Advanced Synthesis of Voglibose Impurity II for Commercial Pharmaceutical Quality Control
The pharmaceutical industry continuously demands higher standards for quality control reference substances, particularly for complex aminosugar derivatives like Voglibose, a widely prescribed alpha-glucosidase inhibitor used in the management of diabetes mellitus. Patent CN113105344B introduces a groundbreaking preparation method for high-purity Voglibose Impurity II, addressing the critical need for reliable reference standards mandated by the Chinese Pharmacopoeia 2020 edition. This technical breakthrough shifts the paradigm from inefficient multi-step syntheses to a streamlined, one-step reductive amination process catalyzed by weak acids. For R&D directors and procurement specialists, this innovation represents a significant opportunity to enhance the reliability of analytical method validation while simultaneously optimizing the cost structure of reference material acquisition. The ability to produce this specific impurity with high fidelity ensures that pharmaceutical manufacturers can maintain rigorous compliance with regulatory impurity profiling requirements without incurring the prohibitive costs associated with legacy synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
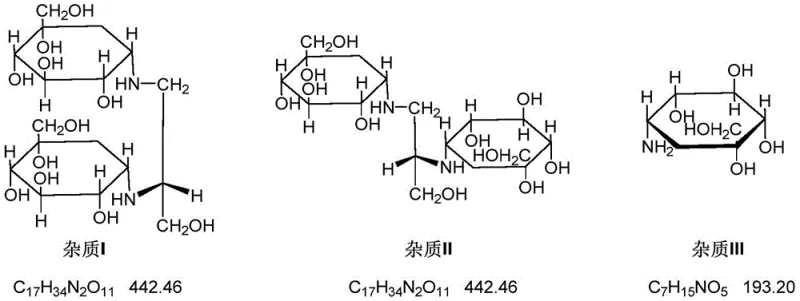
Historically, the industrial synthesis of Voglibose and its associated impurities has relied heavily on semi-synthetic pathways that are fraught with inefficiencies and environmental hazards. As detailed in prior art such as CN110511152, the conventional preparation of Voglibose Impurity II involves a cumbersome five-step sequence that necessitates the use of benzyl groups as hydroxyl protecting agents. This protection strategy introduces significant complexity, requiring the use of heavy metal palladium catalysts for the subsequent hydrogenolysis step to remove the protecting groups, which creates substantial challenges in meeting strict heavy metal residue limits for pharmaceutical ingredients. Furthermore, the cumulative yield loss across these multiple reaction stages results in a total process yield of merely 9.4%, rendering the method economically unsustainable for large-scale production. The reliance on non-commercial intermediates that must be prepared in advance further exacerbates supply chain vulnerabilities, leading to extended lead times and increased inventory holding costs for manufacturers seeking to secure high-purity reference standards for quality control operations.
The Novel Approach
In stark contrast to the legacy methodologies, the novel approach disclosed in patent CN113105344B utilizes a direct reductive amination reaction between Valiolamine and 1,3-dihydroxyacetone, catalyzed specifically by weak acids rather than traditional strong mineral acids. This strategic shift in catalytic conditions allows for the direct formation of the target impurity structure without the need for complex protection and deprotection sequences, thereby drastically reducing the number of unit operations required. The use of commercially available starting materials eliminates the dependency on specialized intermediates, significantly enhancing the robustness and continuity of the supply chain for this critical reference substance. By optimizing the reaction conditions to favor the formation of Impurity II, this method achieves a crude HPLC purity of over 40% in the reaction mixture, which is a substantial improvement over the 1% purity observed with hydrochloric acid catalysis. This efficiency gain translates directly into reduced solvent consumption, lower waste generation, and a more environmentally friendly manufacturing process that aligns with modern green chemistry principles and regulatory expectations for sustainable pharmaceutical production.
Mechanistic Insights into Weak Acid-Catalyzed Reductive Amination
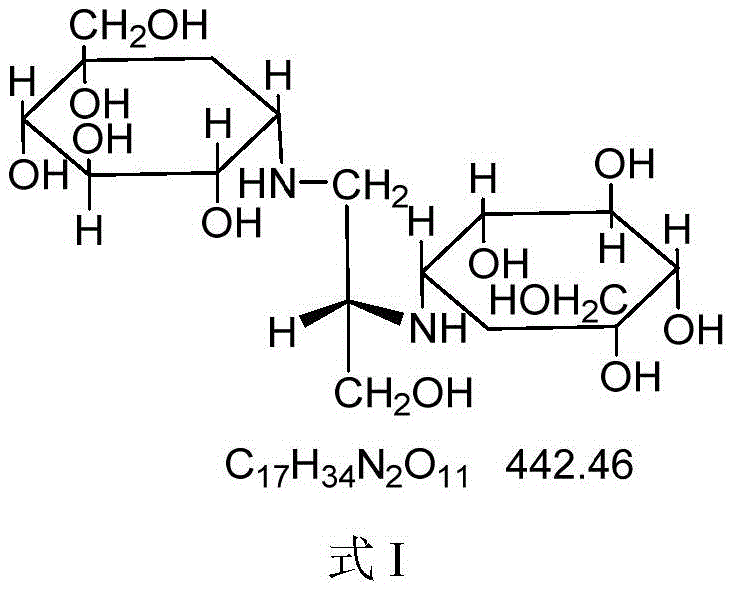
The core chemical innovation of this process lies in the precise modulation of the reaction kinetics through the selection of a weak acid catalyst with a pKa value greater than 2.7, such as acetic acid, formic acid, or citric acid. In the reductive amination pathway, the reaction proceeds through the formation of a Schiff base (imine) intermediate, the rate of which is critically dependent on the acidity of the reaction medium. Under strong acid conditions, the rapid generation of the Schiff base often leads to uncontrolled side reactions and the formation of undesired byproducts like Voglibose Impurity I or III. However, under weakly acidic conditions, the generation rate of the Schiff base is effectively slowed, allowing for a more controlled and selective reaction pathway that favors the specific stereochemical configuration of Voglibose Impurity II. This mechanistic control is essential for R&D teams focused on impurity profiling, as it ensures that the reference standard produced is chemically identical to the impurity generated during the actual drug substance manufacturing process, thereby validating the accuracy of HPLC detection methods used for batch release testing.
Furthermore, the purification strategy employs a sophisticated sequence of ion exchange resin chromatography to achieve the requisite purity levels for reference standards. The process utilizes a combination of strong acid cation exchange resins, weak acid cation exchange resins, and strong base anion exchange resins to selectively adsorb and elute the target impurity while removing unreacted amines, ketones, and ionic byproducts. This multi-stage purification protocol is capable of elevating the HPLC purity from the crude reaction levels to optimally more than 99%, meeting the stringent requirements for pharmacopoeial reference substances. The ability to achieve such high purity without the need for preparative HPLC or complex crystallization steps represents a significant advantage in terms of process scalability and cost efficiency. For quality control laboratories, this ensures a consistent supply of high-fidelity reference materials that are critical for the accurate quantification of impurities in finished Voglibose drug products, thereby safeguarding patient safety and regulatory compliance.
How to Synthesize Voglibose Impurity II Efficiently
The synthesis of Voglibose Impurity II via this novel pathway is designed for operational simplicity and industrial scalability, making it an ideal candidate for technology transfer to commercial manufacturing facilities. The process begins with the dissolution of Valiolamine and 1,3-dihydroxyacetone in a polar organic solvent such as ethanol, acetonitrile, or dimethyl sulfoxide, followed by the addition of a catalytic amount of a weak organic acid. The reaction mixture is then treated with a reducing agent, such as sodium cyanoborohydride or sodium triacetoxyborohydride, and heated to moderate temperatures ranging from 20°C to 80°C to drive the reductive amination to completion. Detailed standardized synthesis steps see the guide below.
- Mix Valiolamine and 1,3-dihydroxyacetone in a polar organic solvent with a weak acid catalyst such as acetic or formic acid.
- Add a reducing agent like sodium cyanoborohydride and heat the mixture to facilitate the reductive amination reaction.
- Purify the crude reaction product using a sequence of cation and anion exchange resins to achieve over 99% HPLC purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this weak acid-catalyzed synthesis route offers profound advantages for procurement managers and supply chain heads tasked with optimizing the cost and reliability of pharmaceutical raw material sourcing. The elimination of multi-step protection and deprotection chemistry significantly reduces the consumption of expensive reagents and solvents, leading to a drastic simplification of the manufacturing workflow. This streamlining of the process not only lowers the direct material costs but also reduces the labor and overhead expenses associated with managing complex multi-stage reactions. For procurement teams, this translates into a more competitive pricing structure for the final reference standard, allowing for significant cost savings in the overall quality control budget without compromising on the purity or quality of the material supplied. The use of commodity chemicals further insulates the supply chain from volatility associated with specialized intermediates, ensuring a stable and predictable sourcing environment.
- Cost Reduction in Manufacturing: The removal of heavy metal palladium catalysts and benzyl protecting groups eliminates the need for expensive hydrogenation equipment and complex heavy metal scavenging processes, which are traditionally cost-prohibitive steps in fine chemical synthesis. By relying on simple weak acid catalysts and standard reducing agents, the process significantly reduces the capital expenditure required for reactor setup and the operational expenditure related to waste treatment and catalyst recovery. This fundamental shift in the cost drivers of the synthesis allows for a more lean manufacturing model, where resources are allocated more efficiently towards value-added purification steps rather than wasteful protection chemistry. Consequently, the overall cost of goods sold for the impurity standard is substantially lowered, providing a clear economic advantage for manufacturers looking to optimize their supply chain expenditures.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials such as Valiolamine and 1,3-dihydroxyacetone ensures that the production of this impurity is not bottlenecked by the availability of custom-synthesized intermediates. This accessibility of raw materials significantly reduces the lead time for production planning and execution, allowing for more responsive inventory management and faster turnaround times for customer orders. For supply chain heads, this means a reduced risk of stockouts and a greater ability to scale production up or down in response to market demand fluctuations. The robustness of the supply chain is further enhanced by the simplicity of the process, which minimizes the risk of batch failures due to complex reaction conditions, thereby ensuring a continuous and reliable flow of high-purity reference standards to support global pharmaceutical manufacturing operations.
- Scalability and Environmental Compliance: The one-step nature of the reaction and the use of ion exchange purification make this process highly amenable to scale-up from laboratory to commercial production volumes without the need for significant process re-engineering. The reduction in solvent usage and the avoidance of heavy metal waste streams align with increasingly stringent environmental regulations, reducing the regulatory burden and potential liability associated with hazardous waste disposal. This environmental compliance is a critical factor for supply chain sustainability, as it ensures that the manufacturing process can be maintained over the long term without facing regulatory shutdowns or costly remediation efforts. The ability to scale this process efficiently ensures that the supply of Voglibose Impurity II can meet the growing demands of the global diabetes treatment market, supporting the continuous production of high-quality generic and branded Voglibose formulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Voglibose Impurity II, based on the specific technical disclosures within patent CN113105344B. These insights are designed to provide clarity for R&D and procurement professionals evaluating the feasibility of integrating this new supply source into their quality control workflows. Understanding the specific advantages of the weak acid catalysis and ion exchange purification helps stakeholders make informed decisions about reference standard procurement. The answers below reflect the verified data and process capabilities described in the patent documentation.
Q: Why is a weak acid catalyst preferred over hydrochloric acid for this synthesis?
A: Using a weak acid catalyst with a pKa greater than 2.7 significantly slows the generation rate of the Schiff base intermediate, which favors the formation of the specific Voglibose Impurity II structure over other byproducts, thereby increasing the HPLC purity in the crude reaction mixture from 1% to over 40%.
Q: How is the final purity of the impurity standard achieved?
A: The process utilizes a multi-stage ion exchange purification strategy involving strong acid cation, weak acid cation, and strong base anion resins, which effectively removes unreacted starting materials and side products to achieve a final HPLC purity of optimally more than 99%.
Q: What are the scalability advantages of this new method?
A: This method eliminates the need for multi-step protection and deprotection reactions and avoids the use of heavy metal palladium catalysts, resulting in a drastically simplified workflow that uses commercially available raw materials and is highly suitable for industrial-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Voglibose Impurity II Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity reference standards play in ensuring the safety and efficacy of pharmaceutical products, and we are committed to delivering Voglibose Impurity II with the highest level of technical excellence. Our CDMO team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistent quality. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of Voglibose Impurity II meets or exceeds the requirements of the Chinese Pharmacopoeia 2020 edition and other international standards. Our commitment to quality is backed by a robust technical support team that can assist with method validation and impurity profiling, ensuring a seamless integration of our materials into your quality control systems.
We invite you to collaborate with us to optimize your supply chain for Voglibose reference standards and achieve significant operational efficiencies. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific annual volume needs. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our advanced synthesis method can enhance your quality control capabilities while reducing overall costs. Partnering with NINGBO INNO PHARMCHEM ensures a reliable, compliant, and cost-effective source for your critical pharmaceutical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
