Advanced Industrial Synthesis Of Terpyridine Ligands For Commercial Scale Production
The chemical industry continuously seeks more efficient pathways for synthesizing complex heterocyclic ligands, particularly terpyridine derivatives which serve as critical building blocks in pharmaceutical and agrochemical applications. Patent CN1390835A introduces a groundbreaking methodology for the preparation of terpyridine compounds of formula (I), addressing long-standing inefficiencies in yield and raw material costs. This innovation pivots away from expensive picolinate esters, opting instead for the more economically viable 2-cyanopyridine as the primary starting material. The structural versatility of these compounds allows for various substituents, making them indispensable in the coordination chemistry sector.
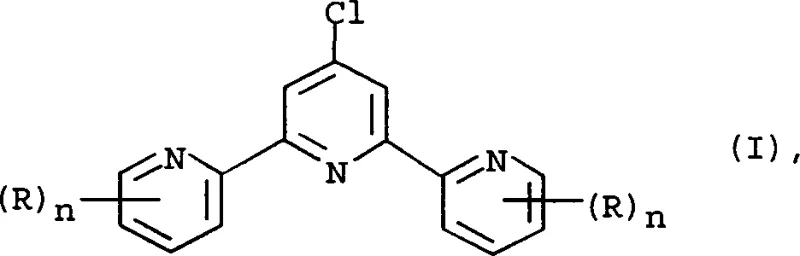
The core breakthrough lies in a multi-step sequence that optimizes every stage of the molecular assembly, from the initial hydrolysis to the final chlorination. By integrating an improved acid hydrolysis step followed by a controlled Claisen condensation, the process ensures high purity intermediates. Furthermore, the strategic use of azeotropic distillation during the cyclization phase effectively drives the equilibrium towards the desired pyridone product, minimizing side reactions. This comprehensive approach not only enhances the theoretical yield but also simplifies the downstream purification processes required for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4'-chloro-2,2':6',2''-terpyridine has been plagued by economic and safety hurdles that hinder large-scale adoption. Traditional routes, such as those described by Constable and Ward, rely heavily on ethyl picolinate, a raw material that commands a significantly higher market price compared to nitrile precursors. Moreover, the conventional condensation step typically necessitates the use of sodium hydride, a reagent notorious for its pyrophoric nature and extreme sensitivity to moisture, which complicates reactor operations and increases safety protocols. The subsequent chlorination often involves phosphorus pentachloride, generating substantial amounts of corrosive waste and requiring specialized disposal methods. When calculated from the basic 2-cyanopyridine starting point, the cumulative yield of these legacy methods drops precipitously to approximately 16 percent, rendering them commercially unviable for bulk manufacturing.
The Novel Approach
The methodology disclosed in CN1390835A fundamentally restructures the synthetic landscape by introducing a robust hydrolysis protocol that converts inexpensive 2-cyanopyridine derivatives into esters with exceptional efficiency. As illustrated in the starting material structure, the nitrile group is selectively transformed under controlled acidic conditions without degrading the pyridine ring.
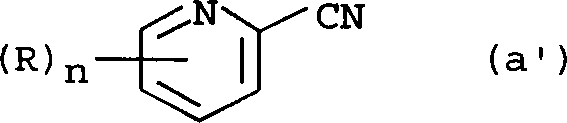
This novel route replaces hazardous sodium hydride with safer alkali metal alkoxides, such as sodium methoxide, facilitating a smoother Claisen condensation with acetone. The process further distinguishes itself by employing phosphorus oxychloride mixtures with organic solvents like toluene or xylene, which moderates the reaction exotherm and improves the handling of the chlorination step. By optimizing each transformation, the overall yield from the cheap nitrile precursor is drastically improved, transforming a previously marginal process into a highly attractive industrial option for producing high-purity terpyridine ligands.
Mechanistic Insights into Claisen Condensation and Cyclization
The heart of this synthesis lies in the precise execution of the Claisen condensation followed by the Hantzsch-type cyclization. In the condensation phase, the pyridine-2-carboxylic acid ester reacts with acetone in an aprotic solvent like tetrahydrofuran (THF) in the presence of a base. The mechanism involves the deprotonation of the ester alpha-position or the acetone to form an enolate, which then attacks the carbonyl carbon of the second molecule. This step is critical because it forms the 1,5-bis(2-pyridyl)pentane-1,3,5-trione skeleton, a linear precursor that sets the stage for ring closure. The choice of base is paramount; alkali metal alkoxides provide the necessary nucleophilicity without the violent reactivity associated with hydrides, ensuring a cleaner reaction profile and easier workup procedures.
Following the formation of the trione intermediate, the cyclization step converts this linear chain into the central pyridine ring. This transformation is driven by the reaction with ammonia or ammonium salts, which introduces the nitrogen atom required for the heterocycle.
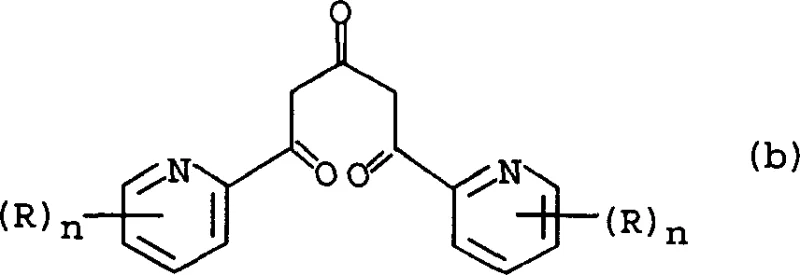
A key mechanistic feature here is the continuous removal of water generated during the condensation. The process utilizes C1-C4 alkanols, such as ethanol, as entrainers to form an azeotrope with water. By physically removing the water byproduct via a Dean-Stark trap or similar distillation setup, the equilibrium is shifted decisively towards the formation of the 2,6-bis(2-pyridyl)-4(1H)pyridone derivative. This thermodynamic control prevents the hydrolysis of the intermediate and ensures high conversion rates. The resulting pyridone is then ready for the final activation step, where the hydroxyl group is replaced by a chlorine atom to yield the final electrophilic terpyridine product.
How to Synthesize 4'-Chloro-2,2':6',2''-terpyridine Efficiently
The synthesis of this high-value ligand requires strict adherence to the optimized reaction parameters defined in the patent to ensure maximum yield and purity. The process begins with the careful hydrolysis of the nitrile, followed by the base-mediated condensation and the crucial water-removal cyclization. Each step builds upon the previous one to construct the terpyridine backbone efficiently. For detailed operational parameters, temperature controls, and specific molar ratios required to replicate this industrial-grade synthesis, please refer to the standardized guide below.
- Perform acid hydrolysis of 2-cyanopyridine derivatives with anhydrous inorganic acid in the presence of water and C1-C4 alcohol to form pyridine-2-carboxylic acid esters.
- Condense the resulting ester with acetone in an aprotic solvent using alkali metal alkoxides to generate 1,5-bis(2-pyridyl)pentane-1,3,5-trione derivatives.
- React the trione derivative with ammonia or ammonium salts while removing reaction water via azeotropic distillation to form pyridone intermediates.
- Chlorinate the pyridone derivative using phosphorus oxychloride (POCl3) optionally with organic solvents like toluene to yield the final terpyridine compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to this novel synthesis route represents a strategic opportunity to optimize spending and secure supply lines. The primary driver of value is the substitution of high-cost ethyl picolinate with low-cost 2-cyanopyridine. This raw material swap alone creates a substantial margin for cost reduction in fine chemical manufacturing, allowing for more competitive pricing in the final API or ligand market. Furthermore, the elimination of sodium hydride reduces the need for specialized hazardous material storage and handling infrastructure, lowering operational overheads related to safety compliance and insurance.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from both raw material arbitrage and process efficiency. By utilizing 2-cyanopyridine, which is abundantly available and priced significantly lower than ester alternatives, the direct material cost per kilogram of product is drastically lowered. Additionally, the improved overall yield means less waste of reagents and solvents, further compressing the cost of goods sold. The avoidance of expensive and difficult-to-handle reagents like phosphorus pentachloride also reduces waste disposal costs, contributing to a leaner manufacturing budget without compromising on output quality.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like 2-cyanopyridine and acetone insulates the production schedule from the volatility often seen with specialized fine chemical intermediates. These starting materials are produced on a massive global scale, ensuring consistent availability and reducing the risk of supply disruptions. Moreover, the simplified safety profile of the process, which avoids pyrophoric reagents, minimizes the likelihood of production stoppages due to safety incidents or regulatory inspections, thereby guaranteeing a more stable and predictable delivery timeline for downstream customers.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard unit operations such as reflux, distillation, and filtration that are common in multipurpose chemical plants. The use of recyclable solvents like toluene and ethanol aligns with modern green chemistry principles, facilitating easier permitting and environmental compliance. The reduction in hazardous waste generation, particularly through the moderated chlorination step, simplifies effluent treatment requirements, making the technology sustainable for long-term, high-volume commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this terpyridine synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield improvements, safety modifications, and scalability factors for potential licensees or manufacturing partners.
Q: What is the primary advantage of using 2-cyanopyridine over ethyl picolinate for terpyridine synthesis?
A: 2-Cyanopyridine is significantly cheaper than ethyl picolinate. The novel process described in CN1390835A achieves a much higher overall yield (over 40% compared to roughly 16% in prior art) when starting from 2-cyanopyridine, making it economically superior for large-scale production.
Q: How does the new process improve safety compared to conventional methods?
A: Conventional methods often utilize sodium hydride, which is highly sensitive to air and moisture, posing significant handling risks. The new process employs safer alkali metal alkoxides like sodium methoxide and avoids the use of excessive corrosive phosphorus pentachloride, utilizing phosphorus oxychloride mixtures instead.
Q: Can this synthesis route be scaled for industrial manufacturing?
A: Yes, the process is specifically designed for industrial scalability. It utilizes common solvents like ethanol and toluene, avoids extremely hazardous reagents where possible, and includes robust purification steps such as recrystallization and aqueous workups that are standard in chemical engineering.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4'-Chloro-2,2':6',2''-terpyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality ligands play in the development of next-generation pharmaceuticals and advanced materials. Our technical team has thoroughly analyzed the pathway described in CN1390835A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with rigorous QC labs and stringent purity specifications to ensure that every batch of terpyridine compound meets the exacting standards required for catalytic applications and drug synthesis. Our facility is optimized to handle the specific solvent systems and reaction conditions outlined in this patent, ensuring a seamless transition from lab scale to industrial volume.
We invite procurement leaders to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging this optimized synthesis route, we can offer a supply solution that balances performance with economic efficiency. Please contact our technical procurement team to request specific COA data and route feasibility assessments for your upcoming projects, ensuring you secure a reliable supply of this essential chemical building block.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
