Scalable Synthesis of 1-Azaspiro[4.4]nonan-6-one via Gold-Catalyzed Rearrangement
Scalable Synthesis of 1-Azaspiro[4.4]nonan-6-one via Gold-Catalyzed Rearrangement
The development of efficient synthetic routes for complex spirocyclic scaffolds remains a paramount challenge in modern organic chemistry, particularly for applications in asymmetric catalysis and pharmaceutical intermediates. Patent CN107879967B introduces a groundbreaking preparation method for 1-azaspiro[4.4]nonan-6-one, a critical backbone for SPD chiral ligands and catalysts. This technology leverages a sophisticated semipinacol rearrangement strategy initiated by gold catalysis to construct the quaternary spiro center with high precision. Unlike traditional methods that often suffer from lengthy sequences and difficult purifications, this novel approach streamlines the construction of the azaspiro skeleton from readily available homopropargyl alcohol. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this patent represents a significant leap forward in process efficiency, offering a pathway to high-purity chiral building blocks that are essential for next-generation drug discovery and catalytic applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of azaspiro[4.4]nonane skeletons has been fraught with significant operational hurdles that impede commercial viability. Early methodologies, such as those reported by the Sha group in 1991, while foundational, often involved multi-step sequences with poor atom economy and required rigorous purification techniques like column chromatography at multiple stages. Furthermore, previous attempts to access chiral variants frequently relied on chiral induction routes that were not only synthetically long but also resulted in troublesome separation and purification processes. These conventional pathways often failed to deliver the necessary throughput for industrial applications, limiting the availability of these valuable ligands for large-scale asymmetric synthesis. The inability to realize large-scale preparation effectively restricted the broader application of azaspiro-based catalysts in the fine chemical industry, creating a supply bottleneck for downstream users requiring consistent quality and volume.
The Novel Approach
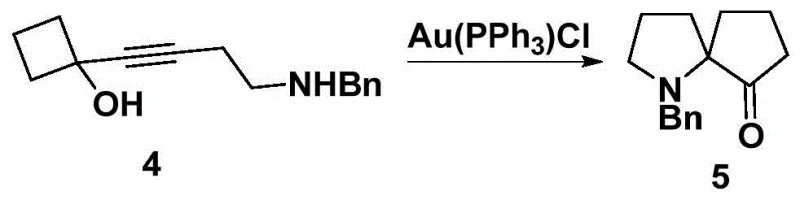
The methodology disclosed in CN107879967B fundamentally reimagines the construction of the spiro core by employing a semipinacol rearrangement driven by a gold catalyst. This innovative route begins with simple homopropargyl alcohol, undergoing hydroxyl protection, 1,2-addition, and substitution to set up the precursor for the critical cyclization. The use of Au(PPh3)Cl facilitates a rapid and high-yielding formation of the racemic aza[4.4]spiro compound, bypassing the need for complex chiral pool starting materials at the early stages. By deferring the chirality introduction to a robust resolution step involving tartaric and camphorsulfonic acids, the process achieves exceptional flexibility, allowing for the isolation of both (S) and (R) enantiomers in high optical purity. This strategic shift not only shortens the overall synthetic timeline but also drastically simplifies the workup procedures, making it an ideal candidate for cost reduction in chiral ligand manufacturing.
![Overall synthetic route from homopropargyl alcohol to chiral 1-azaspiro[4.4]nonan-6-one derivatives](/insights/img/1-azaspiro-synthesis-gold-catalysis-pharma-supplier-20260305032804-01.webp)
Mechanistic Insights into Au(PPh3)Cl-Catalyzed Semipinacol Rearrangement
The heart of this synthetic innovation lies in the gold-catalyzed cyclization step, which elegantly constructs the congested quaternary spiro center. In this transformation, the gold(I) species coordinates to the alkyne moiety of the benzylamine-substituted precursor, activating it towards nucleophilic attack by the proximal hydroxyl group or facilitating the migration of the carbon framework. This activation lowers the energy barrier for the semipinacol rearrangement, allowing the reaction to proceed smoothly at moderate temperatures ranging from 40°C to 60°C. The mild conditions are crucial for maintaining the integrity of the sensitive functional groups present in the molecule, thereby minimizing the formation of degradation byproducts. The catalytic cycle is highly efficient, requiring only 0.1 to 1 mol% of the gold catalyst, which underscores the economic feasibility of the process for large-scale operations where catalyst cost is a significant factor.
From an impurity control perspective, the specificity of the gold catalyst plays a pivotal role in ensuring the high quality of the final API intermediate. The rearrangement proceeds with high regioselectivity, favoring the formation of the desired five-membered lactam ring fused to the cyclobutane system. This inherent selectivity reduces the burden on downstream purification units, as fewer structural isomers are generated compared to non-catalyzed thermal rearrangements. Furthermore, the subsequent chiral resolution strategy employs a dual-system approach, first using tartaric acid for a crude separation followed by camphorsulfonic acid for fine tuning. This layered resolution technique ensures that the final products, such as (S)-9 and (R)-9, achieve enantiomeric excess values exceeding 99%, meeting the stringent purity specifications required for use in asymmetric catalysis and pharmaceutical synthesis.
How to Synthesize 1-Azaspiro[4.4]nonan-6-one Efficiently
The synthesis protocol outlined in the patent provides a clear, step-by-step guide for producing both enantiomers of the target spiro ketone. The process is designed to be robust and scalable, utilizing common laboratory reagents and standard equipment such as four-necked flasks and double-layer reactors. Key to the success of this route is the careful control of temperature during the lithiation and substitution steps, as well as the precise stoichiometry of the resolving agents. The following sections detail the operational parameters necessary to replicate the high yields and purity described in the intellectual property, serving as a foundational reference for process chemists aiming to implement this technology.
- Protect homopropargyl alcohol with TsCl and triethylamine to form the tosyl ester.
- Perform 1,2-addition with cyclobutanone using n-BuLi to generate the hydroxy-alkyne intermediate.
- Substitute the tosyl group with benzylamine to introduce the nitrogen functionality.
- Execute the key Semipinacol rearrangement using Au(PPh3)Cl catalyst to form the spiro cyclic core.
- Remove the benzyl group via hydrogenation and perform sequential chiral resolution using tartaric acid and camphorsulfonic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from legacy synthesis methods to this novel gold-catalyzed route offers substantial strategic benefits. The most immediate impact is seen in the simplification of the manufacturing workflow, which directly translates to improved operational efficiency and reduced overhead costs. By eliminating the reliance on column chromatography for the bulk of the purification, the process becomes far more amenable to continuous flow or large-batch processing, significantly enhancing the reliability of supply. This robustness is critical for maintaining uninterrupted production schedules in the fast-paced pharmaceutical and agrochemical sectors, where delays in intermediate delivery can have cascading effects on final drug substance manufacturing.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the high efficiency of the key cyclization step and the use of inexpensive, commercially available starting materials like homopropargyl alcohol and cyclobutanone. The low loading of the gold catalyst (0.1-1 mol%) ensures that precious metal costs do not become a prohibitive factor, while the high yields reported in the examples (e.g., 90% yield for the cyclization step) maximize material throughput. Furthermore, the avoidance of expensive chiral starting materials in favor of a resolution strategy allows for the utilization of cheaper racemic precursors, driving down the overall cost of goods sold for the final chiral intermediates.
- Enhanced Supply Chain Reliability: The ability to produce both (S) and (R) enantiomers from a common racemic intermediate provides a significant buffer against supply chain volatility. Instead of managing two distinct synthetic lines for opposite enantiomers, manufacturers can consolidate production into a single streamlined process up to the resolution stage. This flexibility ensures that demand fluctuations for either stereoisomer can be met rapidly without the need for retooling or sourcing different raw materials. Additionally, the use of stable resolving agents like tartaric acid and camphorsulfonic acid, which are widely available in the global market, mitigates the risk of raw material shortages that often plague more exotic synthetic routes.
- Scalability and Environmental Compliance: The process has been demonstrated to scale effectively, with examples showing successful execution on multi-kilogram scales (e.g., 3000g batches). The reaction conditions are relatively mild, avoiding extreme pressures or temperatures that would require specialized high-cost reactor infrastructure. From an environmental standpoint, the high atom economy of the rearrangement and the potential for solvent recovery in the crystallization steps contribute to a greener manufacturing profile. This alignment with green chemistry principles facilitates easier regulatory approval and supports corporate sustainability goals, making the supply of these intermediates more resilient in an increasingly regulated global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a transparent view of the process capabilities and limitations for potential partners and licensees.
Q: What is the primary advantage of this synthesis route over prior art?
A: The primary advantage is the elimination of column chromatography for the core spiro structure and the ability to access both (S) and (R) enantiomers efficiently through a robust resolution process, significantly simplifying large-scale manufacturing.
Q: What catalyst is used for the critical cyclization step?
A: The process utilizes Triphenylphosphine Gold Chloride (Au(PPh3)Cl) at a low loading of 0.1-1 mol% to drive the Semipinacol rearrangement, ensuring high efficiency and minimizing heavy metal contamination risks.
Q: Is this method suitable for industrial scale-up?
A: Yes, the patent explicitly demonstrates successful execution on a multi-kilogram scale (e.g., 3000g batches in Example 3), utilizing standard reagents and avoiding exotic conditions, making it highly viable for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Azaspiro[4.4]nonan-6-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this gold-catalyzed semipinacol rearrangement technology for the production of high-value chiral ligands. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from benchtop discovery to full-scale manufacturing. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, including the high enantiomeric excess required for advanced asymmetric catalysis applications. We are committed to delivering consistent quality and supply continuity for complex pharmaceutical intermediates.
We invite you to collaborate with us to leverage this efficient synthetic route for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this process can optimize your budget without compromising on quality. Please contact our technical procurement team today to request specific COA data for our spiro-intermediate portfolio and to discuss detailed route feasibility assessments for your upcoming campaigns.
