Scalable Electrochemical Oxidation for High-Purity Trifluoromethylated Pharmaceutical Intermediates
Scalable Electrochemical Oxidation for High-Purity Trifluoromethylated Pharmaceutical Intermediates
The landscape of fine chemical manufacturing is undergoing a paradigm shift towards greener, more sustainable methodologies, particularly in the synthesis of fluorinated building blocks essential for modern drug discovery. Patent CN114214646A introduces a groundbreaking electrochemical oxidation strategy for the efficient synthesis of 2-trifluoromethyl-alpha-carbonyl dithioketene compounds. This technology represents a significant departure from conventional stoichiometric oxidation methods, leveraging electric current as a traceless oxidant to drive the trifluoromethylation of alpha-carbonyl dithioketenes. By utilizing readily available sodium trifluoromethanesulfinate as the CF3 source and operating under mild, open-system conditions, this process addresses critical pain points regarding environmental impact, operational safety, and cost efficiency. For R&D directors and procurement specialists seeking reliable sources of complex fluorinated intermediates, this patent outlines a pathway that eliminates the need for expensive transition metal catalysts and harsh chemical oxidants, thereby streamlining the supply chain for high-value pharmaceutical and agrochemical precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of trifluoromethyl groups into organic scaffolds has been a challenging endeavor, often necessitating the use of specialized, high-cost reagents such as (trifluoromethyl)trimethylsilane (TMSCF3) or hypervalent iodine reagents like Togni's Reagent. These traditional protocols frequently require the presence of transition metal catalysts, which pose significant downstream processing challenges due to the strict regulatory limits on residual heavy metals in active pharmaceutical ingredients (APIs). Furthermore, the reliance on stoichiometric chemical oxidants generates substantial amounts of hazardous waste, complicating disposal and increasing the overall environmental footprint of the manufacturing process. The harsh reaction conditions often associated with these methods, including the need for inert atmospheres and elevated temperatures, further exacerbate operational costs and safety risks, making the commercial scale-up of complex fluorinated intermediates a logistical burden for many fine chemical manufacturers.
The Novel Approach
In stark contrast to these legacy techniques, the electrochemical method described in CN114214646A offers a streamlined, atom-economical alternative that fundamentally redefines the synthesis of 2-trifluoromethyl-alpha-carbonyl dithioketenes. By employing a diaphragm-free electrolytic cell with a carbon felt anode and an iron cathode, the process utilizes electrons as the primary reagent, effectively replacing toxic chemical oxidants. This approach not only simplifies the reaction setup by allowing operations under ambient air and room temperature conditions but also ensures that the only byproduct is typically hydrogen gas or simple salts, drastically reducing waste generation. The use of sodium trifluoromethanesulfinate, a commercially available and stable solid, as the trifluoromethyl source further enhances the economic viability of the process, providing a robust platform for the production of diverse fluorinated building blocks without the baggage of heavy metal contamination.

Mechanistic Insights into Electrochemical Oxidative Trifluoromethylation
The core of this innovation lies in the anodic oxidation mechanism, where the application of a constant current facilitates the generation of reactive radical species from the sodium trifluoromethanesulfinate precursor. At the carbon felt anode, the sulfinate anion undergoes single-electron oxidation to generate a trifluoromethyl radical, which subsequently adds to the electron-rich double bond of the alpha-carbonyl dithioketene substrate. This radical addition is followed by a second oxidation event and deprotonation to restore the conjugated system, yielding the desired 2-trifluoromethyl product. The choice of electrode materials is critical; the high surface area of the carbon felt anode maximizes the efficiency of the electron transfer process, while the iron cathode provides a stable counter-electrode environment. This mechanistic pathway avoids the formation of metal-complex intermediates, ensuring that the final product is free from transition metal residues, a feature that is paramount for meeting the stringent purity specifications required by global regulatory bodies for pharmaceutical intermediates.
From an impurity control perspective, the mild nature of the electrochemical conditions plays a pivotal role in maintaining high product fidelity. Unlike thermal radical initiators that can lead to non-selective decomposition or polymerization side reactions, the controlled potential applied in this system selectively targets the oxidation of the sulfinate species. The tolerance of the reaction to water and oxygen further mitigates the risk of moisture-sensitive side products that often plague traditional organometallic trifluoromethylation reactions. As demonstrated in the patent examples, this selectivity allows for the successful functionalization of substrates containing sensitive heterocycles such as furan and thiophene, which might otherwise degrade under harsher oxidative conditions. This broad functional group compatibility ensures that the synthetic route is versatile enough to accommodate a wide array of structural motifs found in modern drug candidates.
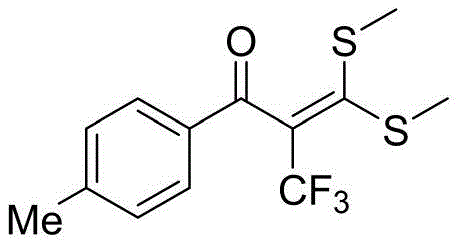
How to Synthesize 2-Trifluoromethyl-alpha-carbonyl Dithioketene Efficiently
The practical implementation of this electrochemical synthesis is designed for ease of operation, requiring standard laboratory equipment adapted for electrolysis. The process begins with the assembly of a simple undivided cell, into which the substrate, electrolyte, and solvent are charged. The reaction is driven by a constant current power supply, eliminating the need for complex potential control systems. Following the electrolysis, the workup procedure is straightforward, involving standard aqueous extraction and drying techniques familiar to any process chemist. The detailed standardized synthesis steps, including precise molar ratios and purification parameters, are outlined below to facilitate immediate technology transfer and process validation.
- Assemble a diaphragm-free electrolytic cell equipped with a carbon felt anode and an iron plate cathode.
- Add alpha-carbonyl dithioketene, sodium trifluoromethanesulfinate, tetra-n-butylammonium perchlorate electrolyte, and acetonitrile/water solvent to the reactor.
- Stir the mixture under open-air conditions at room temperature while applying a constant current of 10 mA for approximately 4 hours.
- Upon completion, extract the crude product with dichloromethane, wash with brine, dry, and purify via flash column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrochemical methodology translates into tangible strategic advantages regarding cost stability and supply continuity. By shifting away from proprietary, high-cost fluorinating reagents to commodity chemicals like sodium trifluoromethanesulfinate, manufacturers can decouple their production costs from the volatile pricing of specialized organofluorine reagents. The elimination of transition metal catalysts removes the necessity for expensive scavenging resins and additional purification steps, directly reducing the bill of materials and processing time. Furthermore, the ability to run the reaction in an open system at room temperature significantly lowers energy consumption compared to processes requiring cryogenic cooling or high-pressure reactors, contributing to a leaner and more sustainable manufacturing overhead.
- Cost Reduction in Manufacturing: The replacement of expensive reagents like Togni's Reagent or TMSCF3 with inexpensive sodium trifluoromethanesulfinate results in a drastic reduction in raw material expenditure. Additionally, the absence of transition metal catalysts eliminates the costs associated with metal removal technologies and the disposal of heavy metal waste, leading to substantial overall cost savings in the production of fluorinated intermediates.
- Enhanced Supply Chain Reliability: The starting materials for this process, including alpha-carbonyl dithioketenes and sodium trifluoromethanesulfinate, are commercially available in bulk quantities from multiple global suppliers. This diversification of the supply base reduces the risk of single-source bottlenecks and ensures a steady flow of materials, which is critical for maintaining uninterrupted production schedules for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The simplicity of the reactor design (diaphragm-free cell) and the mild reaction conditions make this process highly amenable to scale-up from gram to multi-ton scales. The green nature of the process, characterized by the use of electricity as a clean oxidant and the generation of minimal waste, aligns perfectly with increasingly stringent environmental regulations, facilitating easier permitting and long-term operational sustainability for chemical manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of 2-trifluoromethyl-alpha-carbonyl dithioketene compounds. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the technology's capabilities and limitations for potential partners and licensees.
Q: How does this electrochemical method compare to traditional trifluoromethylation reagents?
A: Traditional methods often rely on expensive and toxic reagents like TMSCF3 or Togni's Reagent, along with transition metal catalysts. This electrochemical approach utilizes inexpensive sodium trifluoromethanesulfinate and electricity as the oxidant, eliminating heavy metal contamination and significantly reducing raw material costs.
Q: Is the reaction sensitive to moisture or oxygen during scale-up?
A: No, one of the key advantages of this protocol is its robustness. The reaction proceeds smoothly under open-air conditions at room temperature and tolerates the presence of water in the solvent system (acetonitrile/water mixture), making it highly suitable for industrial scale-up without stringent inert atmosphere requirements.
Q: What is the substrate scope for this electrochemical oxidation?
A: The method demonstrates excellent functional group compatibility. It successfully converts various alpha-carbonyl dithioketenes, including those substituted with aromatic groups (phenyl, naphthyl), heteroaromatic rings (furan, thiophene), and alkyl chains, into the corresponding 2-trifluoromethyl derivatives with moderate to good yields.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl-alpha-carbonyl Dithioketene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in the production of advanced fluorinated intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN114214646A can be seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl-alpha-carbonyl dithioketene delivered meets the exacting standards required for API synthesis and fine chemical applications.
We invite forward-thinking R&D and procurement leaders to collaborate with us to leverage this cutting-edge technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your supply chain is optimized for both performance and profitability in the competitive global market.
