materials ensures a stable and abundant supply base, mitigating risks associated with raw material scarcity or price volatility that can disrupt production schedules. Furthermore, the simplicity of the isolation process, which involves basic neutralization and filtration rather than energy-intensive distillation or extraction, translates directly into reduced utility consumption and lower operational expenditures. For procurement managers focused on cost reduction in polymer additive manufacturing, this process represents a highly optimized solution that maximizes yield while minimizing waste generation and processing time.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of a one-pot synthesis strategy significantly lower the direct material costs associated with monomer production. By avoiding complex multi-step sequences and utilizing efficient acid catalysis, the process reduces the consumption of solvents and reagents, leading to a leaner manufacturing footprint. Additionally, the high yield reported in the patent examples indicates minimal loss of valuable intermediates, ensuring that the overall cost per kilogram of the final diamine remains competitive against traditional alternatives that suffer from lower efficiency.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate standard industrial heating and stirring equipment, facilitates easy scale-up from laboratory to pilot and commercial production scales without requiring specialized hardware. The stability of the final product at room temperature allows for flexible inventory management and extended shelf life, reducing the pressure on logistics teams to maintain cold chains or expedited shipping. This operational flexibility ensures a continuous supply of high-purity monomers, enabling downstream polymer manufacturers to plan their production runs with greater certainty and reduced risk of stockouts.
- Scalability and Environmental Compliance: The aqueous workup and solvent recycling potential inherent in this process align well with modern environmental regulations and sustainability goals. The absence of heavy metal residues simplifies wastewater treatment requirements, lowering the cost of compliance and reducing the environmental impact of the manufacturing facility. Moreover, the ability to produce soluble polyarylamides that do not require hazardous solvents like concentrated sulfuric acid for processing adds a layer of green chemistry value to the final product, appealing to end-users who prioritize eco-friendly materials in their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this rigid aromatic diamine monomer. These insights are derived directly from the patent data to provide clarity on the feasibility and benefits of adopting this technology for your specific manufacturing needs. Understanding these details is crucial for making informed decisions about integrating this intermediate into your existing product lines or developing new high-performance materials.
Q: What are the key advantages of using fluorene-based diamines in polyaramid synthesis?
A: The introduction of bulky fluorenyl structures disrupts the tight packing of polymer chains, significantly improving solubility in organic solvents while maintaining high thermal stability and excellent film-forming properties essential for separation membranes.
Q: How does the new synthesis method improve cost efficiency compared to traditional routes?
A: The process utilizes cheap, commercially available starting materials like 2-isopropylaniline and avoids expensive transition metal catalysts, relying instead on efficient acid catalysis and simple precipitation workups that drastically reduce purification costs.
Q: What represents the typical yield and purity profile for this monomer?
A: The patented method achieves high yields ranging from 81% to 82% with a melting point of 227-228°C, indicating a highly crystalline and pure product suitable for demanding polymerization applications without extensive further purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9,9-Bis(4-amino-3-isopropylphenyl)fluorene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality monomers play in the development of advanced polymeric materials, and we are committed to delivering this specific fluorene-based diamine with uncompromising quality standards. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the R&D phase or full-scale manufacturing. We operate stringent purity specifications and utilize rigorous QC labs to verify every batch, guaranteeing that the 9,9-bis(4-amino-3-isopropylphenyl)fluorene you receive is perfectly suited for the synthesis of high-performance separation membranes and electronic materials.
We invite you to collaborate with us to explore how this innovative monomer can enhance your product portfolio and drive efficiency in your operations. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our supply capabilities can support your long-term strategic goals in the competitive landscape of specialty polymers and advanced materials.
