Breakthrough Pd-Catalyzed Synthesis of Gem-1,3-Enynes for Commercial Scale Pharmaceutical Manufacturing
The landscape of organic synthesis for conjugated enyne structures is undergoing a significant transformation driven by the need for more efficient, scalable, and selective manufacturing processes. Patent CN113354511B introduces a groundbreaking methodology for the synthesis of gem-1,3-enyne compounds, utilizing a cross-dimerization strategy between alkynol derivatives and aromatic alkyne compounds. This technology addresses long-standing challenges in the field, specifically the difficulty in controlling regio- and stereo-selectivity when coupling two different terminal alkynes. By leveraging a simple yet highly effective palladium and tetramethylethylenediamine (TMEDA) catalytic system, this approach offers a robust pathway for producing high-value pharmaceutical intermediates. The implications for industrial production are profound, as it replaces complex, expensive catalytic systems with readily available reagents that operate under mild thermal conditions. For R&D directors and procurement specialists alike, this represents a shift towards more sustainable and cost-effective supply chains for critical fine chemical building blocks used in the development of bioactive molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of conjugated 1,3-enyne compounds has relied on methodologies that present substantial hurdles for large-scale commercial adoption. Traditional routes often involve Wittig or Horner-Wadsworth-Emmons reactions, which can suffer from poor atom economy and the generation of significant stoichiometric waste. Furthermore, transition metal-catalyzed cross-coupling methods frequently necessitate the use of precious metals such as rhodium or titanium, accompanied by sophisticated, air-sensitive ligands that drive up raw material costs exponentially. These conventional catalytic systems often struggle with selectivity issues, particularly when attempting the cross-dimerization of two distinct terminal alkynes, leading to a complex mixture of homodimerization and heterodimerization byproducts. The presence of up to 12 potential dimerization products in unoptimized systems creates a nightmare for downstream purification, drastically reducing overall yield and increasing the environmental footprint of the manufacturing process due to excessive solvent usage in chromatography.
The Novel Approach
In stark contrast, the novel synthetic method disclosed in the patent data utilizes a streamlined palladium catalyst system paired with TMEDA, which serves a dual function as both a ligand and a base. This innovation simplifies the reaction setup by eliminating the need for pre-formed complex organometallic species or harsh reaction conditions. The process operates efficiently at temperatures ranging from 30°C to 50°C, significantly reducing energy consumption compared to high-temperature alternatives. The strategic use of alkynol derivatives allows the hydroxyl group to act as a directing moiety, guiding the palladium center to form a specific cyclopalladated intermediate. This mechanistic feature ensures that the reaction proceeds with high gem-selectivity, effectively suppressing the formation of unwanted symmetric dimerization products. The result is a cleaner reaction profile that facilitates easier isolation of the target gem-1,3-enyne compound, thereby enhancing the overall viability of the process for industrial scale-up.
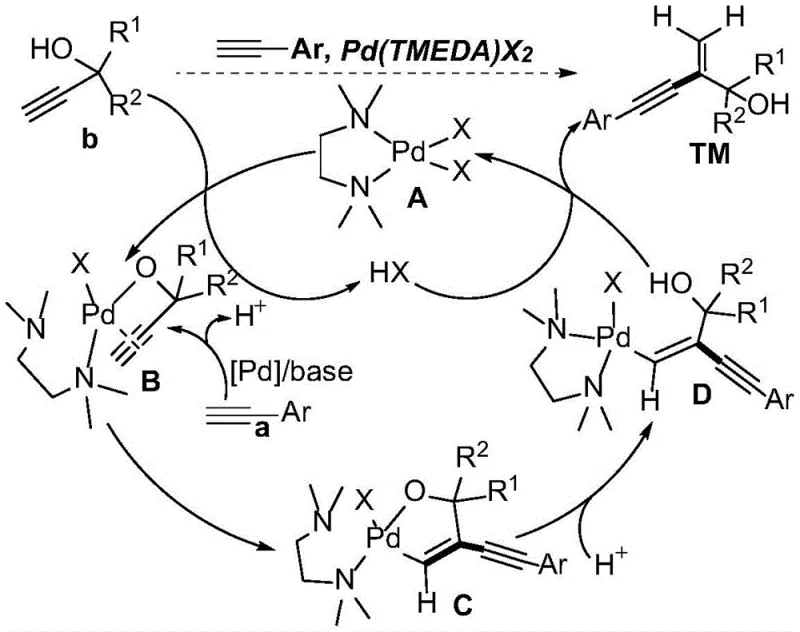
Mechanistic Insights into Pd/TMEDA-Catalyzed Cross Dimerization
To fully appreciate the technical superiority of this synthesis route, one must examine the intricate catalytic cycle that governs the transformation. The reaction initiates with the coordination of the palladium catalyst with the TMEDA ligand, forming an active species capable of engaging with the alkyne substrates. A critical step involves the interaction between the palladium center and the alkynol derivative, where the hydroxyl group facilitates a ligand exchange process. This interaction leads to the formation of a five-membered cyclopalladated intermediate, a key structural motif that locks the conformation of the substrate. This intermediate is crucial because it dictates the subsequent regioselectivity of the alkyne insertion. Unlike traditional syn-carbopalladation pathways often seen in other alkyne couplings, this system promotes an anti-carbopalladation mechanism. This unique stereochemical course is responsible for the exclusive formation of the gem-1,3-enyne architecture, distinguishing it from other dimerization modes that might yield head-to-head or tail-to-tail products.
Furthermore, the impurity control mechanism inherent in this catalytic cycle is of paramount importance for pharmaceutical applications. The specific geometry of the cyclopalladated intermediate sterically hinders the approach of a second molecule of the same alkynol derivative, thereby minimizing homodimerization side reactions. Instead, the intermediate is primed to react with the aromatic alkyne component, which attacks the activated complex to extend the carbon chain. The TMEDA ligand not only stabilizes the palladium center throughout this cycle but also acts as a weak base to facilitate proton transfer steps necessary for product release and catalyst regeneration. This dual functionality reduces the need for additional exogenous bases, simplifying the workup procedure. The ability to suppress 11 out of 12 theoretically possible dimerization byproducts demonstrates a level of chemoselectivity that is rarely achieved with such simple reagent combinations, ensuring that the final product meets stringent purity specifications required for downstream drug synthesis.
How to Synthesize Gem-1,3-Enyne Compounds Efficiently
The practical implementation of this synthesis route is designed to be accessible for standard laboratory and pilot plant facilities without requiring specialized equipment. The general procedure involves charging a reaction vessel with the palladium source, such as palladium chloride or palladium acetate, and the TMEDA ligand in a molar ratio optimized for catalytic turnover. Subsequently, the alkynol derivative and the aromatic alkyne compound are introduced into the mixture along with a compatible organic solvent like chloroform or dichloromethane. The reaction is conducted under an inert nitrogen atmosphere to prevent oxidation of the catalyst or substrates, maintaining a temperature between 30°C and 50°C for a duration of 12 to 24 hours. Following the reaction period, the mixture is cooled to ambient temperature and subjected to a standard aqueous workup involving washing with saturated sodium chloride solution. The organic layer is then separated, dried, and concentrated under reduced pressure. Final purification is achieved through column chromatography, yielding the target gem-1,3-enyne compound with high stereochemical integrity. For detailed standardized operating procedures and safety data sheets regarding this specific synthesis pathway, please refer to the technical documentation provided below.
- Charge a reaction vessel with palladium catalyst (e.g., PdCl2), TMEDA ligand, alkynol derivative, and aromatic alkyne compound in a suitable solvent like chloroform.
- Maintain the reaction mixture under a nitrogen atmosphere and stir continuously at a controlled temperature between 30-50°C for 12 to 24 hours.
- Upon completion, cool to room temperature, wash with saturated NaCl, extract with ethyl acetate, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this Pd/TMEDA catalytic technology offers compelling advantages that directly impact the bottom line and supply chain resilience. The primary driver for cost optimization lies in the drastic simplification of the catalyst system. By replacing expensive, proprietary titanium or rhodium complexes with commodity-grade palladium salts and TMEDA, manufacturers can significantly reduce the raw material cost per kilogram of the active intermediate. This reduction is not merely marginal; it fundamentally alters the cost structure of the synthesis, making it competitive even in high-volume markets. Additionally, the mild reaction conditions translate to lower energy expenditures, as there is no need for cryogenic cooling or high-temperature heating mantles, further contributing to operational efficiency. The simplified workup procedure also reduces solvent consumption and waste disposal costs, aligning with modern green chemistry initiatives that are increasingly mandated by global regulatory bodies.
- Cost Reduction in Manufacturing: The elimination of complex, air-sensitive ligands and precious metal catalysts like rhodium results in substantial cost savings on reagent procurement. Since the catalyst loading is low and the reagents are commercially abundant, the dependency on volatile specialty chemical markets is minimized. Furthermore, the high selectivity of the reaction reduces the burden on purification processes, meaning less silica gel and fewer solvents are required to achieve pharmaceutical-grade purity. This efficiency gain allows for a more predictable budget allocation for production runs, shielding the project from sudden price spikes in niche catalytic materials.
- Enhanced Supply Chain Reliability: Sourcing reliability is greatly improved because the key reagents, such as palladium chloride and TMEDA, are produced by multiple global suppliers, reducing the risk of single-source bottlenecks. Unlike custom-synthesized ligands that may have long lead times, these commodities can often be sourced with short notice, ensuring continuity of supply even during market disruptions. The robustness of the reaction conditions also means that the process is less susceptible to variations in utility quality, such as minor fluctuations in cooling water temperature, making it easier to transfer between different manufacturing sites or contract development and manufacturing organizations (CDMOs) without extensive re-validation.
- Scalability and Environmental Compliance: The process is inherently scalable due to its exothermic profile being manageable under mild conditions, allowing for safe operation in large-scale reactors without complex heat exchange requirements. The high atom economy and reduced byproduct formation mean that the environmental load per unit of product is significantly lower, facilitating easier compliance with environmental protection regulations. This sustainability advantage is increasingly becoming a prerequisite for partnerships with major pharmaceutical companies who are committed to reducing their carbon footprint. The ability to run the reaction at near-ambient temperatures also enhances operator safety, reducing the risk of thermal runaway incidents associated with more aggressive coupling chemistries.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this gem-1,3-enyne synthesis technology. These insights are derived directly from the experimental data and mechanistic studies presented in the patent literature, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for project managers evaluating the feasibility of integrating this route into their existing pipeline. The answers reflect the consensus on the method's performance regarding selectivity, substrate scope, and operational parameters, ensuring that stakeholders have accurate expectations before initiating pilot studies.
Q: What are the primary advantages of this Pd/TMEDA system over traditional Titanium or Rhodium catalysts?
A: The Pd/TMEDA system utilizes commercially available, inexpensive reagents compared to complex, air-sensitive Ti or Rh catalysts. It operates under significantly milder conditions (30-50°C) and achieves superior regio- and stereo-selectivity, effectively suppressing the formation of 11 potential byproduct isomers common in alkyne dimerization.
Q: Does this synthesis method support functional group compatibility for complex drug intermediates?
A: Yes, the method demonstrates excellent functional group tolerance. It is compatible with sensitive groups including silicon-based moieties, aldehydes, hydroxyls, and amines, making it highly suitable for the late-stage modification of complex pharmaceutical intermediates without requiring extensive protecting group strategies.
Q: How does the hydroxyl group in the alkynol derivative influence the reaction selectivity?
A: The hydroxyl group plays a critical directing role by facilitating ligand exchange and metal-pi coordination with the palladium center. This forms a specific cyclopalladated intermediate that guides the anti-carbopalladation pathway, ensuring high gem-selectivity and preventing random homodimerization of the terminal alkynes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gem-1,3-Enyne Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the acceleration of drug discovery and development programs. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch of gem-1,3-enyne compounds meets the exacting standards required for pharmaceutical applications. Our commitment to excellence extends beyond mere manufacturing; we actively collaborate with our clients to optimize reaction parameters for maximum yield and minimal environmental impact, leveraging our deep understanding of palladium-catalyzed transformations.
We invite you to discuss your specific project requirements with our technical procurement team to explore how this advanced synthesis method can benefit your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clearer picture of the potential economic advantages tailored to your volume needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments for your target molecules. Let us partner with you to deliver reliable, high-purity chemical solutions that drive your innovation forward.
