Advanced Synthesis of Dehydro Amino Acid Derivatives for Enhanced Polypeptide Drug Stability and Commercial Scalability
Advanced Synthesis of Dehydro Amino Acid Derivatives for Enhanced Polypeptide Drug Stability and Commercial Scalability
The pharmaceutical industry is constantly seeking innovative solutions to overcome the inherent instability of polypeptide and protein-based therapeutics, which often suffer from rapid degradation in physiological environments. Patent CN110642753A introduces a groundbreaking class of amino acid derivatives specifically designed to modify polypeptide drugs, effectively resisting proteolytic enzyme degradation and prolonging drug half-life. These novel compounds feature a unique unsaturated structure that provides steric hindrance against enzymatic cleavage while maintaining biological compatibility. As a leading manufacturer, we recognize the immense potential of these derivatives in transforming the pharmacokinetic profiles of next-generation biologics. The structural versatility allows for various protecting groups such as Fmoc and Boc, catering to diverse solid-phase peptide synthesis requirements. 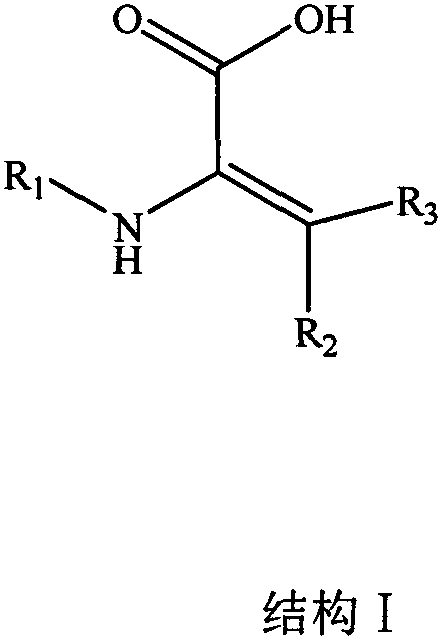
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional strategies for stabilizing polypeptide drugs often rely on PEGylation or the incorporation of non-natural D-amino acids, which can introduce significant immunogenicity risks or alter the binding affinity of the drug to its target receptor. Furthermore, conventional chemical modifications frequently require harsh reaction conditions that are incompatible with sensitive peptide sequences, leading to racemization or side reactions that compromise the final product's purity. The reliance on enzymatic methods for specific modifications often results in prohibitive costs and complex downstream processing due to the difficulty of removing proteinaceous impurities. Additionally, standard L-amino acids are inherently susceptible to protease attack at specific cleavage sites, limiting the oral bioavailability and systemic circulation time of many promising therapeutic candidates. These challenges create a critical bottleneck in the development of long-acting injectables and oral peptide formulations, driving the urgent need for more robust chemical scaffolds.
The Novel Approach
The methodology disclosed in the patent offers a sophisticated chemical solution by introducing a carbon-carbon double bond at the alpha-beta position of the amino acid backbone, creating a dehydro-amino acid structure that is chemically distinct from its saturated counterparts. This unsaturation locks the conformation of the amino acid residue, providing a rigid framework that physically blocks the access of proteolytic enzymes to the peptide bond. The synthesis routes described utilize straightforward organic transformations such as dehydration of hydroxyl-containing side chains or dehydrogenation of saturated precursors, avoiding the need for complex biocatalytic systems. By employing standard protecting group strategies like Fmoc and benzyl esters, these derivatives can be seamlessly integrated into existing solid-phase peptide synthesis workflows without requiring specialized equipment. This approach not only enhances metabolic stability but also simplifies the manufacturing process by utilizing readily available raw materials and scalable reaction conditions suitable for multi-kilogram production.
Mechanistic Insights into Dehydration and Dehydrogenation Strategies
The core chemical innovation lies in the precise control of elimination reactions to generate the desired alkene functionality without compromising the chiral integrity of adjacent centers or the stability of the amine protecting group. In the dehydration pathway, the hydroxyl group of serine or threonine derivatives is first activated, typically through conversion to a leaving group such as a tosylate, followed by base-mediated elimination to form the double bond. This mechanism requires careful optimization of temperature and base strength to prevent over-reaction or polymerization, ensuring high selectivity for the Z or E isomer as required by the specific drug design. Alternatively, the dehydrogenation route employs N-chlorosuccinimide to generate an N-chloro intermediate, which subsequently undergoes elimination to establish the unsaturation, offering a complementary route for amino acids lacking side-chain hydroxyl groups. 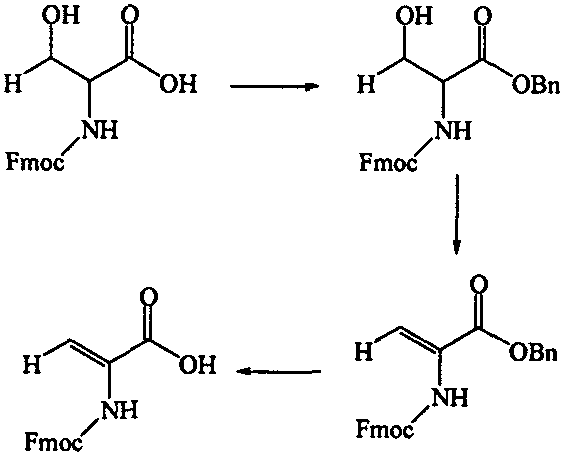
Impurity control is paramount in the synthesis of these intermediates, as trace amounts of saturated starting materials or over-reacted byproducts can significantly impact the efficacy of the final polypeptide drug. The patent details rigorous purification protocols, including recrystallization from specific solvent systems like ethyl acetate and petroleum ether, to achieve the high purity levels demanded by regulatory agencies. The use of benzyl ester protection serves a dual purpose: it masks the carboxylic acid during the harsh dehydration conditions and facilitates purification through precipitation, thereby reducing the burden on chromatographic separation steps. Understanding these mechanistic nuances allows process chemists to anticipate potential failure modes, such as the formation of N-dichloroamines in the dehydrogenation route, and implement mitigation strategies like controlled addition rates and inert atmosphere handling. This depth of chemical understanding ensures that the transition from laboratory bench to commercial plant is smooth and reproducible.
How to Synthesize Fmoc-Dehydroxyserine Efficiently
The synthesis of Fmoc-dehydroxyserine serves as a prime example of the practical application of these patented methods, demonstrating a clear path from commodity chemicals to high-value intermediates. The process begins with the protection of Fmoc-Serine, followed by a dehydration step using p-toluenesulfonyl chloride in pyridine, and concludes with the removal of the benzyl group to reveal the free acid. This sequence highlights the balance between reaction efficiency and product quality, utilizing common reagents that are easily sourced from global chemical suppliers. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below which outlines the critical control points for ensuring batch-to-batch consistency.
- Protect the carboxylic acid group of Fmoc-Serine using benzyl bromide and sodium carbonate in DMF to form the benzyl ester intermediate.
- Perform dehydration by reacting the protected intermediate with p-toluenesulfonyl chloride in dry pyridine at low temperatures followed by room temperature stirring.
- Remove the benzyl protecting group using HBr in TFA solution to yield the final Fmoc-dehydroxyserine product with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of these chemically synthesized dehydro amino acid derivatives offers substantial advantages over biologically derived alternatives, primarily driven by the stability and predictability of the supply chain. The reliance on abundant natural amino acids like serine and valine as starting materials insulates the production process from the volatility associated with fermentation-based feedstocks, ensuring consistent availability even during market fluctuations. The synthetic routes described avoid the use of precious metal catalysts or exotic reagents, which significantly reduces the raw material cost profile and minimizes the environmental footprint associated with heavy metal waste disposal. Furthermore, the robustness of the chemical steps allows for processing in standard glass-lined or stainless steel reactors, eliminating the need for specialized single-use bioreactors and lowering the capital expenditure required for manufacturing setup.
- Cost Reduction in Manufacturing: The elimination of enzymatic steps and the use of crystallization-based purification instead of preparative HPLC drastically lower the operational expenses associated with producing these intermediates. By avoiding expensive biocatalysts and reducing solvent consumption through efficient precipitation techniques, the overall cost of goods sold is significantly optimized, allowing for more competitive pricing of the final polypeptide therapeutics. The ability to recycle solvents like pyridine and DMF further contributes to long-term cost savings and sustainability goals.
- Enhanced Supply Chain Reliability: The synthetic nature of this process ensures that production is not limited by biological growth cycles or contamination risks inherent in fermentation, providing a reliable and continuous supply of critical intermediates. The use of stable protecting groups like Fmoc ensures that the intermediates have a long shelf life, reducing the risk of inventory spoilage and allowing for strategic stockpiling to buffer against demand surges. This reliability is crucial for pharmaceutical companies managing complex global supply chains where continuity of supply is a key performance indicator.
- Scalability and Environmental Compliance: The reaction conditions described, such as room temperature stirring and ambient pressure operations, are inherently safer and easier to scale from pilot plants to multi-ton commercial facilities. The waste streams generated are primarily organic salts and solvents which can be treated using standard industrial wastewater treatment protocols, ensuring compliance with increasingly stringent environmental regulations. This scalability ensures that as the demand for the modified polypeptide drug grows, the supply of the key intermediate can be ramped up rapidly without requiring fundamental process redesigns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these amino acid derivatives in drug development pipelines. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details helps in assessing the feasibility of incorporating these building blocks into new molecular entities.
Q: What is the primary advantage of using dehydro amino acid derivatives in polypeptide drugs?
A: The introduction of a double bond at the alpha-beta position creates a rigid structure that resists proteolytic enzyme degradation, significantly extending the half-life of the therapeutic polypeptide in vivo.
Q: Are the starting materials for these derivatives commercially available?
A: Yes, the synthesis utilizes widely available natural amino acids such as Serine, Threonine, and Valine as starting materials, ensuring a stable and reliable supply chain for large-scale production.
Q: How does the dehydration method compare to enzymatic modification?
A: The chemical dehydration method described in the patent offers superior scalability and avoids the high costs and strict storage conditions associated with enzymatic processes, making it ideal for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dehydro Amino Acid Derivative Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from clinical trials to market launch is supported by a robust manufacturing infrastructure. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications for complex peptide intermediates, guaranteeing that every batch meets the highest international standards. We understand the critical nature of these building blocks in the context of polypeptide drug stability and are committed to delivering materials that enable your R&D success. Our team of expert chemists is ready to collaborate on process optimization to further enhance yield and reduce impurities tailored to your specific needs.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis for your specific project requirements. By engaging with us early in the development cycle, you can secure specific COA data and route feasibility assessments that will de-risk your supply chain and accelerate your time to market. Let us be your partner in bringing these innovative polypeptide therapies to patients worldwide through superior chemical manufacturing excellence.
