Advanced Synthesis of Perfluoroalkyl Sulfonyl Isoflavones for High-Potency Anticancer Drug Development
The pharmaceutical industry is constantly seeking novel scaffolds that can overcome the limitations of existing anticancer therapies, particularly in the realm of leukemia treatment. Patent CN101003530A introduces a groundbreaking methodology for the preparation of perfluoroalkyl group sulfonyl isoflavones, a class of compounds demonstrating potent anticarcinogenic properties. This technical disclosure outlines a robust synthetic route that transforms readily available hydroxyisoflavones into highly bioactive derivatives through a strategic sulfonylation process. The innovation lies not only in the biological efficacy against HL-60 human leukemia cell lines but also in the operational simplicity of the chemical transformation. By leveraging a nucleophilic substitution mechanism under mild basic conditions, this process bypasses the complex multi-step sequences often required for fluorination, offering a direct path to high-value pharmaceutical intermediates. For R&D teams and procurement specialists alike, understanding the nuances of this patent is critical for evaluating its potential integration into existing drug discovery pipelines and supply chain strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to modifying the isoflavone backbone to enhance bioactivity have historically faced significant hurdles, particularly regarding the introduction of fluorine-containing groups. Conventional methods often rely on the introduction of trifluoromethyl or difluoromethyl groups, which, while useful, frequently fail to deliver substantial improvements in both anticancer activity and solubility profiles. Furthermore, the synthetic routes to install these groups are often fraught with difficulties, requiring harsh reaction conditions, expensive reagents, or complex protection-deprotection strategies that drive up manufacturing costs and extend lead times. The low absorption of native isoflavones in the intestinal tract further complicates their utility, necessitating structural modifications that do not compromise the core pharmacophore. Many existing protocols suffer from poor regioselectivity, leading to mixtures of isomers that are difficult and costly to separate, thereby reducing the overall process efficiency and yield. These limitations create a bottleneck for the commercial scale-up of complex pharmaceutical intermediates, making the search for alternative, more efficient synthetic methodologies a top priority for forward-thinking chemical enterprises.
The Novel Approach
The methodology disclosed in CN101003530A represents a paradigm shift by utilizing perfluoroalkylsulfonyl fluorides as the key electrophilic reagents. This novel approach simplifies the synthetic landscape by employing a direct reaction between hydroxyisoflavones and perfluoroalkylsulfonyl fluorides in the presence of a mild base like potassium carbonate. The process operates effectively in polar aprotic solvents such as N,N-dimethylformamide (DMF), allowing for excellent solubility of the reactants and facilitating smooth nucleophilic attack. Unlike traditional fluorination techniques that might require cryogenic temperatures or specialized catalysts, this method proceeds at moderate temperatures ranging from 40°C to 120°C, significantly reducing energy consumption and equipment stress. The reaction demonstrates remarkable tolerance to various substituents on the isoflavone ring, enabling the synthesis of a diverse library of derivatives without the need for extensive optimization for each substrate. This streamlined workflow not only accelerates the R&D phase but also lays a solid foundation for cost reduction in API manufacturing by minimizing unit operations and waste generation.
Mechanistic Insights into Perfluoroalkylsulfonylation of Isoflavones
The core of this synthetic strategy relies on a nucleophilic substitution mechanism where the phenolic hydroxyl groups of the isoflavone act as nucleophiles. In the presence of potassium carbonate, the hydroxyl protons are abstracted to form phenoxide anions, which are significantly more reactive towards the sulfur center of the perfluoroalkylsulfonyl fluoride. The electron-withdrawing nature of the perfluoroalkyl chain enhances the electrophilicity of the sulfur atom, making it highly susceptible to attack even under relatively mild thermal conditions. This interaction results in the formation of a stable sulfonate ester linkage, effectively tethering the perfluoroalkyl chain to the isoflavone scaffold. The choice of DMF as a solvent is critical, as it stabilizes the ionic intermediates and ensures that the inorganic base remains sufficiently dispersed to drive the reaction to completion. The nitrogen protection atmosphere prevents moisture ingress, which could otherwise hydrolyze the sensitive sulfonyl fluoride reagent, thereby preserving reagent integrity and maximizing atom economy.
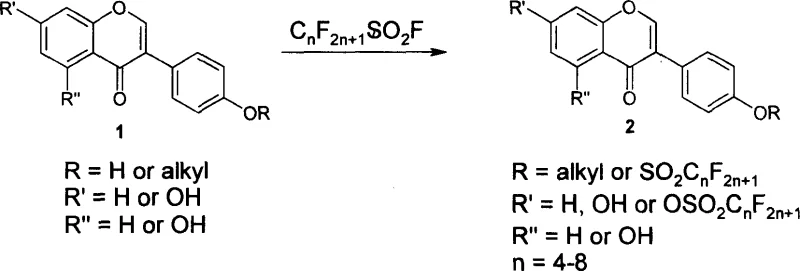
One of the most compelling aspects of this chemistry is its inherent regioselectivity, which is governed by the electronic and steric environment of the isoflavone ring system. When the substrate possesses a single hydroxyl group at the 7-position, the sulfonylation occurs exclusively at this site, yielding a single, well-defined product. However, in more complex scenarios where hydroxyl groups are present at both the 7 and 4' positions, or even at the 5, 7, and 4' positions, the reaction displays a distinct preference for the 4'-hydroxyl group. This selectivity is likely driven by the higher nucleophilicity of the 4'-phenoxide compared to the 7-phenoxide, possibly due to conjugation effects with the carbonyl group at the 4-position of the isoflavone core. Such predictable selectivity is invaluable for impurity control, as it minimizes the formation of regioisomeric by-products that would otherwise complicate downstream purification. This level of control ensures that the final high-purity pharmaceutical intermediates meet the stringent specifications required for clinical applications, reducing the burden on quality control laboratories.
How to Synthesize Perfluoroalkyl Sulfonyl Isoflavone Efficiently
Implementing this synthesis on a production scale requires careful attention to reagent quality and process parameters to ensure consistent outcomes. The protocol begins with the dissolution of high-purity hydroxyisoflavone in anhydrous DMF, followed by the addition of dried potassium carbonate to generate the reactive phenoxide species. Once the mixture is homogenized, the perfluoroalkylsulfonyl fluoride is introduced under an inert atmosphere, and the temperature is carefully ramped to the optimal range of 50°C to 70°C. Maintaining this temperature window is crucial for balancing reaction rate with the stability of the perfluoroalkyl chain. Following the reaction period, the workup involves a straightforward quench into ice water, which precipitates the product due to its reduced solubility in the aqueous medium. The detailed standardized synthesis steps, including specific molar ratios and purification techniques, are outlined in the guide below to assist technical teams in replicating this high-value process.
- Dissolve 97% hydroxyisoflavone in dry DMF and add dry K2CO3 powder, stirring at room temperature for 30 minutes to activate the phenolic hydroxyl groups.
- Under nitrogen protection, add perfluoroalkylsulfonyl fluoride to the mixture and heat to 40-120°C, maintaining the reaction for 2 to 24 hours depending on substrate reactivity.
- Cool the reaction mixture, pour into ice water, filter the precipitate, wash with water, vacuum dry, and recrystallize from ethanol to obtain the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial benefits for procurement managers and supply chain heads looking to optimize their sourcing strategies for oncology intermediates. The primary advantage stems from the use of readily available starting materials; hydroxyisoflavones are accessible from natural sources or via established synthetic routes, ensuring a stable supply base that is not subject to the volatility of exotic reagents. Furthermore, the reaction conditions are mild and do not require specialized high-pressure or cryogenic equipment, which translates to lower capital expenditure for manufacturing partners. The simplicity of the workup procedure, involving basic filtration and recrystallization, significantly reduces processing time and solvent consumption compared to more complex chromatographic purifications. These factors collectively contribute to a more resilient supply chain capable of meeting the demanding timelines of pharmaceutical development projects without compromising on quality or compliance.
- Cost Reduction in Manufacturing: The elimination of complex catalytic systems and the use of inexpensive inorganic bases like potassium carbonate drastically simplify the cost structure of the synthesis. By avoiding the need for expensive transition metal catalysts, the process removes the necessity for costly metal scavenging steps, which are often a significant portion of the downstream processing budget. The high atom economy of the sulfonylation reaction ensures that a large proportion of the input mass is converted into the desired product, minimizing waste disposal costs. Additionally, the ability to use standard stainless steel reactors rather than specialized lined vessels further reduces the barrier to entry for contract manufacturing organizations, fostering a competitive bidding environment that drives down overall production costs for the final API intermediate.
- Enhanced Supply Chain Reliability: The robustness of this chemical transformation enhances supply chain reliability by reducing the risk of batch failures due to sensitive reaction parameters. Since the reaction tolerates a range of temperatures and times without significant degradation of the product, manufacturing partners have greater flexibility in scheduling and resource allocation. The use of common solvents like DMF, which are widely stocked by chemical suppliers, mitigates the risk of raw material shortages that can plague projects relying on niche reagents. This stability allows for better forecasting and inventory management, ensuring that critical milestones in drug development are met without delay. For supply chain heads, this means a lower risk profile and a more predictable flow of materials from the synthesis site to the formulation unit.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of exothermic hazards or gas evolution that typically complicate scale-up. The reaction mixture remains homogeneous or easily manageable throughout the process, facilitating efficient heat transfer and mixing in large-scale reactors. From an environmental standpoint, the process generates minimal hazardous waste, as the primary by-products are inorganic salts that can be easily treated or disposed of according to standard regulations. The reduced solvent usage and energy requirements align with green chemistry principles, helping pharmaceutical companies meet their sustainability goals. This environmental compatibility simplifies the regulatory approval process for new manufacturing sites, accelerating the time to market for new therapeutic candidates based on this scaffold.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of perfluoroalkyl sulfonyl isoflavones. These answers are derived directly from the technical specifications and experimental data provided in the patent literature to ensure accuracy and relevance for industry professionals. Understanding these details is essential for making informed decisions about technology transfer and procurement.
Q: What is the regioselectivity of the perfluoroalkylsulfonylation reaction on isoflavones?
A: The reaction exhibits high regioselectivity based on the number of hydroxyl groups. For isoflavones with a single hydroxyl at the 7-position, substitution occurs exclusively at the 7-position. However, for substrates containing hydroxyl groups at both the 7 and 4' positions, or at the 7, 4', and 5 positions, the perfluoroalkylsulfonylation selectively targets the 4'-hydroxyl group, ensuring consistent product profiles.
Q: Why is the perfluoroalkylsulfonyl group introduced into isoflavone structures?
A: Introducing perfluoroalkylsulfonyl groups significantly enhances the biological and physicochemical properties of isoflavones. Unlike simple trifluoromethyl groups which may not significantly improve solubility or activity, the perfluoroalkylsulfonyl moiety provides strong electron-withdrawing effects and lipophilicity, resulting in potent inhibitory activity against HL-60 human leukemia cell lines, making them valuable lead compounds for oncology.
Q: What are the optimal reaction conditions for maximizing yield in this synthesis?
A: According to patent data, optimal conditions involve using dry N,N-dimethylformamide (DMF) as the solvent and potassium carbonate (K2CO3) as the base. The reaction temperature should be maintained between 50°C and 70°C, with a reaction time of approximately 5 to 7 hours. These parameters balance reaction kinetics with product stability to achieve satisfactory yields.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Perfluoroalkyl Sulfonyl Isoflavone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation anticancer therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent to plant is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify every batch. Our expertise in fluorine chemistry and heterocyclic synthesis positions us as an ideal partner for executing the complex transformations required for perfluoroalkyl sulfonyl isoflavones. By leveraging our technical capabilities, clients can accelerate their development timelines while maintaining the highest standards of quality and regulatory compliance.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific project requirements. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain. Whether you require small quantities for preclinical studies or large volumes for commercial launch, NINGBO INNO PHARMCHEM is equipped to support your journey from molecule to medicine with reliability and precision.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
