Revolutionizing Epsilon-Caprolactone Manufacturing via Efficient Oxygen Oxidation Co-production
Revolutionizing Epsilon-Caprolactone Manufacturing via Efficient Oxygen Oxidation Co-production
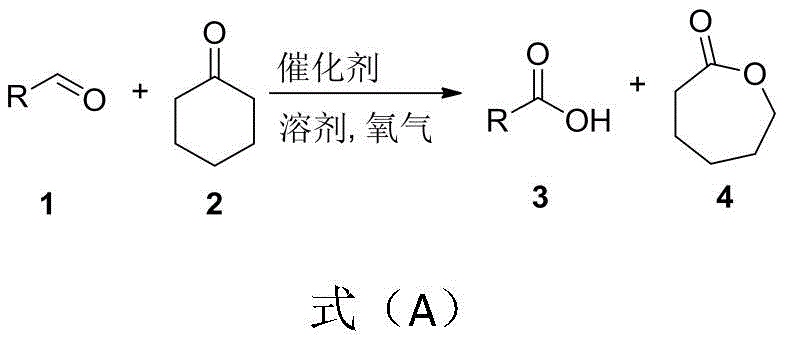
The chemical industry is constantly seeking sustainable pathways to produce high-value intermediates, and the recent disclosure in patent CN112479860B presents a transformative approach to synthesizing epsilon-caprolactone. This intellectual property details a novel method for the co-production of carboxylic acids and epsilon-caprolactone based on efficient oxygen oxidation. By leveraging inexpensive iron-based catalysts and molecular oxygen, this technology addresses critical pain points in traditional Baeyer-Villiger oxidation, such as high reagent costs and safety hazards associated with peracids. For R&D directors and procurement managers alike, this represents a significant opportunity to optimize synthetic routes for biodegradable polymers and pharmaceutical intermediates. The ability to generate two valuable products simultaneously from simple starting materials like cyclohexanone and aldehydes underscores the economic and environmental potential of this innovation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of epsilon-caprolactone has relied heavily on the Baeyer-Villiger oxidation of cyclohexanone using organic peracids such as peracetic acid or m-chloroperbenzoic acid (m-CPBA). While effective, these traditional pathways suffer from severe drawbacks that impact both cost and safety profiles. Organic peracids are inherently unstable and pose significant explosion risks during storage and transport, necessitating stringent safety protocols that drive up operational expenditures. Furthermore, these reagents are stoichiometric oxidants, meaning they are consumed in equimolar amounts relative to the substrate, generating substantial quantities of acidic waste by-products that require complex neutralization and disposal procedures. Alternatively, hydrogen peroxide-based processes have been developed to mitigate some safety concerns, yet they often struggle with lower reactivity requiring highly active catalysts, and the presence of water can lead to undesirable hydrolysis of the lactone product. These limitations create a pressing need for a cleaner, more atom-economical oxidation strategy.
The Novel Approach
The methodology described in CN112479860B offers a compelling solution by utilizing molecular oxygen as the terminal oxidant in the presence of a catalyst. This approach fundamentally shifts the paradigm from stoichiometric oxidation to catalytic aerobic oxidation. In this system, an aldehyde is oxidized in situ to form a peroxy acid intermediate, which subsequently reacts with cyclohexanone to yield epsilon-caprolactone and a carboxylic acid. This dual-output mechanism not only maximizes atom economy but also converts the aldehyde, often considered a sacrificial reductant in other contexts, into a valuable co-product. The reaction proceeds under remarkably mild conditions, typically between 30°C and 50°C, eliminating the need for energy-intensive heating. By replacing hazardous peracids with air or oxygen, the process drastically reduces the safety footprint and waste generation, aligning perfectly with modern green chemistry principles and regulatory demands for sustainable manufacturing.
Mechanistic Insights into Fe-Catalyzed Aerobic Oxidation
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to implement this technology. The core of the reaction involves the catalytic activation of molecular oxygen to oxidize an aldehyde substrate into a peroxy acid species. This step is facilitated by the iron nitrate catalyst, potentially in synergy with a nitroxyl radical co-catalyst like TEMPO. The generated peroxy acid then acts as the oxygen donor in a classic Baeyer-Villiger rearrangement with cyclohexanone. This involves the formation of a Criegee intermediate, followed by a migration of the alkyl group to the oxygen atom, resulting in ring expansion to form the seven-membered lactone ring of epsilon-caprolactone. Simultaneously, the peroxy acid is reduced to its corresponding carboxylic acid. This tandem process ensures that the oxidizing power of oxygen is efficiently transferred to both substrates, minimizing side reactions and maximizing the yield of both target molecules.
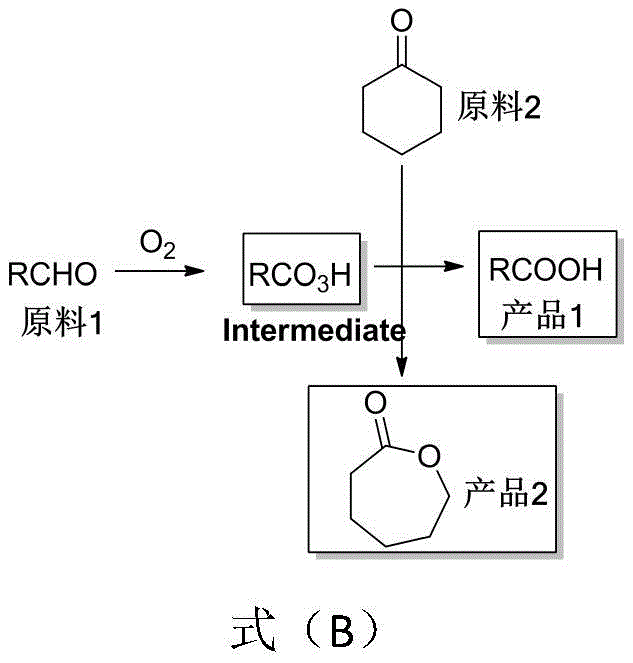
Impurity control is another critical aspect where this mechanism offers distinct advantages. In traditional peracid methods, residual peracids can be difficult to remove and may degrade the product over time. In this aerobic system, the peroxy acid is generated in situ and consumed immediately in the reaction with cyclohexanone, preventing its accumulation. Furthermore, the use of mild temperatures helps suppress thermal degradation pathways that might lead to ring-opening polymerization or other decomposition products. The choice of solvent, such as 1,2-dichloroethane, plays a vital role in solubilizing both the organic substrates and the inorganic catalyst species, ensuring a homogeneous reaction environment that promotes consistent kinetics. The addition of additives like sodium chloride in certain embodiments suggests a potential role in modulating the catalyst's electronic environment or stabilizing intermediate species, further refining the impurity profile of the final crude mixture.
How to Synthesize Epsilon-Caprolactone Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to achieve the high yields reported in the patent data. The process generally involves charging a reactor with the iron catalyst, solvent, and cyclohexanone, followed by oxygen purging to establish the necessary oxidative atmosphere. The aldehyde component is then introduced slowly, often via dropwise addition over several hours, to maintain a steady concentration of the reactive peroxy intermediate and prevent exothermic runaway. Detailed standard operating procedures regarding specific molar ratios, stirring rates, and work-up protocols are essential for reproducibility.
- Prepare the reaction mixture by adding ferric nitrate nonahydrate catalyst, organic solvent (preferably 1,2-dichloroethane), and cyclohexanone into a reactor.
- Purge the system with oxygen gas three times to ensure an aerobic environment, then slowly add the aldehyde solution dropwise over approximately 5 hours.
- Maintain the reaction temperature between 30°C and 50°C for 12 to 24 hours, then isolate products via column chromatography or distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this oxygen oxidation technology translates into tangible strategic benefits beyond mere technical feasibility. The shift from expensive, hazardous stoichiometric oxidants to cheap, abundant molecular oxygen represents a fundamental restructuring of the cost base for epsilon-caprolactone production. By eliminating the need to purchase and store large quantities of peracids or high-concentration hydrogen peroxide, facilities can significantly reduce their raw material expenditure and insurance premiums associated with hazardous chemical storage. Moreover, the co-production model effectively subsidizes the cost of the lactone; the simultaneous generation of valuable carboxylic acids creates an additional revenue stream or internal feedstock, thereby lowering the net production cost of the primary target molecule. This economic efficiency is compounded by the simplified waste management profile, as the process avoids the generation of heavy metal waste or large volumes of acidic aqueous waste typical of older methods.
- Cost Reduction in Manufacturing: The utilization of molecular oxygen as the primary oxidant removes the dependency on costly organic peracids, which are among the most expensive reagents in oxidation chemistry. Since oxygen is derived from air, its marginal cost is negligible compared to stoichiometric oxidants. Additionally, the catalyst system relies on iron nitrate, an abundant and inexpensive base metal salt, rather than precious metals like palladium or platinum. This combination of low-cost oxidant and catalyst, paired with the value-added co-production of carboxylic acids, drives a substantial reduction in the overall cost of goods sold (COGS) for epsilon-caprolactone manufacturing.
- Enhanced Supply Chain Reliability: Relying on specialized peracid suppliers can introduce bottlenecks and lead time vulnerabilities, especially during global supply chain disruptions. In contrast, the inputs for this process—cyclohexanone, simple aldehydes, iron salts, and oxygen—are commodity chemicals with robust, multi-vendor supply chains. This diversification of raw material sources ensures greater continuity of supply and reduces the risk of production stoppages due to reagent shortages. The stability of the catalyst system also allows for longer campaign runs without frequent catalyst replenishment, further stabilizing production schedules and delivery commitments to downstream customers.
- Scalability and Environmental Compliance: The mild reaction conditions (30-50°C) and the absence of highly energetic peracid intermediates make this process inherently safer and easier to scale from pilot plant to commercial tonnage. Safety is a major driver of scalability; avoiding the thermal hazards associated with peracid accumulation allows for larger batch sizes or continuous flow processing. From an environmental perspective, the high atom economy and the use of oxygen minimize the E-factor (mass of waste per mass of product), facilitating compliance with increasingly stringent environmental regulations. This green profile not only reduces waste disposal costs but also enhances the marketability of the final product to eco-conscious consumers in the biodegradable plastics and pharmaceutical sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this co-production technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a reliable foundation for feasibility assessments.
Q: What are the primary advantages of this oxygen oxidation method over traditional Baeyer-Villiger oxidation?
A: Unlike traditional methods using hazardous organic peracids (like m-CPBA) or unstable hydrogen peroxide, this method utilizes molecular oxygen as the terminal oxidant. This significantly reduces safety hazards, eliminates the need for expensive stoichiometric oxidants, and improves atom economy by co-producing valuable carboxylic acids alongside epsilon-caprolactone.
Q: Which catalysts are most effective for this co-production process?
A: The patent identifies inexpensive metal nitrates, specifically ferric nitrate nonahydrate (Fe(NO3)3·9H2O), as the primary catalyst. The addition of a co-catalyst such as TEMPO (2,2,6,6-tetramethylpiperidine-1-oxyl) further enhances yield and selectivity, particularly for aryl aldehydes.
Q: Can this process be scaled for industrial manufacturing of biodegradable polymer precursors?
A: Yes, the process operates under mild conditions (30-50°C) and uses readily available raw materials like cyclohexanone and simple aldehydes. The use of oxygen gas and simple iron catalysts suggests high scalability and environmental compliance, making it suitable for large-scale production of epsilon-caprolactone for biodegradable plastics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Epsilon-Caprolactone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the oxygen oxidation co-production method described in CN112479860B for the global supply of epsilon-caprolactone and carboxylic acids. As a leading CDMO partner, we possess the technical expertise to adapt and optimize this novel catalytic system for your specific production needs. Our facilities are equipped to handle diverse synthetic pathways, ranging from early-stage process development to full-scale commercial manufacturing. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that guarantee every batch meets the highest international standards for pharmaceutical and polymer grade intermediates.
We invite you to explore how this innovative technology can enhance your supply chain resilience and cost structure. Our team is ready to collaborate with you to evaluate the feasibility of this route for your specific portfolio. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are eager to provide specific COA data and route feasibility assessments to demonstrate how we can become your trusted partner in delivering high-quality epsilon-caprolactone and related carboxylic acids.
