Revolutionizing Oteracil Potassium Production: A High-Purity Pharmaceutical Intermediate Strategy
The pharmaceutical landscape for anticancer agents continues to evolve, driven by the demand for safer and more efficient biochemical regulators. Patent CN110655493A introduces a groundbreaking synthesis method for Oteracil Potassium, a critical component in oral fluoropyrimidine anticancer therapies. This compound acts as a potent inhibitor of pyrimidine phosphoribosyl transferase, effectively reducing the gastrointestinal toxicity associated with 5-fluorouracil treatment. The disclosed technology represents a significant leap forward in pharmaceutical intermediate manufacturing, shifting away from hazardous oxidation processes toward a mild, acid-catalyzed cyclization strategy. By leveraging readily available starting materials such as 2-carbamoylamino-2-oxyacetic acid and urea, this innovation addresses long-standing challenges in yield stability and environmental compliance. For global supply chains, this development signals a move towards more robust and sustainable production capabilities for high-value oncology ingredients.
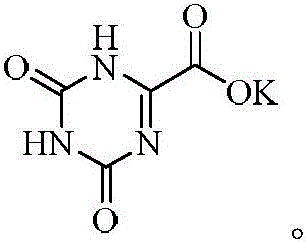
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of Oteracil Potassium has been plagued by complex multi-step sequences involving dangerous reagents and harsh conditions. Early methodologies, such as the condensation of biuret with potassium ethyl oxalate, required absolute ethanol and potassium ethoxide, resulting in prohibitively high raw material costs and difficult operational parameters. Subsequent attempts utilizing uric acid as a precursor necessitated oxidative ring-opening using agents like iodine, hydrogen peroxide, or potassium permanganate, as illustrated in the reaction schemes below. These oxidation-based routes suffered from severe drawbacks, including the generation of manganese dioxide sludge which created substantial environmental disposal burdens. Furthermore, the use of strong oxidants often led to over-oxidation, generating peroxide impurities that were notoriously difficult to separate, thereby capping the overall yield at approximately 55 percent. Alternative approaches employing bromine or hypohalites introduced significant safety risks due to the toxicity and instability of these halogenated species, making them unsuitable for modern green chemistry standards.
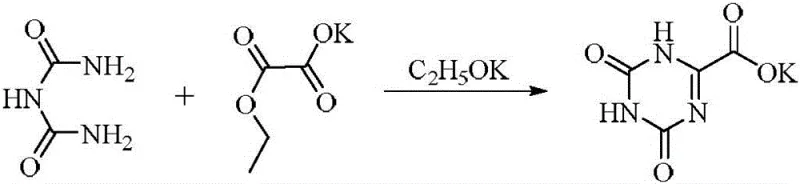
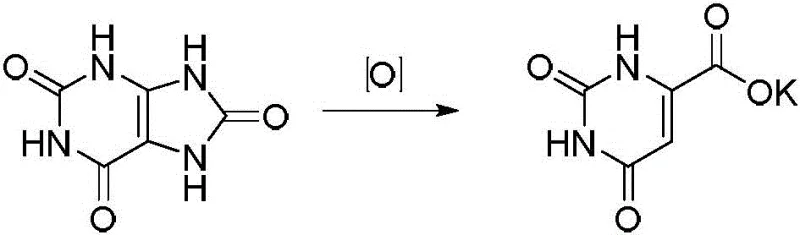
The Novel Approach
In stark contrast to these legacy methods, the patented process utilizes a direct condensation and cyclization strategy that fundamentally simplifies the molecular construction of the triazine core. As depicted in the reaction pathway, 2-carbamoylamino-2-oxyacetic acid reacts directly with urea in the presence of a catalytic amount of organic acid, such as p-toluenesulfonic acid. This approach completely bypasses the need for oxidative cleavage steps, thereby eliminating the formation of heavy metal waste and toxic halogenated byproducts. The reaction proceeds smoothly in common alcohol solvents like ethanol at moderate temperatures ranging from 35°C to 70°C, ensuring energy efficiency and operational safety. The simplicity of the workup procedure, which involves merely adjusting the pH to precipitate the potassium salt followed by washing with water and cold alcohol, allows for the isolation of the product with exceptional purity. This streamlined workflow not only reduces the number of unit operations but also drastically cuts down on solvent consumption and processing time, offering a clear economic advantage for large-scale API intermediate production.
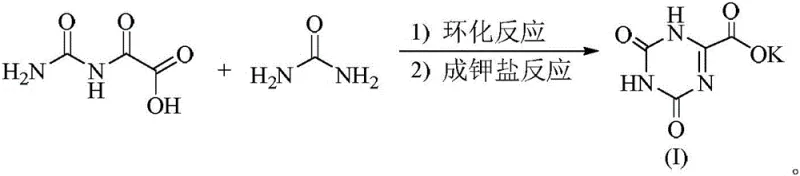
Mechanistic Insights into Acid-Catalyzed Triazine Cyclization
The core of this technological advancement lies in the efficient acid-catalyzed cyclization mechanism that constructs the 1,3,5-triazine-2,4,6-trione skeleton. In this process, the carbonyl group of the 2-carbamoylamino-2-oxyacetic acid is activated by the proton donor catalyst, facilitating a nucleophilic attack by the amino groups of the urea molecule. This condensation leads to the formation of intermediate species that subsequently undergo dehydration and ring closure to form the stable heterocyclic structure. The choice of catalyst is critical; strong organic acids like p-toluenesulfonic acid provide the optimal proton activity to drive the equilibrium towards the product without degrading the sensitive functional groups. Unlike oxidative pathways where radical species can cause indiscriminate bond breaking, this ionic mechanism ensures high regioselectivity, preserving the integrity of the carboxylic acid moiety intended for salt formation. The reaction kinetics are favorable enough to reach completion within 4 to 8 hours, demonstrating a robust conversion rate that minimizes the accumulation of unreacted starting materials.
From an impurity control perspective, this mechanism offers superior selectivity compared to oxidation-based routes. Traditional methods often generated complex mixtures of over-oxidized byproducts and metal complexes that required extensive chromatographic purification. In the new cyclization route, the primary impurities are limited to unreacted urea or hydrolysis products, which are highly soluble in the aqueous wash steps employed during isolation. The absence of transition metals means there is no risk of metal-catalyzed degradation during storage, enhancing the shelf-life stability of the final high-purity pharmaceutical intermediate. Furthermore, the precise control over pH during the salt formation step ensures that the potassium ion coordinates specifically with the carboxylate anion, preventing the formation of mixed salts or polymorphs that could affect bioavailability. This mechanistic clarity provides R&D teams with a predictable and controllable process window, essential for regulatory filing and quality assurance.
How to Synthesize Oteracil Potassium Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and thermal management to maximize yield and crystal quality. The process begins with the dissolution of the acid precursor and urea in a lower alcohol solvent, followed by the addition of the catalyst under ambient conditions before heating. Maintaining the reaction temperature within the specified range of 35°C to 70°C is crucial to balance reaction rate against potential thermal degradation. Once the cyclization is complete, the cooling phase must be controlled to induce uniform nucleation during the pH adjustment stage, which directly impacts the filtration characteristics of the final solid. For a detailed breakdown of the specific molar ratios, solvent volumes, and crystallization times optimized in the patent examples, please refer to the standardized protocol below.
- Combine 2-carbamoylamino-2-oxyacetic acid, urea, and an acid catalyst (e.g., p-toluenesulfonic acid) in a solvent like ethanol at room temperature.
- Heat the mixture to 35-70°C to facilitate the cyclization reaction, monitoring progress until completion.
- Cool the solution, adjust pH to 5-6 using potassium hydroxide to form the salt, crystallize, filter, and dry to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to this non-oxidative synthesis route presents compelling opportunities for cost optimization and risk mitigation. The reliance on commodity chemicals like urea and glyoxylic acid derivatives removes the volatility associated with sourcing specialized oxidants or halogenated reagents. This stability in raw material sourcing translates directly into more predictable pricing models and reduced exposure to supply disruptions caused by environmental regulations on hazardous chemical transport. Additionally, the simplified downstream processing eliminates the need for expensive resin treatments or complex extraction protocols typically required to remove metal residues, further driving down the cost of goods sold. The ability to produce material with consistently high purity reduces the burden on quality control laboratories and minimizes the risk of batch rejection, ensuring a smoother flow of materials into the final drug product manufacturing lines.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous oxidizing agents such as bromine or potassium permanganate significantly lowers the direct material costs associated with production. By removing the need for heavy metal scavengers and complex purification steps to address over-oxidation impurities, the overall processing time and utility consumption are drastically reduced. This streamlined approach allows for higher throughput in existing reactor vessels without the need for specialized corrosion-resistant equipment required for handling strong acids or halogens. Consequently, the total manufacturing cost per kilogram is substantially decreased, improving the margin profile for the final anticancer formulation.
- Enhanced Supply Chain Reliability: The starting materials for this process, specifically urea and 2-carbamoylamino-2-oxyacetic acid, are widely available from multiple global suppliers, reducing dependency on single-source vendors. The mild reaction conditions and use of common solvents like ethanol mean that production can be easily transferred between different manufacturing sites without significant requalification efforts. This flexibility ensures business continuity and allows for rapid scaling of production capacity to meet fluctuating market demands for oncology therapies. Furthermore, the reduced hazard profile of the process simplifies logistics and storage requirements, minimizing regulatory hurdles associated with transporting dangerous goods.
- Scalability and Environmental Compliance: This synthesis method aligns perfectly with modern green chemistry principles by avoiding the generation of toxic heavy metal waste and halogenated organic byproducts. The absence of manganese dioxide sludge or bromine waste streams simplifies wastewater treatment and reduces the environmental footprint of the manufacturing facility. The high atom economy of the cyclization reaction ensures that the majority of the input mass is converted into the desired product, minimizing waste disposal costs. These factors make the process highly scalable from pilot plant to commercial tonnage, facilitating a smoother regulatory approval process in regions with stringent environmental protection laws.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation to ensure accuracy and relevance for industry stakeholders. Understanding these details is crucial for evaluating the feasibility of adopting this route for your specific supply chain needs.
Q: What are the primary advantages of the new cyclization route over traditional oxidation methods?
A: The new route eliminates the need for hazardous oxidants like bromine or potassium permanganate, significantly reducing environmental pollution and simplifying purification steps while achieving yields over 83%.
Q: How does this synthesis method impact the purity profile of Oteracil Potassium?
A: By avoiding over-oxidation side reactions common in uric acid pathways, this method consistently delivers purity exceeding 99.7% with maximum single impurities below 0.1%.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the use of mild reaction temperatures (35-70°C), inexpensive raw materials like urea, and standard solvents like ethanol makes it highly scalable and safe for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oteracil Potassium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving anticancer medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical clients. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of Oteracil Potassium meets the highest international standards. Our expertise in implementing advanced cyclization technologies allows us to deliver products with superior impurity profiles, supporting your regulatory filings and clinical trial timelines.
We invite you to collaborate with us to leverage these technological advancements for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency and product quality.
