Scalable Purification of Desmopressin Acetate: Eliminating Toxic Impurities for Global Pharma Supply
Introduction to Advanced Desmopressin Purification Technologies
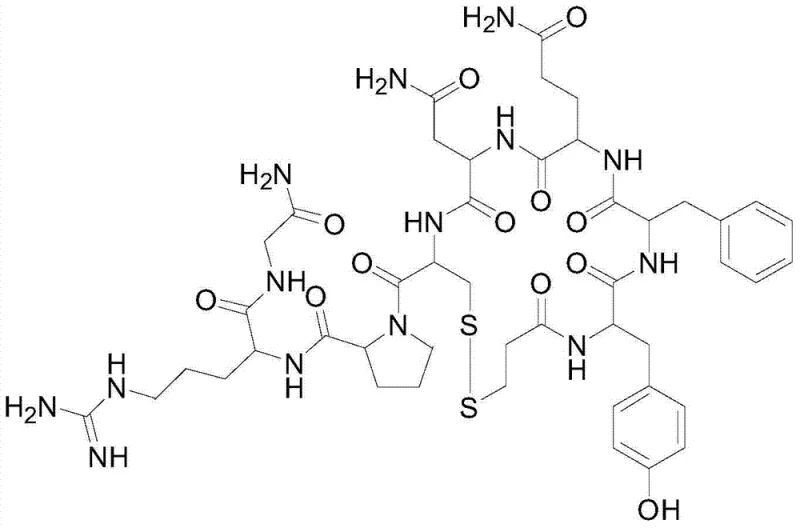
The pharmaceutical landscape for peptide hormones demands increasingly rigorous standards for purity and impurity control, particularly for critical therapeutics like Desmopressin Acetate. Patent CN103467574A introduces a transformative purification methodology that addresses the persistent challenge of removing the toxic des-Gln4-desmopressin impurity, a deletion variant that compromises product stability and safety. This technical breakthrough utilizes a specialized polymer reversed-phase chromatographic system, diverging from traditional silica-based approaches to achieve unprecedented separation efficiency. By leveraging a precise acetic acid-acetonitrile gradient on a robust polymer stationary phase, the process ensures that the final Active Pharmaceutical Ingredient (API) meets the stringent requirement of less than 0.05% for this specific genotoxic-related impurity. For global procurement leaders, this represents a shift towards more reliable pharmaceutical intermediate supplier capabilities, where consistency in impurity profiles is as critical as overall yield. The technology not only enhances the safety margin for patients treating hemophilia and diabetes insipidus but also streamlines the downstream processing workflow by eliminating complex salt conversion steps associated with older methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of Desmopressin has relied heavily on octadecylsilane (ODS) chemically bonded silica columns, often employing mobile phases containing trifluoroacetic acid (TFA) or phosphate buffers. While effective to a degree, these conventional methods present significant operational and chemical drawbacks that hinder large-scale cost reduction in pharmaceutical manufacturing. The use of non-volatile phosphate buffers necessitates an additional anion exchange step to convert the final product into the acetate salt form, adding complexity, time, and potential yield loss to the production cycle. Furthermore, silica-based stationary phases have limited pH stability and can suffer from batch-to-batch variability in selectivity, making the consistent removal of the elusive des-Gln4 impurity difficult. In many documented cases using prior art methods, the content of this specific impurity remains stubbornly above 0.4%, failing to meet the premium quality standards required for modern regulatory submissions. The reliance on TFA also introduces environmental and handling concerns, as removing trace fluoride residues requires extensive washing and drying protocols, further impacting the overall process economics and throughput efficiency.
The Novel Approach
The innovative strategy outlined in the patent data replaces fragile silica supports with a high-performance polymer reversed-phase packing material, such as UniPS, which offers superior chemical inertness and mechanical stability. This fundamental shift in stationary phase chemistry allows for the use of a simpler, volatile mobile phase system consisting of 0.3% aqueous acetic acid and acetonitrile, completely bypassing the need for phosphate buffers and the subsequent salt conversion unit operations. The core of this novelty lies in the optimized gradient elution program, which initiates at a low organic modifier concentration (5% acetonitrile) and employs a shallow, multi-stage ramp to 40% over 30 minutes. This precise control over the mobile phase strength maximizes the resolution between the target Desmopressin peak and the closely eluting des-Gln4 impurity, achieving a separation factor that silica columns cannot match. Consequently, the process delivers a purified product with purity exceeding 99.9% and impurity levels reduced to 0.01%, demonstrating a clear technological leap forward for any high-purity pharmaceutical intermediate production line seeking to minimize risk and maximize quality.
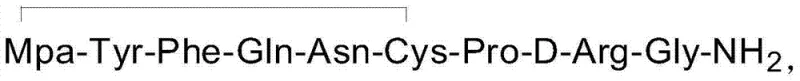
Mechanistic Insights into Polymer Reversed-Phase Chromatography
The success of this purification protocol is rooted in the unique interaction mechanisms between the hydrophobic polymer surface and the amphiphilic peptide molecules. Unlike silica, which possesses residual silanol groups that can cause secondary ionic interactions and peak tailing, the polymer matrix provides a purely hydrophobic environment that promotes ideal partitioning behavior based on the peptide's conformational state. The des-Gln4 impurity, lacking the polar glutamine side chain, exhibits slightly different hydrophobicity compared to the full-length Desmopressin. On a polymer column, this subtle difference is amplified, allowing the gradient to effectively resolve the two species. The initial low-percentage hold of acetonitrile ensures that all peptide components are strongly retained at the column head, focusing the band before the separation begins. As the acetonitrile concentration increases linearly, the solvent strength gradually disrupts the hydrophobic interactions, eluting the slightly less hydrophobic impurity first, followed by the target molecule. This mechanism ensures that the commercial scale-up of complex peptides is not limited by column overload effects, as the polymer support maintains its structural integrity and selectivity even under preparative loading conditions.
Furthermore, the choice of acetic acid as the ion-pairing agent plays a crucial role in the mechanistic pathway of impurity control. Acetic acid acts as a volatile buffer that suppresses the ionization of acidic and basic residues on the peptide without forming non-volatile salts. This volatility is key to the final isolation step; upon lyophilization, the acetic acid sublimes, leaving behind the pure Desmopressin Acetate without the need for diafiltration or resin treatment. This contrasts sharply with TFA, which binds strongly to peptides and is notoriously difficult to remove completely, often requiring repeated co-evaporation with acetic acid. By eliminating strong ion-pairing agents like TFA, the process reduces the risk of introducing new impurities during the workup phase. The result is a cleaner impurity profile where the primary concern is solely the separation of synthesis-related byproducts like des-Gln4, rather than managing reagent-derived contaminants, thereby simplifying the analytical burden on quality control laboratories.
How to Synthesize Desmopressin Acetate Efficiently
The implementation of this purification strategy requires strict adherence to the defined gradient parameters and column specifications to ensure reproducibility. The process begins with the dissolution of the crude peptide in water, followed by pH adjustment to ensure the peptide is in a cationic state compatible with the acidic mobile phase. Once loaded onto the equilibrated polymer column, the gradient elution must be executed with high-precision pumps to maintain the shallow slope required for resolving the critical impurity pair. Detailed standard operating procedures regarding flow rates (typically 80 mL/min for the specified column dimensions) and detection wavelengths (220 nm) are essential for monitoring the elution profile in real-time. For a comprehensive guide on the exact operational parameters and step-by-step execution, please refer to the standardized protocol below.
- Dissolve the Desmopressin crude product in purified water and adjust the pH to an acidic range (approx. 4.5) using acetic acid to ensure solubility and column compatibility.
- Equilibrate the polymer reversed-phase chromatographic column (e.g., UniPS 10-100) by flushing with Mobile Phase A (0.3% aqueous acetic acid) to establish a stable baseline.
- Load the acidified crude peptide solution onto the column and initiate a precise gradient elution, increasing acetonitrile concentration from 5% to 40% over 30 minutes to separate impurities.
- Collect the specific elution fractions containing the target Desmopressin peak while excluding the early-eluting des-Gln4 impurity fractions.
- Lyophilize (freeze-dry) the collected pure fractions to obtain the final Desmopressin Acetate powder with purity exceeding 99.9%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this polymer-based purification method translates directly into tangible operational efficiencies and risk mitigation. The elimination of phosphate buffers and the associated anion exchange salt conversion step significantly simplifies the manufacturing workflow, reducing the number of unit operations and the corresponding labor and equipment time required. This streamlining effect leads to substantial cost savings in terms of consumables, as expensive silica columns with shorter lifespans are replaced by durable polymer columns that withstand rigorous cleaning and sanitization cycles. Moreover, the use of acetic acid instead of trifluoroacetic acid reduces the hazard profile of the process, lowering the costs associated with hazardous waste disposal and worker safety compliance. These factors combined create a more resilient supply chain capable of meeting high-volume demand without the bottlenecks typically associated with complex peptide purification.
- Cost Reduction in Manufacturing: The transition to a volatile acetic acid mobile phase eliminates the need for post-purification salt conversion, a step that traditionally consumes significant resources and time. By removing this unit operation, manufacturers can reduce solvent consumption and avoid the yield losses inherent in multi-step isolation processes. Additionally, the higher selectivity of the polymer column minimizes the need for re-processing batches that fail impurity specifications, thereby protecting margins and ensuring that raw material costs are fully realized in the final product yield.
- Enhanced Supply Chain Reliability: Polymer chromatography media offers superior lot-to-lot consistency compared to silica-based alternatives, reducing the risk of batch failures due to column variability. This reliability is critical for maintaining continuous production schedules and meeting strict delivery timelines for global pharmaceutical clients. The robustness of the polymer support also extends column lifetime, reducing the frequency of column replacement and the associated supply chain disruptions caused by sourcing specialized chromatography hardware.
- Scalability and Environmental Compliance: The method is inherently scalable, as the polymer packing material maintains its physical properties under the high pressures and flow rates required for industrial-scale production. The use of acetic acid and acetonitrile facilitates easier solvent recovery and recycling, aligning with green chemistry principles and reducing the environmental footprint of the manufacturing site. This compliance with environmental standards future-proofs the supply chain against tightening regulations on volatile organic compounds and hazardous waste discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this purification technology. They are derived from the specific performance data and comparative advantages highlighted in the patent literature, providing clarity on how this method outperforms legacy techniques. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this process into existing GMP facilities.
Q: Why is the removal of des-Gln4-desmopressin critical in API manufacturing?
A: The impurity des-Gln4-desmopressin is structurally similar but lacks the glutamine residue, making it unstable and prone to discoloration. Its presence compromises the safety profile and shelf-life of the final hemostatic medication, necessitating reduction to below 0.1% levels.
Q: How does polymer chromatography improve upon traditional silica-based purification?
A: Traditional silica (C18) columns often require harsh mobile phases like trifluoroacetic acid (TFA) or phosphate buffers, which complicate salt conversion and waste treatment. Polymer columns offer superior chemical stability, allow the use of volatile acetic acid, and provide better resolution for separating truncated peptide impurities.
Q: What are the yield implications of this novel purification method?
A: By optimizing the gradient elution profile specifically for the polymer stationary phase, the method achieves a purification yield exceeding 90%, significantly reducing material loss compared to conventional multi-step purification processes that often suffer from lower recovery rates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Desmopressin Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the purity of peptide APIs like Desmopressin is non-negotiable for patient safety and regulatory approval. Our technical team has extensively analyzed advanced purification routes, including the polymer chromatography method described in CN103467574A, to ensure our manufacturing capabilities align with the highest industry standards. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, utilizing state-of-the-art preparative HPLC systems and rigorous QC labs to guarantee stringent purity specifications. Our commitment to quality ensures that every batch of Desmopressin Acetate we supply is free from critical impurities like des-Gln4, delivering the reliability that global pharmaceutical partners demand.
We invite you to collaborate with us to optimize your supply chain for this critical hormone therapeutic. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our advanced purification capabilities can reduce your total cost of ownership. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our engineering excellence can support your long-term commercial goals.
