Advanced Nickel-Catalyzed Hydrogenation for High-Purity Camptothecin Intermediates
The pharmaceutical industry continuously seeks more efficient pathways for synthesizing complex oncology intermediates, and patent CN102046634A presents a significant breakthrough in the production of camptothecin derivatives. This intellectual property details a novel method for the selective hydrogenation of camptothecin or its derivatives to produce corresponding 1,2,6,7-tetrahydro compounds, which serve as critical precursors for potent anticancer agents like 10-hydroxy-20-(S)-camptothecin and irinotecan. Historically, this transformation has been plagued by issues of over-reduction and the reliance on prohibitively expensive noble metal catalysts. The disclosed innovation replaces these costly systems with a robust nickel-based catalytic framework, fundamentally altering the economic and technical landscape for manufacturers of high-purity pharmaceutical intermediates. By eliminating the need for complex catalyst modifiers and reducing the risk of forming unwanted over-reduced byproducts, this technology offers a streamlined route that aligns perfectly with modern green chemistry principles and cost-efficiency mandates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those described in Japanese Patent Documents 59-5188 and 2848958, predominantly relied on platinum or palladium catalysts to effect the reduction of the quinoline skeleton in camptothecin. A major technical hurdle with these noble metal systems was their tendency to drive the reaction beyond the desired 1,2,6,7-tetrahydro stage, leading to the formation of fully reduced 9,10,11,12-tetrahydro byproducts which are difficult to separate and represent a significant yield loss. To mitigate this, previous processes necessitated the addition of hydrogenation catalyst regulators or poisons, such as dimethyl sulfoxide (DMSO) or sulfur compounds, to dampen the catalyst activity. However, this approach introduced new complications, including the requirement for exceptionally high catalyst loadings (up to 50% by weight relative to the substrate) and the subsequent challenge of removing these toxic sulfur additives from the final API intermediate, thereby complicating the purification workflow and increasing environmental disposal costs.
The Novel Approach
In stark contrast, the method disclosed in CN102046634A utilizes a stable nickel catalyst system that inherently possesses the appropriate activity profile to stop reduction at the desired 1,2,6,7-tetrahydro stage without the need for external modifiers. This approach leverages specific nickel formulations, such as those supported on diatomaceous earth, which provide a unique surface environment that favors the selective hydrogenation of the heterocyclic rings while leaving other sensitive functionalities intact. 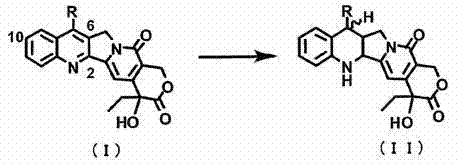 This elimination of catalyst poisons not only simplifies the reaction mixture but also drastically reduces the raw material costs associated with both the catalyst and the auxiliary chemicals. Furthermore, the process demonstrates remarkable robustness against over-reduction even under elevated hydrogen pressures, allowing for faster reaction kinetics and higher throughput compared to the sluggish, modifier-dependent platinum systems of the past.
This elimination of catalyst poisons not only simplifies the reaction mixture but also drastically reduces the raw material costs associated with both the catalyst and the auxiliary chemicals. Furthermore, the process demonstrates remarkable robustness against over-reduction even under elevated hydrogen pressures, allowing for faster reaction kinetics and higher throughput compared to the sluggish, modifier-dependent platinum systems of the past.
Mechanistic Insights into Nickel-Catalyzed Selective Hydrogenation
The core of this technological advancement lies in the specific nature of the "stable nickel catalyst" employed, which differs significantly from standard Raney nickel or reduced nickel powders. These catalysts are prepared by completely reducing nickel oxide and then intentionally re-oxidizing the surface to form a protective oxide film. This passivation layer prevents pyrophoricity, allowing the catalyst to be handled safely in air, while still maintaining high hydrogenation activity once introduced into the reaction medium. Mechanistically, the nickel active sites interact with the quinoline moiety of the camptothecin substrate in a manner that facilitates the addition of hydrogen across the 1,2 and 6,7 positions preferentially. The electronic and steric properties of the nickel-diatomaceous earth interface appear to disfavor the further reduction of the remaining aromatic rings, effectively acting as a built-in selectivity filter that noble metals lack without artificial poisoning.
From an impurity control perspective, this mechanism is vital for ensuring the quality of the resulting pharmaceutical intermediate. The patent data indicates that the process yields a favorable ratio of diastereomers (specifically the RRT 0.62 and RRT 0.90 isomers) while minimizing the formation of the RRT 1.24 over-reduced byproduct to negligible levels. This high selectivity is crucial because downstream oxidation steps to generate 10-hydroxy derivatives are highly sensitive to the structural integrity of the starting tetrahydro material. By avoiding the formation of deep-reduction impurities, the nickel-catalyzed route simplifies the downstream purification burden, reducing the need for extensive chromatography or recrystallization steps that typically erode overall process yield and increase the cost of goods sold for the final active pharmaceutical ingredient.
How to Synthesize 1,2,6,7-Tetrahydrocamptothecin Efficiently
The operational protocol for this synthesis is designed for scalability and ease of execution in standard chemical manufacturing facilities. The process begins by suspending the camptothecin starting material in a non-reactive solvent, with acetic acid being the preferred medium due to its excellent solubility characteristics for the substrate and compatibility with the nickel catalyst. The reaction is conducted in a pressure vessel capable of withstanding moderate hydrogen pressures, typically ranging from 1 to 3 MPa. Heating the mixture to temperatures between 100°C and 140°C accelerates the reaction rate, allowing for complete conversion within a practical timeframe of 1 to 10 hours. The detailed standardized synthesis steps, including specific workup procedures and isolation techniques, are outlined below.
- Suspend camptothecin substrate in a non-reactive solvent such as acetic acid within a high-pressure autoclave reactor.
- Add a stable nickel catalyst (e.g., N113 or N103 supported on diatomaceous earth) at a loading of 10-40% relative to the substrate.
- Replace the atmosphere with hydrogen, pressurize to 1-3 MPa, and heat to 100-140°C for 1-10 hours until conversion is complete.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from noble metal to base metal catalysis represents a substantial opportunity for cost optimization and risk mitigation. The volatility of platinum and palladium prices has long been a concern for the financial planning of API manufacturing, and replacing these with abundant nickel significantly stabilizes the raw material cost structure. Moreover, the elimination of expensive and hazardous catalyst modifiers like DMSO removes an entire class of chemicals from the supply chain, simplifying logistics and reducing the regulatory burden associated with handling and disposing of sulfur-containing waste streams. This streamlined chemical profile translates directly into a more resilient and cost-effective manufacturing process.
- Cost Reduction in Manufacturing: The substitution of platinum with nickel results in a drastic reduction in catalyst expenditure, as nickel is orders of magnitude cheaper than precious metals. Additionally, the process operates effectively with lower catalyst loadings compared to the modified platinum systems, further driving down the variable cost per kilogram of product. The simplified workup, which avoids the complex removal of sulfur poisons, reduces solvent consumption and energy usage during purification, contributing to substantial overall cost savings in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: Nickel catalysts are widely available from multiple global suppliers, reducing the risk of supply bottlenecks that can occur with specialized noble metal catalysts. The robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in operating parameters, leading to more consistent batch-to-batch quality and reliable delivery schedules. This stability is critical for maintaining continuous production lines for high-demand oncology drugs, ensuring that downstream customers receive their materials on time without interruption.
- Scalability and Environmental Compliance: The use of a non-pyrophoric, stable nickel catalyst enhances operational safety during large-scale handling and charging operations, facilitating easier scale-up from pilot plant to commercial production volumes. Furthermore, the absence of toxic sulfur modifiers aligns the process with increasingly stringent environmental regulations regarding waste discharge and worker safety. This eco-friendly profile not only reduces compliance costs but also enhances the sustainability credentials of the supply chain, a key factor for modern pharmaceutical partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nickel-catalyzed hydrogenation technology. These answers are derived directly from the experimental data and comparative examples provided in the patent literature, offering a clear picture of the process capabilities and limitations for potential adopters.
Q: Why is nickel preferred over platinum for camptothecin hydrogenation?
A: Traditional platinum catalysts often require expensive modifiers like DMSO and still risk over-reduction to 9,10,11,12-tetrahydro byproducts. Nickel catalysts offer superior selectivity for the 1,2,6,7-position without needing toxic modifiers, significantly lowering raw material costs.
Q: Does this process require specialized catalyst poisons to control selectivity?
A: No. Unlike prior art methods that necessitate sulfur compounds or CO to poison noble metal catalysts, this nickel-based system achieves high selectivity inherently through the use of stable nickel formulations on diatomaceous earth supports.
Q: What are the typical reaction conditions for this transformation?
A: The reaction typically proceeds in acetic acid solvent at temperatures between 100°C and 140°C under hydrogen pressures of 1 to 3 MPa, offering a balance between reaction rate and safety suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,6,7-Tetrahydrocamptothecin Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting advanced catalytic technologies like the nickel-mediated hydrogenation described in CN102046634A to enhance our portfolio of oncology intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our facility is equipped with rigorous QC labs and stringent purity specifications that guarantee every batch of 1,2,6,7-tetrahydrocamptothecin meets the exacting standards required for GMP API synthesis, providing our partners with absolute confidence in material quality.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cost-effective and robust synthetic route. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this nickel-based process can optimize your bill of materials. We encourage you to contact us today to discuss route feasibility assessments and to obtain specific COA data for our camptothecin derivative intermediates, securing a competitive advantage in the global oncology market.
