Scalable Synthesis of Key Biotin Intermediate (3aS, 6aR)-1,3-Dibenzyltetrahydro-4H-Thieno[3,4-D]Imidazol-2,4-(1H)-Dione
The pharmaceutical and nutraceutical industries continuously demand more efficient pathways for synthesizing essential vitamins, particularly Vitamin H, commonly known as Biotin. A pivotal breakthrough in this domain is documented in Chinese patent CN103880859A, which details a robust preparation method for (3aS, 6aR)-1,3-dibenzyltetrahydro-4H-thieno[3,4-d]imidazol-2,4-(1H)-dione. This compound serves as a critical chiral intermediate in the total synthesis of D-Biotin, a cofactor essential for carboxylase enzymes in human metabolism. The disclosed methodology represents a significant departure from legacy protocols by utilizing L-cystine derivatives as a chiral starting point, thereby circumventing the need for complex resolution steps or expensive chiral auxiliaries. For R&D directors and procurement specialists, understanding this route is vital as it offers a streamlined approach to accessing high-purity pharmaceutical intermediates with improved operational safety and cost profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of this key biotin intermediate has relied on methodologies that pose substantial engineering and economic challenges. The classic approach employed by major entities like Roche involves the reaction of a furo-imidazole precursor with potassium thioacetate in dimethylformamide at elevated temperatures reaching 150°C. This sulfurizing reagent, potassium thioacetate, is notoriously difficult to prepare with high consistency and exhibits poor thermal stability, creating potential safety hazards during large-scale manufacturing. Alternative routes disclosed in literature, such as those starting from fumaroyl derivatives, necessitate asymmetric alcoholysis induced by heavy amounts of alkaloids like quinine. These traditional pathways are not only laden with tedious purification steps to remove chiral inducers but also suffer from lower atom economy and higher waste generation, making them less attractive for modern green chemistry standards.
The Novel Approach
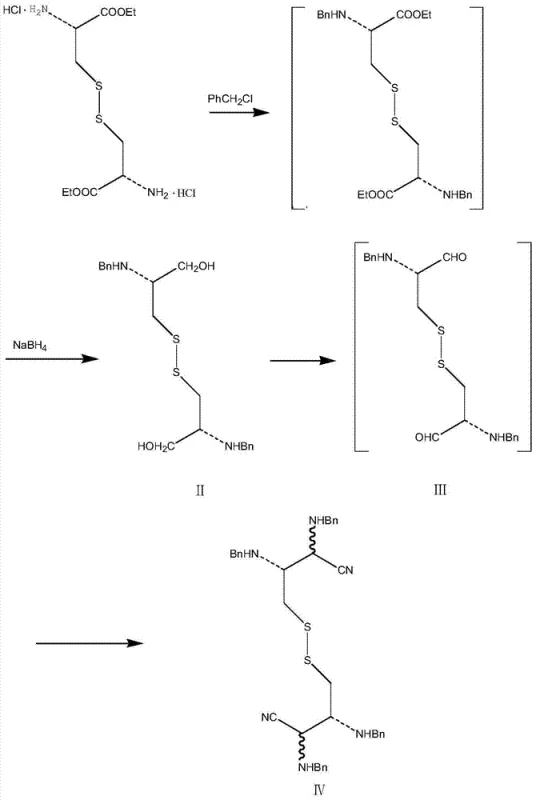
In stark contrast, the novel synthesis route described in the patent data initiates from L-cystine methyl or ethyl ester hydrochloride, a commercially abundant and inexpensive chiral pool material. The process begins with an N-benzyl protection followed by a direct reduction of the ester group to a glycol using sodium borohydride or potassium borohydride. This sequence avoids the harsh thermal conditions of the thioacetate method entirely. As illustrated in the reaction scheme below, the pathway proceeds through a mild oxidation to a dialdehyde and a subsequent Strecker-type reaction to form a dicyano intermediate. This strategic design eliminates the requirement for unstable sulfurizing agents and expensive chiral catalysts, replacing them with stable, commodity chemicals that are easier to source and handle in a GMP environment.
Mechanistic Insights into Zinc-Mediated Disulfide Reduction and Cyclization
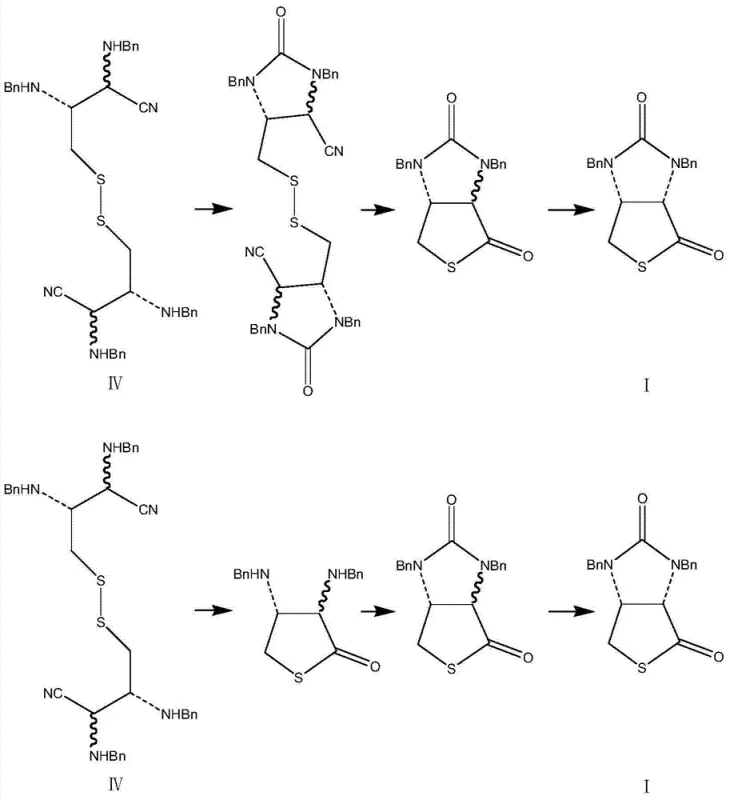
The core innovation of this synthesis lies in the final transformation stages where the linear dicyano precursor is converted into the bicyclic thieno-imidazole framework. The mechanism involves a sophisticated tandem sequence initiated by amidation using solid phosgene or diphosgene, followed by a critical reduction step. The patent specifies the use of a zinc powder-acetic acid-hydrochloric acid system, which is mechanistically distinct from standard hydride reductions. In this system, acetic acid reacts with zinc powder to generate active hydrogen in situ, which selectively reduces the disulfide bond to generate free mercaptan (sulfhydryl) groups. The presence of hydrochloric acid is crucial as it promotes the nucleophilic attack of the newly formed mercaptan onto the nitrile carbon, facilitating lactonization and ring closure. This careful balancing of acid strength and reducing power ensures that the sensitive nitrile groups are converted to amides and subsequently cyclized without degrading the stereochemical integrity established by the L-cystine starting material.
Following the initial cyclization, a thermal rearrangement step is employed to secure the correct stereochemistry at the bridgehead carbons. The crude mixture containing both (3aR, 6aR) and (3aS, 6aR) isomers is subjected to reflux in polar aprotic solvents like DMF or N,N-dimethylacetamide at temperatures between 100°C and 125°C. This thermodynamic equilibration drives the conversion towards the desired (3aS, 6aR) configuration, which is the requisite stereoisomer for downstream biotin synthesis. The ability to correct stereochemistry through a simple thermal rearrangement rather than a complex enzymatic or chromatographic resolution is a major advantage for process chemists aiming for high throughput. The final product is obtained with high liquid phase purity, often exceeding 99%, demonstrating the efficacy of this mechanistic design in controlling impurity profiles.
How to Synthesize (3aS, 6aR)-1,3-Dibenzyltetrahydro-4H-Thieno[3,4-D]Imidazol-2,4-(1H)-Dione Efficiently
The synthesis protocol outlined in the patent provides a clear, step-by-step guide for laboratory and pilot-scale production. The process is divided into three logical phases: the preparation of the chiral glycol backbone, the construction of the dicyano framework, and the final cyclization-rearrangement sequence. Each step utilizes standard organic synthesis unit operations such as extraction, crystallization, and distillation, making it highly adaptable to existing multipurpose reactors. The detailed standardized synthesis steps, including specific molar ratios, temperature controls, and workup procedures, are provided in the technical guide below to assist process engineers in replicating these results.
- Perform N-benzyl glycosylation on L-cystine methyl/ethyl ester hydrochloride followed by borohydride reduction to obtain the glycol compound (Formula II).
- Oxidize the glycol using sulfur trioxide-pyridine complex to form a dialdehyde, then react with benzylamine and sodium cyanide to yield the dicyano compound (Formula IV).
- Execute amidation followed by disulfide reduction using a zinc-acetic acid-hydrochloric acid system to induce cyclization and rearrangement into the final target molecule.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift to this L-cystine-based route offers tangible benefits regarding cost stability and operational reliability. By removing the dependency on specialized reagents like potassium thioacetate or quinine alkaloids, the supply chain becomes significantly more resilient to market fluctuations. The raw materials required, such as benzyl chloride, sodium cyanide, and zinc powder, are bulk commodities with well-established global supply networks, ensuring consistent availability and pricing. Furthermore, the elimination of high-temperature sulfurization steps reduces the energy load on manufacturing facilities and lowers the risk of thermal runaway incidents, contributing to a safer and more sustainable production environment.
- Cost Reduction in Manufacturing: The most significant economic driver of this process is the substitution of expensive chiral inducers and unstable reagents with cheap, achiral commodity chemicals. The use of L-cystine ester as a chiral pool starter means that the cost of chirality is effectively embedded in the low-cost starting material rather than added via expensive catalysts. Additionally, the mild reaction conditions (often between 0°C and 40°C for key steps) reduce utility costs associated with heating and cooling, while the high selectivity minimizes the loss of valuable intermediates to side reactions, thereby improving overall mass balance and yield efficiency.
- Enhanced Supply Chain Reliability: Sourcing potassium thioacetate or high-purity quinine can sometimes lead to bottlenecks due to their niche manufacturing bases. In contrast, the precursors for this novel method are produced by a wide range of chemical manufacturers globally. This diversification of the supplier base mitigates the risk of single-source dependency. Moreover, the stability of the intermediates, such as the glycol and dicyano compounds, allows for potential storage and batch consolidation, providing greater flexibility in production scheduling and inventory management for long-term contracts.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, avoiding the use of heavy metal catalysts that require complex and costly removal steps to meet regulatory limits for pharmaceutical ingredients. The waste streams generated are primarily aqueous salts and organic solvents that can be managed through standard treatment protocols. The avoidance of harsh sulfurizing agents also reduces the generation of malodorous sulfur-containing waste gases, simplifying compliance with environmental regulations and reducing the burden on scrubber systems in the manufacturing plant.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this synthesis route. They are derived from the specific comparative advantages and mechanistic details found in the patent documentation, aimed at clarifying the feasibility of adopting this technology for commercial production.
Q: Why is the traditional Roche method using potassium thioacetate considered problematic for industrial scale-up?
A: The conventional method relies on potassium thioacetate as a sulfurizing reagent, which presents significant handling challenges due to its difficult preparation and poor thermal stability. Furthermore, the reaction requires severe conditions, typically heating to 150°C in DMF, which increases energy costs and safety risks compared to the milder ambient temperature processes described in patent CN103880859A.
Q: How does this novel synthesis route ensure stereochemical purity without expensive chiral catalysts?
A: This process utilizes L-cystine ester hydrochloride as the starting material, which acts as a chiral pool source. By leveraging the inherent chirality of the natural amino acid derivative, the synthesis bypasses the need for expensive alkaloid inducers like quinine or complex asymmetric alcoholysis steps, thereby simplifying the purification workflow and reducing raw material costs significantly.
Q: What are the specific advantages of the zinc-acetic acid-hydrochloric acid reduction system?
A: The use of a zinc powder-acetic acid-hydrochloric acid system allows for the controlled generation of active hydrogen in situ. This specific combination facilitates the selective reduction of the disulfide bond to a mercaptan without over-reduction or degradation of other sensitive functional groups, ensuring high reaction selectivity and facilitating the subsequent intramolecular cyclization to form the thieno-imidazole ring system.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (3aS, 6aR)-1,3-Dibenzyltetrahydro-4H-Thieno[3,4-D]Imidazol-2,4-(1H)-Dione Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the efficient manufacture of vitamins and pharmaceuticals. Our technical team has extensively analyzed the route described in CN103880859A and possesses the expertise to optimize these reactions for large-scale output. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to reactor is seamless. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of (3aS, 6aR)-1,3-dibenzyltetrahydro-4H-thieno[3,4-d]imidazol-2,4-(1H)-dione meets the exacting standards required for downstream API synthesis.
We invite global partners to leverage our manufacturing capabilities to secure a stable supply of this essential biotin intermediate. By collaborating with us, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized processes can enhance your supply chain efficiency and reduce your overall cost of goods sold.
