Advanced Synthesis of Novel Benzazepine Isatin Intermediates for Pharmaceutical Manufacturing
The pharmaceutical and organic electronics industries are constantly seeking novel heterocyclic scaffolds that offer enhanced biological activity or superior charge mobility. Patent CN113461679B introduces a significant breakthrough in this domain by disclosing a robust synthesis method for a novel benzazepine isatin intermediate, specifically 1H-pyrrolo[2,3-c]quinoline-1,2(3H)-dione. This compound serves as a critical bioisostere of isatin, possessing a larger conjugated system that is highly advantageous for developing anti-tumor agents and high-performance organic field-effect transistors. The patented methodology addresses long-standing challenges in heterocyclic chemistry by providing a route that is not only chemically efficient but also operationally simple, utilizing mild reaction conditions that are conducive to industrial scalability. By leveraging this intellectual property, manufacturers can access a versatile building block capable of forming strong intermolecular interactions with various enzymes and receptors, thereby unlocking new potential in drug discovery pipelines.
![Chemical structure of the target compound 1H-pyrrolo[2,3-c]quinoline-1,2(3H)-dione (VI)](/insights/img/pyrrolo-quinoline-dione-synthesis-pharma-supplier-20260305120009-01.png)
For R&D teams evaluating this technology, the structural integrity and purity of the final product are paramount. The synthesis culminates in the formation of the target dione structure shown above, which features a fused quinoline and pyrrole ring system. This specific architecture allows for extensive structural modification at the 3-carbonyl active site, similar to traditional isatin derivatives but with improved electronic properties due to the nitrogen atom within the quinoline ring. The ability to produce this scaffold reliably opens doors for creating donor-acceptor (D-A) type organic photoelectric materials with wide absorption spectra. Furthermore, the patent emphasizes that the resulting intermediate can be easily converted into pharmaceutically acceptable salts, solvates, or isotopic derivatives, providing flexibility for medicinal chemists aiming to optimize metabolic stability or solubility profiles in lead optimization campaigns.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex azaisatin derivatives has been plagued by harsh reaction conditions and difficult purification processes. Traditional reduction methods for converting nitro groups to amines, such as using iron powder with ammonium chloride or stannous chloride with concentrated hydrochloric acid, generate substantial amounts of inorganic waste sludge. This not only complicates the work-up procedure, often leading to emulsification during extraction, but also poses significant environmental disposal challenges. Additionally, these conventional reducing agents can be non-selective, potentially affecting other sensitive functional groups on the quinoline ring, leading to a complex impurity profile that requires extensive chromatographic purification. The use of excessive amounts of strong acids in older cyclization protocols further exacerbates equipment corrosion issues and safety hazards in a plant setting, making the overall process economically unattractive for large-scale production.
The Novel Approach
The patented process outlined in CN113461679B offers a transformative alternative by employing a streamlined five-step sequence that prioritizes atom economy and operational safety. As illustrated in the comprehensive reaction scheme below, the route begins with a nucleophilic substitution followed by a clean decarboxylation, setting the stage for a highly efficient catalytic hydrogenation step. This modern approach replaces the messy metal-acid reductions with a catalytic Pd/C hydrogenation system, which operates under mild temperatures and generates water as the primary byproduct. The subsequent cyclization and oxidation steps are finely tuned to maximize yield while minimizing side reactions, ensuring a high-purity output suitable for sensitive pharmaceutical applications without the need for exhaustive downstream processing.
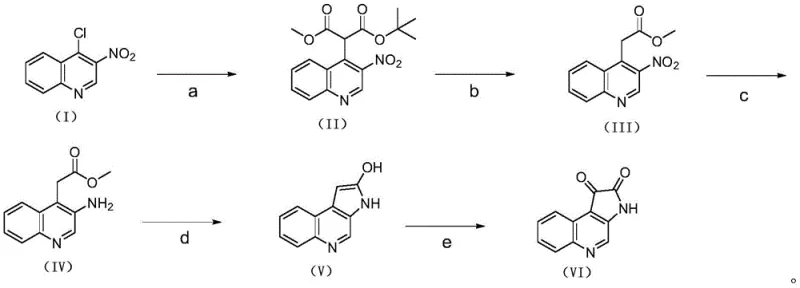
This novel pathway demonstrates exceptional control over reaction parameters, particularly in the reduction and cyclization stages. By optimizing the molar ratios of reagents and strictly controlling temperature profiles, the inventors have achieved a total yield that significantly outperforms legacy methods. The use of tert-butyl malonate methyl ester as a starting material allows for a controlled introduction of the two-carbon unit required for ring closure, avoiding the regioselectivity issues often encountered in direct alkylation strategies. Furthermore, the final oxidation step using selenium dioxide is conducted under conditions that prevent over-oxidation, preserving the integrity of the sensitive heterocyclic core. This level of precision makes the process a reliable [pharmaceutical intermediate] supplier solution for companies looking to secure a stable supply chain for advanced drug candidates.
Mechanistic Insights into Pd/C-Catalyzed Nitro Reduction and Cyclization
The cornerstone of this synthesis lies in the mechanistic efficiency of Step C, where the nitro group is reduced to an amine using a palladium on carbon (Pd/C) catalyst under a hydrogen atmosphere. Unlike stoichiometric reductions that rely on electron transfer from bulk metals, this catalytic cycle involves the adsorption of hydrogen gas onto the palladium surface, followed by the sequential transfer of hydrogen atoms to the nitro group. This mechanism proceeds through nitroso and hydroxylamine intermediates before finally yielding the amine, all while maintaining the aromaticity of the quinoline ring. The patent data indicates that maintaining the reaction temperature between 60°C and 80°C is critical; lower temperatures result in incomplete conversion, while higher temperatures may promote hydrogenolysis of other bonds or catalyst deactivation. This precise thermal control ensures a yield of up to 96%, drastically reducing the burden on purification systems.
Following the reduction, the cyclization mechanism in Step D involves an intramolecular nucleophilic attack of the newly formed amine on the ester carbonyl, facilitated by acid catalysis. The protonation of the carbonyl oxygen by concentrated hydrochloric acid increases the electrophilicity of the carbonyl carbon, making it susceptible to attack by the lone pair of the adjacent amino group. This forms a tetrahedral intermediate which subsequently collapses to eliminate methanol and close the five-membered pyrrole ring. The patent highlights that the ratio of acid to substrate is a governing factor; a ratio of 1:10 to 1:15 provides the optimal acidity to drive the equilibrium towards the cyclic product without causing degradation of the sensitive quinoline nucleus. This mechanistic understanding allows for [cost reduction in API manufacturing] by minimizing reagent waste and maximizing the throughput of the reactor.
How to Synthesize 1H-Pyrrolo[2,3-c]Quinoline-1,2(3H)-dione Efficiently
Implementing this synthesis requires careful attention to the specific conditions outlined in the patent to ensure reproducibility and high quality. The process is designed to be scalable, moving from gram-scale laboratory experiments to multi-kilogram production with minimal adjustment. Operators must ensure that anhydrous conditions are maintained during the initial substitution step to prevent hydrolysis of the malonate ester, and that the hydrogenation step is monitored closely via TLC to prevent over-reduction. The following guide summarizes the critical operational parameters derived from the experimental examples, serving as a foundational reference for process engineers.
- Perform nucleophilic substitution of 4-chloro-3-nitroquinoline with tert-butyl malonate methyl ester using NaH in DMF to form the malonate intermediate.
- Execute acidic decarboxylation using trifluoroacetic acid in dichloromethane to remove the tert-butyl protecting group.
- Conduct catalytic hydrogenation using Pd/C in ethanol at 60-80°C to reduce the nitro group to an amine with high selectivity.
- Induce cyclization by heating the amino ester with concentrated hydrochloric acid in THF to form the pyrrolo-quinolinol core.
- Finalize the synthesis via oxidation with selenium dioxide in DMSO at 100°C to yield the target dione compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers tangible strategic benefits beyond mere chemical elegance. The shift away from heavy metal reductants like iron and tin eliminates the need for complex waste treatment facilities dedicated to handling toxic metal sludge, thereby reducing environmental compliance costs and regulatory risks. The simplified work-up procedures, characterized by straightforward filtrations and extractions, translate directly into shorter batch cycle times and higher equipment utilization rates. This efficiency is crucial for maintaining a continuous supply of high-value intermediates in a competitive market, ensuring that downstream drug development projects are not delayed by raw material shortages.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous stoichiometric reducing agents in favor of a recyclable Pd/C catalyst significantly lowers the variable cost per kilogram of the product. Although palladium is a precious metal, its heterogeneous nature allows for recovery and reuse, amortizing the initial investment over multiple batches. Furthermore, the high selectivity of the reaction reduces the formation of difficult-to-remove impurities, which decreases the consumption of silica gel and solvents during the purification phase. This holistic reduction in material usage and waste disposal fees contributes to a leaner, more cost-effective manufacturing model that enhances overall profit margins.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials, such as 4-chloro-3-nitroquinoline and tert-butyl malonate, mitigates the risk of supply disruptions often associated with exotic or custom-synthesized reagents. The robustness of the reaction conditions, particularly the tolerance of the Pd/C catalyst to minor variations in pressure and temperature, ensures consistent batch-to-batch quality. This reliability is essential for long-term supply agreements with pharmaceutical clients who require stringent adherence to quality specifications. By securing a process that is less prone to failure, supply chain heads can guarantee [reducing lead time for high-purity pharmaceutical intermediates] and maintain inventory levels that support just-in-time manufacturing strategies.
- Scalability and Environmental Compliance: The process is inherently designed for [commercial scale-up of complex pharmaceutical intermediates], utilizing standard unit operations like stirred tank reactors and filtration units that are ubiquitous in fine chemical plants. The absence of pyrophoric reagents or extreme cryogenic conditions simplifies the engineering controls required for safe operation, facilitating a smoother technology transfer from R&D to production. Additionally, the reduced generation of hazardous waste aligns with global sustainability goals and increasingly strict environmental regulations, positioning the manufacturer as a responsible partner in the green chemistry initiative. This compliance advantage future-proofs the supply chain against tightening regulatory frameworks regarding industrial emissions and effluent discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and optimization studies presented in the patent documentation, providing clarity on process capabilities and limitations. Understanding these details is vital for technical teams assessing the feasibility of integrating this route into their existing production portfolios.
Q: Why is Pd/C preferred over iron powder for the nitro reduction step?
A: Pd/C catalytic hydrogenation avoids the generation of large amounts of metal oxide sludge associated with iron powder reduction, significantly simplifying post-reaction filtration and waste treatment while maintaining high yields up to 96%.
Q: What are the optimal conditions for the cyclization step to minimize impurities?
A: The patent specifies that maintaining a molar ratio of substrate to concentrated hydrochloric acid between 1:10 and 1:15, coupled with a reaction temperature of 40-60°C, maximizes yield and minimizes side reactions.
Q: Can this synthesis route be scaled for industrial production?
A: Yes, the route utilizes common solvents like ethanol and THF and avoids extremely hazardous reagents, making it economically applicable and suitable for large-scale commercial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1H-Pyrrolo[2,3-c]Quinoline-1,2(3H)-dione Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in accelerating drug discovery and development. Our team of expert chemists has extensively analyzed the patented route for 1H-pyrrolo[2,3-c]quinoline-1,2(3H)-dione and possesses the technical capability to execute this synthesis with precision and efficiency. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the pre-clinical or commercial phase. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of material we deliver meets the highest industry standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how our optimized process can reduce your overall cost of goods. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate why we are the preferred partner for sourcing complex heterocyclic building blocks. Together, we can drive innovation and bring life-saving therapies to market faster and more efficiently.
