Revolutionizing Indanone Production: A Green Aqueous Phase Synthesis Strategy for Commercial Scale
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex carbocyclic scaffolds, particularly indanone derivatives which serve as critical building blocks for bioactive molecules. Patent CN114560761B discloses a groundbreaking method for the one-time synthesis of 2,3-disubstituted indanone derivatives directly in an aqueous phase, representing a significant leap forward in green chemistry and process intensification. This technology utilizes a rhodium catalyst system to facilitate the coupling of internal alkynes with 2-formyl phenylboronic acid, bypassing the need for hazardous organic solvents that have traditionally plagued this transformation. The ability to conduct this reaction in water under mild conditions not only enhances safety profiles but also drastically simplifies the downstream processing required to isolate high-purity intermediates. For R&D directors and procurement specialists, this patent offers a tangible route to reduce the environmental footprint of API intermediate manufacturing while maintaining rigorous quality standards. The method demonstrates exceptional versatility across various substrate scopes, proving that sustainability and high performance are not mutually exclusive in modern chemical synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the indanone skeleton has relied heavily on intramolecular hydroacylation or transition metal-catalyzed carbocyclization strategies that impose significant operational burdens on manufacturing facilities. Conventional approaches often necessitate the use of hyperstoichiometric reducing agents and high-temperature conditions that drive up energy consumption and increase the risk of thermal runaway incidents in large-scale reactors. Furthermore, many established protocols depend on the presence of exogenous bisphosphine ligands and anhydrous organic solvents, which introduce complex purification challenges and generate substantial volumes of hazardous waste. The requirement for multi-step precursor preparation prior to the actual cyclization event further elongates the production timeline, leading to increased lead times and higher overall operational expenditures. These limitations create bottlenecks in the supply chain, making it difficult to scale production efficiently without compromising on cost or environmental compliance. Consequently, there is an urgent industry demand for a streamlined process that eliminates these inefficiencies while delivering consistent product quality.
The Novel Approach
The methodology outlined in patent CN114560761B introduces a paradigm shift by enabling the direct synthesis of 2,3-disubstituted indanone derivatives in a single operational step using water as the sole reaction medium. This novel approach leverages a specific rhodium catalyst system that functions effectively without the addition of external ligands, thereby removing a major cost driver and potential source of metal contamination from the process flow. By operating at mild temperatures ranging from 50°C to 55°C, the reaction minimizes energy input and enhances safety parameters, making it ideally suited for commercial scale-up of complex pharmaceutical intermediates. The use of water not only reduces toxicity concerns for operators but also simplifies the work-up procedure, as the product can often be isolated through straightforward extraction or filtration techniques. This streamlined workflow significantly reduces the time required for process development and validation, allowing manufacturers to bring critical intermediates to market faster. The robustness of this aqueous phase system ensures that high conversion rates are maintained even with diverse substrate inputs, providing a reliable foundation for continuous manufacturing improvements.
Mechanistic Insights into Rhodium-Catalyzed Cyclization
The core of this technological advancement lies in the unique catalytic cycle facilitated by the [RhCl(cod)]2 complex in an aqueous environment, which promotes the activation of C-H bonds and subsequent cyclization with remarkable efficiency. In this mechanism, the rhodium center coordinates with the internal alkyne and the aldehyde functionality of the boronic acid derivative, initiating a cascade that constructs the five-membered ring system without requiring harsh activation conditions. The absence of exogenous ligands suggests that the water molecules themselves or the amine base may play a crucial role in stabilizing the active catalytic species, preventing catalyst deactivation and ensuring prolonged turnover numbers. This ligand-free environment is particularly advantageous for pharmaceutical applications, as it eliminates the need for rigorous metal scavenging steps that are typically required to meet stringent residual metal specifications in drug substances. The reaction proceeds through a well-defined oxidative addition and reductive elimination pathway, driven by the thermodynamic stability of the resulting indanone scaffold. Understanding this mechanism allows process chemists to fine-tune reaction parameters to maximize yield and minimize the formation of regioisomeric impurities.
Impurity control is a critical aspect of this synthesis, as the presence of side products can complicate purification and impact the overall yield of the desired 2,3-disubstituted indanone derivatives. The aqueous phase conditions inherently suppress certain side reactions that are common in organic solvents, such as polymerization of the alkyne or decomposition of the boronic acid species. The use of triethylamine as a base further aids in maintaining the pH balance necessary for the catalytic cycle to proceed smoothly, while also neutralizing any acidic byproducts that might form during the reaction. The high selectivity observed in this method, with isolated yields reaching up to 98% in optimized examples, indicates a highly controlled reaction environment where the formation of undesired byproducts is minimized. This level of purity is essential for downstream applications in drug discovery, where the presence of trace impurities can alter biological activity or toxicity profiles. The ability to store the final products at room temperature without significant degradation further underscores the stability and robustness of the chemical structure generated through this pathway.
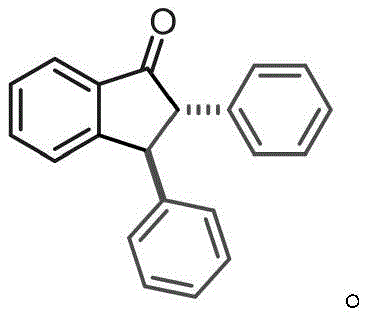
How to Synthesize 2,3-Disubstituted Indanone Derivatives Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry of reagents and the maintenance of anaerobic conditions to ensure the longevity of the rhodium catalyst. The standard protocol involves mixing the rhodium catalyst, triethylamine, internal alkyne, and 2-formyl phenylboronic acid in water, followed by stirring at a controlled temperature of 50°C to 55°C for approximately 16 to 18 hours. It is crucial to exclude oxygen from the reaction vessel, typically by purging with nitrogen, to prevent oxidation of the catalyst or the substrates which could lead to reduced conversion rates. The molar ratio of the internal alkyne to the boronic acid is typically maintained at 1:1.5 to drive the reaction to completion, while the catalyst loading is kept low at around 5 to 6 mol% to optimize cost efficiency. Detailed standardized synthesis steps see the guide below for specific operational parameters and safety precautions required for handling rhodium complexes and boronic acids in a production setting.
- Prepare the reaction mixture by combining the rhodium catalyst [RhCl(cod)]2, triethylamine, internal alkyne, and 2-formyl phenylboronic acid in water under anaerobic conditions.
- Maintain the reaction temperature between 50°C and 55°C while stirring continuously for a duration of 16 to 18 hours to ensure complete conversion.
- Isolate the final 2,3-disubstituted indanone derivative product, which can be stored at room temperature, achieving high yields without exogenous ligands.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this aqueous synthesis method offers profound advantages that directly address the pain points of cost, supply reliability, and environmental compliance faced by procurement and supply chain leaders. The elimination of expensive organic solvents and exogenous ligands results in a drastic reduction in raw material costs, while the simplified work-up procedure lowers labor and utility expenses associated with solvent recovery and waste treatment. The mild reaction conditions reduce the energy load on manufacturing facilities, contributing to lower operational expenditures and a smaller carbon footprint, which is increasingly important for meeting corporate sustainability goals. Furthermore, the use of water as a solvent enhances process safety, reducing the risk of fire and explosion hazards that are common with volatile organic compounds, thereby lowering insurance and compliance costs. These factors combine to create a highly competitive cost structure that allows suppliers to offer high-purity intermediates at more attractive price points without sacrificing quality. The robustness of the process ensures consistent supply continuity, mitigating the risk of production delays that can disrupt downstream drug manufacturing schedules.
- Cost Reduction in Manufacturing: The removal of exogenous ligands and organic solvents from the process flow eliminates significant cost centers associated with raw material procurement and hazardous waste disposal. By utilizing water as the primary medium, manufacturers avoid the volatile pricing and supply chain constraints often linked to specialty organic solvents, leading to substantial cost savings over the lifecycle of the product. Additionally, the ligand-free nature of the catalyst system removes the need for expensive purification steps to remove phosphine residues, further streamlining the production budget. This economic efficiency allows for more competitive pricing strategies in the global market for pharmaceutical intermediates, enhancing the value proposition for partners seeking to optimize their cost of goods sold. The overall reduction in process complexity translates directly into lower capital expenditure requirements for new production lines.
- Enhanced Supply Chain Reliability: The reliance on readily available and stable reagents such as water and triethylamine ensures that the supply chain is less vulnerable to disruptions caused by the scarcity of specialty chemicals. The mild operating conditions reduce the strain on equipment, leading to longer asset life and fewer unplanned maintenance shutdowns that can impact delivery schedules. This stability is crucial for maintaining just-in-time inventory levels and ensuring that critical API intermediates are available when needed for clinical or commercial production runs. The ability to scale this reaction from laboratory to commercial quantities without significant re-optimization provides a predictable timeline for technology transfer, reducing the time to market for new drug candidates. Partners can rely on a consistent supply of high-quality materials, fostering long-term strategic relationships built on trust and performance.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this method, such as the use of water and low energy consumption, align perfectly with increasingly stringent environmental regulations globally. Scaling up an aqueous process is inherently safer and more straightforward than managing large volumes of flammable organic solvents, reducing the regulatory burden and permitting timelines for new facilities. The low toxicity of the reagents and the absence of heavy metal ligands simplify the handling of effluents, making it easier to meet discharge standards and avoid environmental fines. This compliance advantage future-proofs the manufacturing process against tightening environmental laws, ensuring long-term operational viability. Companies adopting this technology can leverage their sustainability achievements for marketing and brand differentiation, appealing to eco-conscious stakeholders and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aqueous phase synthesis technology for indanone derivatives. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing manufacturing portfolios. The clarity provided here aims to reduce uncertainty and accelerate the adoption of this innovative synthetic route. For further technical discussions, our team is available to provide deeper insights into specific substrate compatibilities and process optimization strategies.
Q: What are the primary advantages of using water as a solvent for indanone synthesis?
A: Using water eliminates the need for volatile organic compounds, significantly reducing environmental impact and solvent disposal costs while maintaining high reaction efficiency and product conversion rates.
Q: Does this rhodium-catalyzed method require expensive phosphine ligands?
A: No, the patented method operates effectively without exogenous ligands, which simplifies the purification process and reduces raw material costs associated with ligand removal.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the mild reaction conditions (50-55°C) and use of water make the process highly scalable and safe for industrial manufacturing, ensuring supply chain reliability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Disubstituted Indanone Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the aqueous phase synthesis route described in patent CN114560761B for producing high-value pharmaceutical intermediates. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that this innovative chemistry can be seamlessly transitioned from the laboratory to the manufacturing plant. Our facilities are equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch of 2,3-disubstituted indanone derivatives meets the highest industry standards for quality and consistency. We understand the critical nature of supply chain continuity for our partners and are committed to delivering reliable volumes of these key intermediates to support your drug development pipelines. Our technical team is ready to collaborate with you to optimize this process for your specific needs, ensuring maximum efficiency and yield.
We invite you to engage with our technical procurement team to discuss how this technology can drive value for your organization through a Customized Cost-Saving Analysis. By partnering with us, you gain access to specific COA data and route feasibility assessments that will help you make informed decisions about your sourcing strategy. Let us help you reduce lead time for high-purity indanone derivatives and secure a stable supply of this critical building block for your upcoming projects. Contact us today to initiate a conversation about optimizing your supply chain with our advanced synthesis capabilities. We are dedicated to being your trusted partner in navigating the complexities of fine chemical manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
