Advanced Synthesis of Formoterol Related Substance A for Pharmaceutical Quality Control
Introduction to Formoterol Impurity Control
The pharmaceutical industry relies heavily on the rigorous control of impurities to ensure patient safety, particularly for long-acting beta-agonists like Formoterol used in treating asthma and COPD. Patent CN114213285A introduces a critical advancement in this domain by identifying and providing a robust synthesis for a specific related substance, designated as Compound A. This impurity, structurally distinct from the active pharmaceutical ingredient, was found to possess significant cytotoxicity, thereby mandating its strict monitoring and quantification in final drug formulations. The discovery underscores the necessity for high-purity reference standards that match the exact structural and stereochemical profile of potential process impurities.
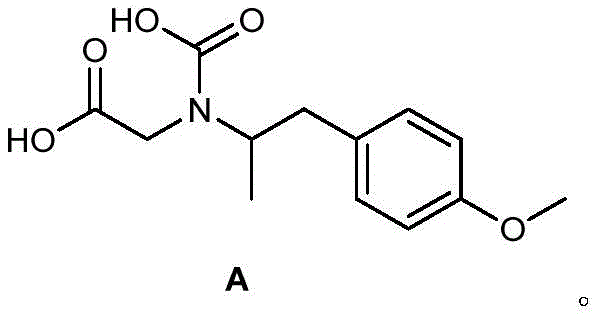
Traditionally, obtaining such specific impurities involved laborious isolation from reaction byproducts, resulting in low yields and inconsistent purity levels that compromised analytical accuracy. The novel approach detailed in the patent circumvents these issues by establishing a dedicated synthetic pathway. This method not only secures a reliable supply of the reference standard but also enhances the overall quality control framework for Formoterol manufacturing. By integrating this standard into HPLC or TLC monitoring protocols, manufacturers can effectively detect and limit Compound A to safe thresholds, aligning with stringent pharmacopoeial requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the identification and quantification of trace impurities in complex pharmaceutical syntheses have been plagued by significant technical hurdles. Conventional methods often relied on the fortuitous formation of impurities during the main API synthesis, followed by difficult chromatographic separation. This approach is inherently inefficient, as the impurities are generated in minute quantities, making isolation costly and time-consuming. Furthermore, the structural confirmation of these isolated traces is often ambiguous without authentic samples, leading to potential gaps in quality assurance. The reliance on unpredictable byproduct formation means that supply chains for critical reference standards are fragile and unable to meet the consistent demand of global regulatory compliance.
The Novel Approach
The methodology presented in the patent represents a paradigm shift towards proactive impurity management through targeted synthesis. Instead of scavenging for trace amounts, the inventors designed a concise three-step route specifically to construct Compound A from readily available starting materials. This deliberate synthesis ensures that the reference standard is produced with high fidelity to the target structure, including the correct stereochemistry at the chiral center. The process utilizes mild reaction conditions and common reagents, which drastically simplifies the operational complexity compared to traditional isolation techniques. Consequently, this approach guarantees a steady availability of high-purity material, empowering quality control laboratories to implement more accurate and reliable testing protocols for Formoterol batches.
Mechanistic Insights into the Synthetic Route
The synthesis of Compound A is a masterclass in efficient functional group manipulation, beginning with the stereoselective reduction of a ketone precursor. The first step involves the reduction of 1-(4-methoxyphenyl)propan-2-one using sodium borohydride in methanol. This transformation is critical as it establishes the chiral alcohol intermediate (Intermediate II) which serves as the scaffold for the subsequent modifications. The choice of sodium borohydride is strategic; it offers excellent chemoselectivity, reducing the ketone without affecting the aromatic methoxy group, and operates under mild temperatures (0-25°C) to prevent racemization or side reactions. The resulting alcohol is then activated via mesylation, converting the hydroxyl group into a superior leaving group (Intermediate III), which primes the molecule for the final nucleophilic attack.
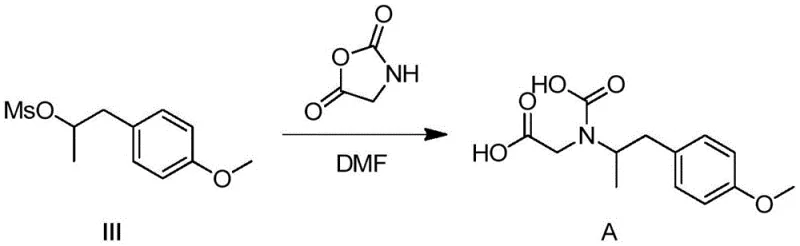
The culmination of the synthesis occurs in the third step, where the mesylate intermediate undergoes a nucleophilic substitution with 2,5-oxazolidinedione. This reaction is facilitated by cesium carbonate in anhydrous DMF at elevated temperatures (60-80°C). The base deprotonates the nitrogen of the heterocyclic reagent, generating a potent nucleophile that displaces the mesylate group. Following this alkylation, the reaction mixture is subjected to acidic workup, which likely induces ring opening or hydrolysis to reveal the final dicarboxylic acid functionality characteristic of Compound A. This mechanistic pathway is highly efficient, minimizing the formation of elimination byproducts and ensuring that the nitrogen atom is successfully dialkylated or functionalized to match the impurity profile observed in Formoterol production.
How to Synthesize Formoterol Related Substance A Efficiently
The preparation of Compound A requires precise control over reaction parameters to maximize yield and purity. The process begins with the careful addition of sodium borohydride to the ketone substrate at low temperatures to manage exothermicity, followed by a controlled warm-up to complete the reduction. The subsequent mesylation step demands anhydrous conditions to prevent hydrolysis of the sulfonyl chloride, utilizing triethylamine as a proton scavenger. Finally, the coupling reaction with 2,5-oxazolidinedione requires rigorous temperature management and stoichiometry optimization to drive the substitution to completion. For the complete standardized operating procedure and specific molar ratios, please refer to the detailed guide below.
- Reduce 1-(4-methoxyphenyl)propan-2-one with sodium borohydride in methanol to form the chiral alcohol intermediate.
- Convert the alcohol intermediate to a mesylate leaving group using methanesulfonyl chloride and triethylamine in dichloromethane.
- React the mesylate intermediate with 2,5-oxazolidinedione in DMF using cesium carbonate, followed by acidification to isolate Compound A.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis offers substantial strategic benefits beyond mere technical feasibility. The primary advantage lies in the decoupling of reference standard production from the variability of bulk API manufacturing. By establishing a dedicated, short synthetic route, suppliers can guarantee the continuous availability of Compound A regardless of fluctuations in Formoterol production schedules. This independence mitigates the risk of supply shortages that could otherwise halt quality control operations, ensuring that batch release testing proceeds without interruption. Furthermore, the use of commodity chemicals and standard unit operations simplifies the sourcing strategy, reducing dependency on exotic or single-source reagents.
- Cost Reduction in Manufacturing: The streamlined three-step process eliminates the need for complex preparative HPLC purification typically required to isolate trace impurities from bulk reactions. By synthesizing the impurity directly from low-cost starting materials like 4-methoxyphenylacetone derivatives, the cost of goods sold for the reference standard is drastically reduced. Additionally, the high yields reported in each step minimize raw material waste and solvent consumption, contributing to a leaner and more economically viable production model. This efficiency translates into significant cost savings for pharmaceutical companies purchasing these critical quality control materials.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route ensures a stable and predictable supply of Compound A. Since the reaction conditions are mild and do not require specialized high-pressure or cryogenic equipment, the process can be easily replicated across multiple manufacturing sites if necessary. This flexibility enhances supply chain resilience, protecting against regional disruptions or logistical bottlenecks. Moreover, the stability of the intermediates allows for potential stockpiling, further securing the supply line for long-term quality control programs.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing solvents like methanol, dichloromethane, and DMF which are well-understood in industrial settings. The avoidance of heavy metal catalysts or hazardous reagents simplifies waste treatment and disposal, aligning with modern environmental, health, and safety (EHS) standards. The ability to scale from gram-scale laboratory synthesis to multi-kilogram production without significant process re-engineering facilitates rapid response to increasing regulatory demands for impurity profiling, ensuring that compliance timelines are met without compromising environmental responsibilities.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical specifications and applications of Formoterol Related Substance A. These answers are derived directly from the experimental data and findings presented in the patent documentation, providing clarity on the compound's properties and synthesis. Understanding these details is crucial for R&D teams implementing new QC methods and for procurement specialists evaluating supplier capabilities.
Q: Why is controlling Formoterol Related Substance A critical?
A: Compound A has demonstrated cytotoxicity against HUVEC cells with an IC50 of 7.6 μM, significantly lower than Formoterol itself, necessitating strict limits below 0.1% in final drug products.
Q: What is the advantage of this new synthetic method?
A: The patented route offers a short, three-step process with mild reaction conditions and high overall yield, avoiding the difficulties of isolating trace impurities from bulk Formoterol production.
Q: Can this process be scaled for commercial reference standard production?
A: Yes, the use of common solvents like methanol, DCM, and DMF, along with stable reagents like sodium borohydride and cesium carbonate, ensures excellent scalability and supply chain reliability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Formoterol Related Substance A Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality reference standards play in ensuring the safety and efficacy of life-saving medications like Formoterol. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency. We are committed to delivering materials with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards.
We invite you to collaborate with us to secure your supply chain for Formoterol impurity standards. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized synthesis of Compound A can enhance your quality control operations while reducing overall costs.
