Advanced Synthesis of Eldecalcitol 20S Isomer for Pharmaceutical Impurity Standards
Advanced Synthesis of Eldecalcitol 20S Isomer for Pharmaceutical Impurity Standards
The pharmaceutical industry faces rigorous challenges in ensuring the purity and safety of active pharmaceutical ingredients (APIs), particularly for complex molecules like Vitamin D analogs used in osteoporosis treatment. Patent CN114656384A introduces a groundbreaking preparation method for the Eldecalcitol 20S isomer, a critical process-related impurity that must be strictly monitored and quantified. As Eldecalcitol gains traction as a superior treatment for improving bone metabolism and calcium absorption, the demand for high-purity reference standards for its impurities has surged. This new technical disclosure offers a robust, three-step synthetic route that addresses the historical bottlenecks of low yield and operational complexity, providing a viable pathway for reliable pharmaceutical intermediate supplier networks to produce essential quality control materials efficiently.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of the Eldecalcitol 20S isomer was fraught with significant technical and economic hurdles. Existing literature, such as the method reported in Chemiform in 2005, relied on convoluted reaction sequences that were not only operationally demanding but also resulted in substantial formation of byproducts. These conventional routes often necessitated the use of highly toxic reagents, posing severe environmental and safety risks in a manufacturing setting. Furthermore, the purification of the target isomer from the complex reaction mixture was notoriously difficult, leading to poor isolation yields and inconsistent product quality. For procurement managers and supply chain heads, these factors translated into unreliable sourcing, extended lead times for high-purity pharmaceutical intermediates, and inflated costs due to excessive raw material consumption and waste disposal requirements.
The Novel Approach
In stark contrast, the methodology outlined in the patent utilizes a streamlined convergent strategy that dramatically simplifies the production landscape. The core of this innovation lies in a concise three-step sequence: selective protection, stereoselective coupling, and global deprotection. By employing a silyl-based protecting group strategy on the CD-ring precursor, the process ensures high chemoselectivity before the critical bond-forming event. The subsequent coupling with the A-ring phosphine oxide proceeds with excellent stereocontrol, minimizing the generation of unwanted isomers. This approach not only boosts the total yield to over 65 percent but also utilizes common organic solvents and reagents that are easier to handle and dispose of. 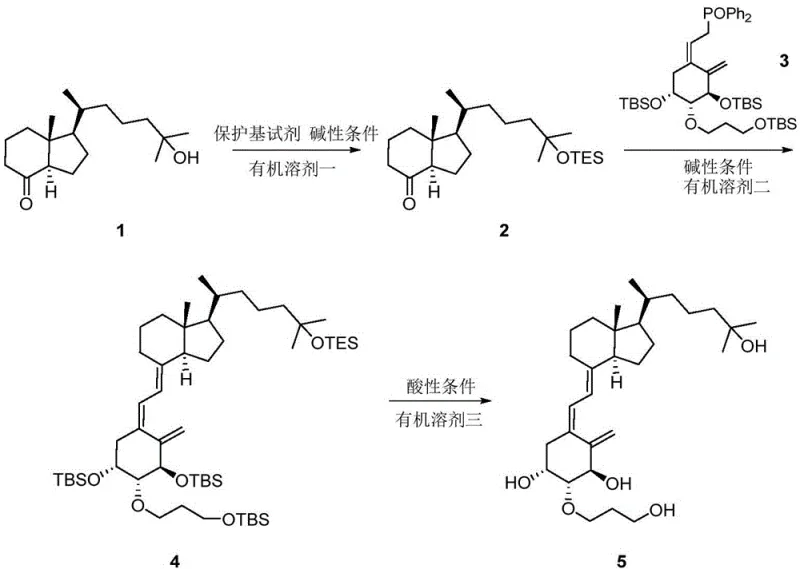
Mechanistic Insights into Silyl Protection and Coupling Strategy
The success of this synthesis hinges on the precise execution of the coupling reaction between the protected CD-ring fragment and the A-ring phosphine oxide. In the second step, the protected Compound 2 reacts with Compound 3 under strongly alkaline conditions, typically utilizing n-butyllithium as the base in tetrahydrofuran at low temperatures around minus 60 degrees Celsius. This generates a reactive carbanion species that attacks the carbonyl equivalent of the phosphine oxide, forging the crucial carbon-carbon double bond that links the A and CD rings of the secosteroid structure. The choice of n-BuLi over other bases like tert-butyllithium is critical, as experimental data indicates a marked improvement in yield, reaching approximately 80 percent compared to significantly lower efficiencies with alternative bases. 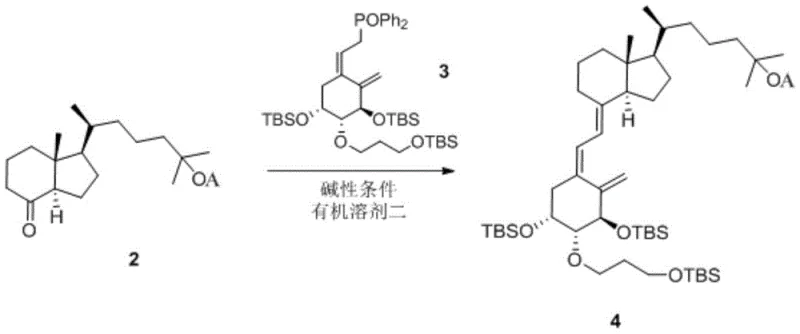
Impurity control is inherently built into the design of this route through the use of robust protecting groups such as triethylsilyl (TES) or tert-butyldiphenylsilyl (TBDPS). These groups effectively mask the hydroxyl functionality on the side chain during the harsh coupling conditions, preventing side reactions that could lead to degradation or polymerization. The final deprotection step, achieved using acidic conditions with reagents like hydrofluoric acid or sulfuric acid, cleanly removes these silyl groups to reveal the triol structure of the final Eldecalcitol 20S isomer. This orthogonal protection strategy ensures that the final product possesses the correct stereochemistry and functional group arrangement required for it to serve as a valid reference standard in analytical testing.
How to Synthesize Eldecalcitol 20S Isomer Efficiently
The synthesis protocol detailed in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing reproducibility and ease of execution. The process begins with the protection of the starting material, followed by the key coupling event, and concludes with a straightforward deprotection workup. Each step has been optimized for solvent choice, temperature, and stoichiometry to maximize throughput while maintaining safety. For technical teams looking to implement this route, the following guide outlines the standardized operational parameters derived directly from the patent examples, ensuring consistent results across different batches.
- Protect the hydroxyl group of the CD-ring precursor (Compound 1) using a silyl chloride reagent like TESCl or TBDPSCl under alkaline conditions to form Compound 2.
- Perform a coupling reaction between the protected CD-ring (Compound 2) and the A-ring phosphine oxide (Compound 3) using a strong base like n-BuLi at low temperature to generate Compound 4.
- Deprotect the silyl groups on Compound 4 using an acidic reagent such as hydrofluoric acid in a mixed solvent system to obtain the final Eldecalcitol 20S isomer (Compound 5).
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, this new synthesis route represents a significant strategic advantage over legacy methods. By shortening the synthetic sequence and improving overall yields, the method drastically reduces the consumption of expensive starting materials and solvents. The elimination of highly toxic reagents simplifies the environmental compliance burden, reducing the costs associated with hazardous waste treatment and disposal. Furthermore, the use of standard reaction conditions and readily available reagents enhances the scalability of the process, allowing manufacturers to respond quickly to fluctuating market demands for impurity standards without requiring specialized or exotic equipment.
- Cost Reduction in Manufacturing: The substantial increase in total yield directly correlates to a reduction in the cost of goods sold. By avoiding the low-yielding steps and difficult purifications characteristic of older methods, manufacturers can achieve significant cost savings in API intermediate manufacturing. The ability to use cheaper protecting group reagents like TESCl instead of more cumbersome alternatives further drives down raw material expenses, making the production of this critical impurity standard economically viable for broader distribution.
- Enhanced Supply Chain Reliability: The reliance on common chemical reagents and solvents such as dichloromethane, tetrahydrofuran, and triethylamine ensures that supply chain disruptions are minimized. Unlike processes dependent on scarce or highly regulated specialty chemicals, this route can be executed in most standard fine chemical facilities. This flexibility allows for diversified sourcing strategies and reduces the risk of production stoppages, ensuring a steady flow of high-purity vitamin D analogs to downstream quality control laboratories.
- Scalability and Environmental Compliance: The simplified workup procedures, which primarily involve aqueous washes and standard column chromatography, are easily adaptable to larger reactor volumes. The absence of complex distillation steps or cryogenic requirements beyond standard low-temperature stirring facilitates a smoother transition from gram-scale development to kilogram-scale commercial production. Additionally, the reduced toxicity profile of the reagents aligns with modern green chemistry principles, helping companies meet increasingly stringent environmental regulations without compromising on output quality.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for making informed procurement decisions. The following questions address common concerns regarding the implementation and benefits of this patented method, drawing directly from the experimental data and comparative analysis provided in the documentation. These insights are designed to clarify the operational advantages and quality assurances associated with this new production route.
Q: What is the total yield of the new Eldecalcitol 20S isomer synthesis route?
A: The novel synthesis method described in patent CN114656384A achieves a total yield of over 65 percent, which is significantly higher than prior art methods that suffer from low yields and difficult purification.
Q: Why is the 20S isomer important for Eldecalcitol drug development?
A: The 20S isomer is a specific process impurity introduced during the convergent synthesis of Eldecalcitol. Regulatory agencies require validated impurity standards to establish strict quality control limits for the final drug substance.
Q: Does this method avoid the use of highly toxic reagents?
A: Yes, compared to previous methods reported in literature which utilized highly toxic reagents and complicated operations, this new route employs standard silyl protection and coupling chemistry with simpler workup procedures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Eldecalcitol 20S Isomer Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality impurity standards play in the drug development lifecycle. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that even complex molecules like the Eldecalcitol 20S isomer are manufactured with precision and consistency. We adhere to stringent purity specifications and utilize rigorous QC labs to verify the identity and purity of every batch, guaranteeing that our products meet the exacting standards required by global regulatory bodies for pharmaceutical applications.
We invite you to collaborate with us to optimize your supply chain for Vitamin D analog intermediates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced synthesis capabilities can support your R&D and quality control objectives effectively.
