Advanced Zinc-Mediated Reduction for Scalable Tandospirone Intermediate Production
Advanced Zinc-Mediated Reduction for Scalable Tandospirone Intermediate Production
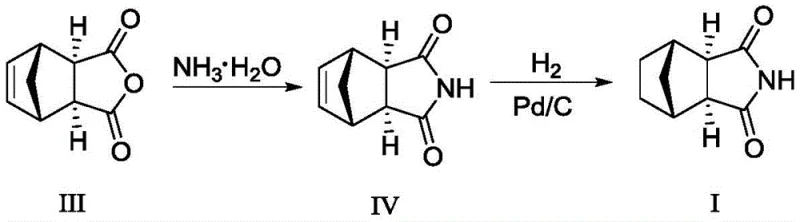
The pharmaceutical industry continuously seeks robust synthetic routes for critical psychotropic agents, particularly for anxiolytics like Tandospirone. A pivotal breakthrough in this domain is documented in Chinese patent CN114685349A, which details a novel preparation method for cis-exo-bicyclo[2.2.1]heptane-2,3-dicarboximide, a key intermediate designated as Formula I. This compound serves as the structural backbone for Tandospirone Citrate, a drug renowned for its selective action on 5-HT1A receptors and favorable safety profile. The significance of this patent lies not merely in the synthesis of the molecule itself, but in the strategic overhaul of the reduction step, transitioning away from hazardous high-pressure hydrogenation towards a safer, metal-mediated reduction protocol. For R&D directors and procurement specialists, understanding this shift is crucial for securing a reliable pharmaceutical intermediate supplier capable of delivering materials that meet the stringent purity standards of the 2020 edition of the Chinese Pharmacopoeia.
![Chemical structure of cis-exo-bicyclo[2.2.1]heptane-2,3-dicarboximide (Formula I)](/insights/img/cis-exo-bicyclo-heptane-dicarboximide-zinc-reduction-pharma-supplier-20260307030418-01.png)
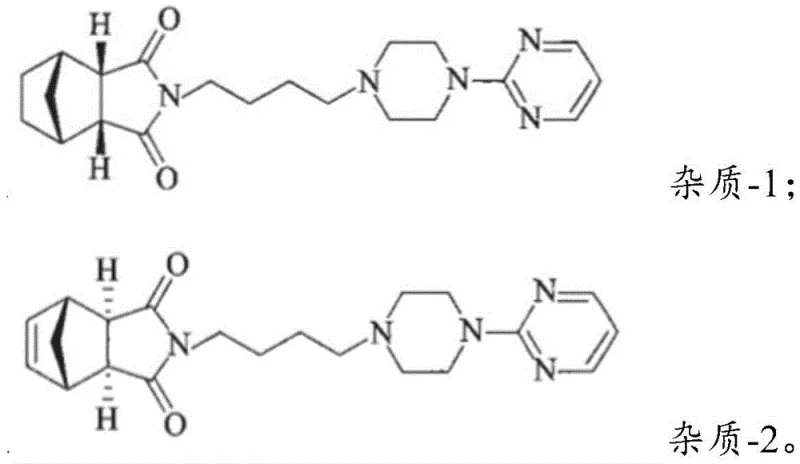
The structural integrity of Formula I is paramount, as any deviation in stereochemistry or the presence of unsaturated impurities can propagate through the synthesis of the final API. The patent highlights that the quality of this intermediate directly dictates the impurity profile of Tandospirone Citrate, specifically regarding Impurity-1 and Impurity-2. Consequently, the development of a manufacturing process that inherently suppresses these contaminants represents a significant value add for the entire supply chain, ensuring that downstream processing remains efficient and compliant with global regulatory expectations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this bicyclic imide has relied heavily on catalytic hydrogenation using palladium on carbon (Pd/C), a method fraught with operational and safety challenges. As illustrated in prior art routes, such as those disclosed in CN102863437A, the process typically involves reducing the olefinic precursor (Formula IV) under hydrogen pressure. This approach necessitates the use of specialized high-pressure reactors, which significantly increases capital expenditure and operational complexity. Moreover, hydrogen gas is inherently flammable and explosive, introducing severe safety liabilities in large-scale manufacturing environments. Beyond safety, the chemical efficacy of Pd/C hydrogenation is often inconsistent; literature and comparative data indicate that this method frequently results in incomplete conversion, leaving behind significant amounts of unreacted starting material (Impurity B).
Furthermore, conventional recrystallization methods utilizing organic solvents like toluene or ethyl acetate have proven inadequate for removing stereoisomeric impurities, specifically the endo-isomer (Impurity A). Data from comparative examples in the patent reveal that even with optimization, Pd/C routes struggle to reduce Impurity A below 0.17% and often leave detectable levels of Impurity B. These residual impurities are not merely cosmetic defects; they react in subsequent condensation steps to form Impurity-1 and Impurity-2 in the final drug substance, potentially causing the batch to fail pharmacopoeial standards. This creates a bottleneck where extensive purification is required, driving up costs and extending lead times for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast to the hazardous hydrogenation protocols, the method disclosed in CN114685349A employs a zinc-mediated reduction system catalyzed by nickel salts. This innovative route operates under atmospheric pressure, completely eliminating the need for flammable hydrogen gas and high-pressure containment vessels. The reaction utilizes a mixed solvent system of water and an organic co-solvent (such as tetrahydrofuran or isopropanol), which facilitates the interaction between the solid zinc reducing agent and the dissolved substrate. This shift in chemistry fundamentally alters the risk profile of the manufacturing process, making it significantly more suitable for industrial mass production. By replacing a precious metal catalyst with abundant base metals, the process also decouples production costs from the volatile market prices of palladium.
The true brilliance of this novel approach, however, lies in its integration with a specific aqueous crystallization workup. Unlike prior art that relies on organic solvents for purification, this method leverages water as the primary crystallization medium. The patent data demonstrates that cooling the aqueous solution to between 0°C and 15°C results in the precipitation of the target exo-isomer with exceptional purity. This step is critical because it exploits the differential solubility of the impurities in water, effectively washing away both the endo-isomer (Impurity A) and any trace unreacted olefin (Impurity B). The result is a product with purity exceeding 99.70%, where both critical impurities are undetectable, thereby streamlining the path to commercial scale-up of complex heterocyclic intermediates.
Mechanistic Insights into Zinc-Nickel Mediated Reduction
The core of this technological advancement is the synergistic interaction between zinc powder and nickel salts (such as nickel chloride hexahydrate or nickel bromide) in a protic environment. Mechanistically, the zinc acts as the stoichiometric electron donor, while the nickel salt is likely reduced in situ to form active nickel species on the zinc surface, creating a highly reactive couple similar to a Raney-type system but generated under milder conditions. This active surface facilitates the transfer of electrons to the olefinic double bond of Formula IV, effecting reduction without the need for external hydrogen gas. The presence of water in the solvent mixture is not merely incidental; it plays a dual role as a proton source for the reduction and as a modulator of the reaction kinetics, preventing overly vigorous exotherms that could compromise stereocontrol. The molar ratio of zinc to substrate is maintained between 3:1 and 5:1, ensuring a substantial excess of reducing power to drive the reaction to full conversion, which is the primary defense against the formation of Impurity B.
Controlling the stereochemical outcome to favor the exo-isomer over the endo-isomer (Impurity A) is equally critical. The reaction conditions, specifically the reflux temperature in a THF/water or Isopropanol/water mixture, appear to favor the thermodynamic stability of the exo-configuration or kinetically prevent isomerization. However, the most robust mechanism for impurity control is the downstream crystallization. The patent explicitly contrasts this with organic solvent crystallization, noting that toluene fails to remove Impurity A and ethyl acetate fails to remove Impurity B effectively. Water, conversely, acts as a highly selective anti-solvent. Upon cooling, the lattice energy of the pure exo-isomer drives its precipitation, while the more soluble endo-isomer and polar impurities remain in the mother liquor. This physical separation mechanism ensures that even if trace amounts of Impurity A are formed during reduction, they do not contaminate the final isolated solid, thus preventing the formation of Impurity-1 and Impurity-2 in the final Tandospirone Citrate API.
How to Synthesize Cis-Exo-Bicyclo[2.2.1]heptane-2,3-dicarboximide Efficiently
Implementing this synthesis requires precise adherence to the solvent ratios and thermal profiles outlined in the patent to maximize yield and purity. The process begins with the preparation of the reaction slurry, where the choice of co-solvent (THF or Isopropanol) influences the solubility of the starting material, while the water content is critical for the reduction mechanism. Following the reduction, the workup involves a liquid-liquid extraction with dichloromethane to separate the organic product from inorganic zinc salts, followed by concentration. The final crystallization step is the gatekeeper of quality; dissolving the concentrate in hot water and controlling the cooling rate to 0-15°C is essential for obtaining the desired crystal habit and purity. For detailed operational parameters and specific molar quantities, please refer to the standardized synthesis guide below.
- Mix compound IV with a mixed solvent of tetrahydrofuran or isopropanol and water, adding zinc powder and a nickel salt catalyst.
- Heat the mixture to reflux for 6 to 13 hours to ensure complete reduction of the double bond.
- Filter the reaction mixture, concentrate the filtrate, extract with dichloromethane, and concentrate to obtain a crude concentrate.
- Dissolve the concentrate in water, heat to dissolve completely, then cool to 0-15°C to crystallize the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from Pd/C hydrogenation to zinc-nickel reduction offers profound strategic benefits beyond simple chemistry. The elimination of high-pressure hydrogenation removes a major bottleneck in facility utilization, allowing production to occur in standard glass-lined or stainless steel reactors without the need for specialized autoclaves. This flexibility significantly enhances supply chain reliability, as production can be easily transferred between different manufacturing sites without extensive requalification of high-pressure equipment. Furthermore, the removal of pyrophoric Pd/C catalysts simplifies waste handling and reduces the environmental compliance burden associated with heavy metal disposal, aligning with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The economic implications of this process change are substantial. By replacing palladium, a precious metal with high and volatile market pricing, with zinc and nickel salts, the direct material cost of the reduction step is drastically simplified. Additionally, the avoidance of high-pressure equipment reduces both capital depreciation costs and maintenance expenses. The high yield and purity achieved in a single crystallization step minimize the need for reprocessing or chromatographic purification, leading to substantial cost savings in labor and solvent consumption. This efficiency translates directly into a more competitive pricing structure for the final intermediate.
- Enhanced Supply Chain Reliability: Relying on hydrogen gas and palladium catalysts introduces specific supply chain vulnerabilities, including gas delivery logistics and precious metal availability. The new method utilizes commodity chemicals—zinc powder and nickel salts—which are widely available from multiple global suppliers, reducing the risk of raw material shortages. Moreover, the safety profile of the process reduces the likelihood of unplanned shutdowns due to safety incidents, ensuring consistent delivery schedules. This stability is crucial for reducing lead time for high-purity pharmaceutical intermediates, allowing downstream API manufacturers to plan their production with greater confidence.
- Scalability and Environmental Compliance: Scaling a high-pressure hydrogenation reaction from the lab to multi-ton production involves significant engineering challenges and safety reviews. In contrast, the zinc-mediated reduction described in this patent is inherently scalable, operating at atmospheric pressure and reflux temperatures. This simplicity accelerates technology transfer and commercial scale-up of complex heterocyclic intermediates. From an environmental perspective, the process generates less hazardous waste; the absence of spent palladium catalyst simplifies effluent treatment, and the use of water as a crystallization solvent reduces the volume of organic waste streams, supporting sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this novel synthesis route. Understanding these details is vital for quality assurance teams evaluating the suitability of this intermediate for GMP production. The answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, ensuring accuracy and relevance to real-world manufacturing scenarios.
Q: Why is the zinc-nickel reduction method preferred over traditional palladium-carbon hydrogenation?
A: Traditional Pd/C hydrogenation requires high-pressure equipment and flammable hydrogen gas, posing significant safety risks. Furthermore, Pd/C often leaves unreacted starting material (Impurity B) and fails to adequately remove endo-isomers (Impurity A). The zinc-nickel method operates at atmospheric pressure, eliminates explosion hazards, and achieves superior impurity profiles through optimized aqueous crystallization.
Q: How does this process control Impurity A and Impurity B levels?
A: Impurity B (unreacted olefin) is minimized by the high efficiency of the zinc-nickel reduction system which drives the reaction to completion. Impurity A (endo-isomer) and residual Impurity B are effectively removed during the final crystallization step using water as the solvent, leveraging specific solubility differences that organic solvents like toluene or ethyl acetate cannot achieve.
Q: What are the optimal solvent systems for this reduction?
A: The patent identifies mixed solvent systems comprising an ether or alcohol solvent (such as tetrahydrofuran or isopropanol) combined with water. A volume ratio of organic solvent to water between 2:1 and 5:1 is critical for balancing reaction kinetics and subsequent workup efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cis-Exo-Bicyclo[2.2.1]heptane-2,3-dicarboximide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of the starting material defines the success of the final drug product. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated zinc-nickel reduction technology is implemented with precision. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of cis-exo-bicyclo[2.2.1]heptane-2,3-dicarboximide meets the undetectable impurity standards required for Tandospirone synthesis. We understand the critical nature of this intermediate in the psychotropic drug supply chain and are committed to maintaining uninterrupted availability.
We invite pharmaceutical partners to leverage our technical expertise to optimize their supply chains. By choosing our advanced manufacturing route, you gain access to a safer, more cost-effective, and higher-purity intermediate. We encourage you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Let us provide you with specific COA data and route feasibility assessments to demonstrate how our process can enhance the efficiency and compliance of your Tandospirone production.
