Scalable Synthesis of GDC-0077 Intermediate for Global Pharmaceutical Supply Chains
The pharmaceutical industry's relentless pursuit of effective oncology treatments has placed significant spotlight on PI3K inhibitors, particularly Inavolisib (GDC-0077), a potent agent for treating HR+, HER2- breast cancer. As demand for this critical therapeutic grows, the efficiency and reliability of its supply chain become paramount. A recent technological breakthrough, documented in patent CN115417884A, introduces a revolutionary synthetic pathway for the key GDC-0077 intermediate. This novel methodology addresses longstanding bottlenecks in traditional manufacturing by utilizing a streamlined three-step sequence: etherification, deprotection, and heterocyclic cyclization. By shifting away from hazardous reagents and complex purification protocols, this innovation offers a robust framework for producing high-purity pharmaceutical intermediates. For global procurement leaders and R&D directors, understanding this shift is essential for securing a stable, cost-effective supply of this vital oncology building block.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the GDC-0077 core structure has been plagued by significant safety and operational challenges that hinder efficient commercial scale-up. Traditional routes often rely on the use of chloroacetaldehyde, a reagent known for its pungent odor, high toxicity, and flammability, posing severe risks to plant personnel and requiring specialized containment infrastructure. Furthermore, these legacy processes typically involve an amination cyclization followed by dissociation and a subsequent reaction with chloroacetaldehyde, which generates substantial quantities of inorganic salt byproducts. This salt formation complicates downstream processing, necessitating extensive washing and purification steps that drive up waste disposal costs. Additionally, the intermediate hydrochloride salts produced in these older methods are often highly hygroscopic and difficult to filter, leading to inconsistent batch quality and extended production cycles. These factors collectively create a fragile supply chain vulnerable to delays and regulatory scrutiny regarding environmental safety.
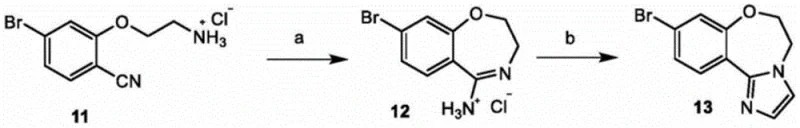
The Novel Approach
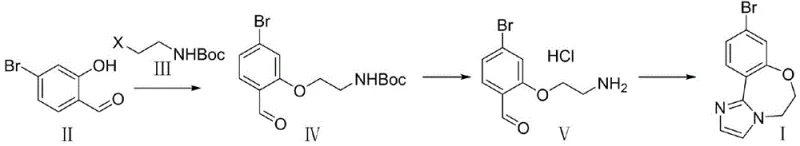
In stark contrast, the innovative strategy outlined in the patent data leverages a much safer and more atom-economical design centered on readily available starting materials. The new route initiates with the nucleophilic substitution of 4-bromo-2-hydroxybenzaldehyde with N-Boc-haloethylamine, effectively constructing the necessary carbon-oxygen backbone without the need for dangerous aldehydes. This is followed by a mild acid-mediated deprotection to reveal the free amine, which then undergoes a concise cyclization with glyoxal and an ammonia source. This approach not only eliminates the hazards associated with chloroacetaldehyde but also simplifies the reaction profile, allowing for easier control of reaction parameters such as temperature and pH. The result is a cleaner reaction mixture that facilitates straightforward isolation of the product, significantly reducing the burden on purification units and enhancing the overall throughput of the manufacturing facility.
Mechanistic Insights into Etherification and Heterocyclic Cyclization
The cornerstone of this improved synthesis lies in the initial etherification step, where 4-bromo-2-hydroxybenzaldehyde reacts with N-Boc-haloethylamine in the presence of a mild inorganic base such as potassium carbonate or cesium carbonate. This transformation proceeds via a classic SN2 mechanism, where the phenoxide anion attacks the haloethyl group, displacing the halide leaving group to form the protected amino-ether linkage. The choice of solvent, ranging from polar aprotic options like DMF and acetonitrile to greener alternatives like 2-methyltetrahydrofuran, plays a critical role in solubilizing the reactants and stabilizing the transition state. Operating at moderate temperatures between 40°C and 90°C ensures rapid kinetics while minimizing side reactions such as aldehyde degradation or over-alkylation. This step is remarkably efficient, with patent data indicating yields approaching 98.7%, demonstrating the high fidelity of the bond formation and the stability of the Boc protecting group under these conditions.
Following the etherification, the process moves to the critical cyclization phase which constructs the imidazo-fused ring system characteristic of the GDC-0077 scaffold. After the Boc group is cleanly removed using hydrochloric acid to generate the free amine hydrochloride salt, the molecule is primed for condensation. The addition of glyoxal and an ammonium source triggers a multi-component condensation reaction. The primary amine reacts with one carbonyl of the glyoxal to form an imine, while the ammonia source facilitates the closure of the second ring nitrogen. This cascade occurs efficiently in alcoholic solvents like methanol or ethanol under reflux conditions (50°C to 100°C). The mechanism is highly selective, favoring the formation of the desired heterocycle over potential oligomerization of the glyoxal. This specificity is crucial for impurity control, ensuring that the final API intermediate meets the stringent purity specifications required for oncology drug substances, often exceeding 99.5% purity after simple recrystallization.
How to Synthesize GDC-0077 Intermediate Efficiently
Implementing this synthesis requires precise control over stoichiometry and thermal conditions to maximize yield and minimize impurity profiles. The process is divided into three distinct operational stages: the initial coupling of the phenol and amine precursors, the acidic deprotection to unmask the reactive amine, and the final thermal cyclization to close the heterocyclic ring. Each step has been optimized in the patent examples to balance reaction rate with product stability, utilizing common industrial solvents and reagents that are easy to source and recycle. Operators should pay particular attention to the temperature control during the deprotection phase to prevent exotherms, and ensure adequate mixing during the cyclization to maintain homogeneity. For a detailed breakdown of the specific operational parameters, reagent equivalents, and workup procedures validated in the patent examples, please refer to the standardized protocol below.
- Perform nucleophilic substitution between 4-bromo-2-hydroxybenzaldehyde and N-Boc-haloethylamine using a carbonate base in solvents like acetone or DMF at 40-90°C.
- Execute Boc deprotection of the resulting ether intermediate using hydrochloric acid in methanol or ethanol at temperatures between -5°C and 30°C.
- Conduct cyclization of the amino-aldehyde hydrochloride with glyoxal and an ammonium source in alcohol/water mixtures under reflux conditions to form the final heterocyclic structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthetic route offers transformative benefits for procurement managers and supply chain directors tasked with optimizing cost structures and ensuring continuity of supply. The shift away from hazardous and regulated reagents like chloroacetaldehyde significantly lowers the barrier to entry for manufacturing partners, expanding the pool of qualified suppliers and reducing geopolitical supply risks. Furthermore, the simplification of the post-reaction workup—moving from complex salt filtrations to straightforward crystallizations—drastically reduces the cycle time per batch. This efficiency gain translates directly into increased production capacity without the need for capital-intensive equipment upgrades. The robustness of the chemistry also implies a lower rate of batch failures, providing greater predictability in delivery schedules and inventory planning for downstream API manufacturers.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous chloroacetaldehyde, combined with the use of commodity chemicals like glyoxal and ammonium salts, results in a substantially lower raw material cost profile. Additionally, the high yields observed in the initial etherification step minimize the consumption of the valuable bromo-hydroxybenzaldehyde starting material, further driving down the cost of goods sold. The simplified purification process reduces solvent usage and waste treatment expenses, contributing to a leaner and more economically viable manufacturing model.
- Enhanced Supply Chain Reliability: By relying on stable, non-hygroscopic intermediates and avoiding reagents with strict transportation regulations, the supply chain becomes inherently more resilient. The starting materials, such as 4-bromo-2-hydroxybenzaldehyde, are widely available from multiple global vendors, mitigating the risk of single-source bottlenecks. This diversification of the supply base ensures that production can continue uninterrupted even if one vendor faces disruptions, thereby securing the continuity of supply for critical cancer therapies.
- Scalability and Environmental Compliance: The process is designed with green chemistry principles in mind, generating fewer inorganic byproducts and avoiding the release of toxic volatiles associated with traditional methods. This reduced environmental footprint simplifies regulatory compliance and permitting for manufacturing sites. Moreover, the reaction conditions are mild and easily transferable from kilogram to multi-ton scales, allowing for seamless commercial scale-up of complex pharmaceutical intermediates without the need for specialized high-pressure or cryogenic equipment.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology, we have compiled answers to common inquiries regarding the reaction specifics and quality outcomes. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a transparent view of the process capabilities. Understanding these details is crucial for assessing the fit of this route within your existing manufacturing infrastructure and quality management systems.
Q: What are the critical advantages of this new GDC-0077 synthesis route over conventional methods?
A: The novel route eliminates the use of toxic and flammable chloroacetaldehyde, replacing it with safer N-Boc-haloethylamine. It also avoids the generation of large amounts of inorganic salts and hygroscopic intermediates that complicate filtration, resulting in a simpler post-treatment process and higher overall yields suitable for industrial scale-up.
Q: What are the optimal reaction conditions for the final cyclization step?
A: The final cyclization is optimally performed by reacting the amino-aldehyde hydrochloride with glyoxal and an ammonium salt (such as ammonium acetate or ammonia water) in a solvent mixture of methanol, ethanol, or water. The reaction is heated to reflux, typically between 50°C and 100°C, ensuring complete conversion to the target heterocyclic structure with purity exceeding 99%.
Q: Is this synthesis method suitable for large-scale commercial manufacturing?
A: Yes, the method is specifically designed for industrial applicability. It utilizes readily available starting materials like 4-bromo-2-hydroxybenzaldehyde and employs robust reaction conditions (e.g., simple etherification and acid deprotection) that are easily controlled. The high yields reported in examples (over 98% for step 1 and 81% for the final step) demonstrate its efficiency for ton-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable GDC-0077 Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of next-generation oncology drugs depends on a partner who can deliver both technical excellence and supply security. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical clients. We are committed to maintaining stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify that every batch of GDC-0077 intermediate meets the highest standards for safety and efficacy. Our facility is equipped to handle the specific solvent systems and thermal requirements of this novel synthesis, guaranteeing a consistent and high-quality supply.
We invite you to engage with our technical procurement team to discuss how this optimized route can benefit your specific project timelines and budgetary goals. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages of switching to this superior manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a seamless integration of this critical intermediate into your supply chain.
