Advanced Synthesis of 3,5-Dihalo Trifluoroacetophenone Derivatives for Industrial Scale
Advanced Synthesis of 3,5-Dihalo Trifluoroacetophenone Derivatives for Industrial Scale
The pharmaceutical and agrochemical industries constantly demand robust, scalable synthetic routes for complex intermediates, particularly those containing electron-withdrawing groups that traditionally hinder functionalization. Patent CN112110804B, published in early 2023, addresses a critical gap in the synthesis of 3,5-dihalo trifluoroacetophenone and its derivatives, compounds that serve as pivotal building blocks for various bioactive molecules. This technology offers a transformative approach by bypassing the severe electronic deactivation typically caused by the trifluoroacetyl and halogen groups, which usually render the para-position of the benzene ring inert to standard electrophilic substitution. By leveraging a strategic sequence of reduction, halogenation, and diazo-substitution, this method enables the precise installation of diverse functional groups at the 4-position with exceptional efficiency. For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier, understanding the mechanistic elegance and commercial viability of this patent is essential for securing a competitive supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,5-dihalo-substituted trifluoroacetyl ketone compounds has been plagued by significant technical hurdles and economic inefficiencies. As detailed in the comparative examples of the patent data, conventional routes often rely on harsh organometallic chemistry, such as the use of tert-butyllithium at cryogenic temperatures of -78°C. This approach not only demands specialized, energy-intensive equipment capable of maintaining ultra-low temperatures but also suffers from poor atom economy and safety risks associated with pyrophoric reagents. Furthermore, the strong electron-withdrawing nature of the trifluoroacetyl group severely limits the scope of subsequent modifications, making it nearly impossible to introduce substituents at the para-position after the initial framework is established. The comparative data highlights a stark reality: traditional lithiation methods yield merely 38.4% of the target product, rendering them commercially unviable for large-scale manufacturing where cost reduction in pharma intermediates manufacturing is a primary directive.
The Novel Approach
In contrast, the methodology disclosed in CN112110804B presents a sophisticated yet operationally simple three-step cascade that circumvents these electronic limitations entirely. The process initiates with the reduction of a readily available 3-halogen-4-nitro trifluoroacetophenone, transforming the deactivating nitro group into a powerful ortho/para-directing amino group. This strategic inversion of electronic properties allows for the subsequent halogenation to occur selectively at the position ortho to the amino group, effectively constructing the 3,5-dihalo pattern. Finally, the amino group is removed or substituted via diazotization, leaving behind the desired 4-substituted 3,5-dihalo scaffold. This route operates under mild conditions, utilizing common solvents like ethanol and toluene, and avoids the need for cryogenic cooling, thereby drastically simplifying the engineering requirements for commercial scale-up of complex polymer additives or pharmaceutical precursors.
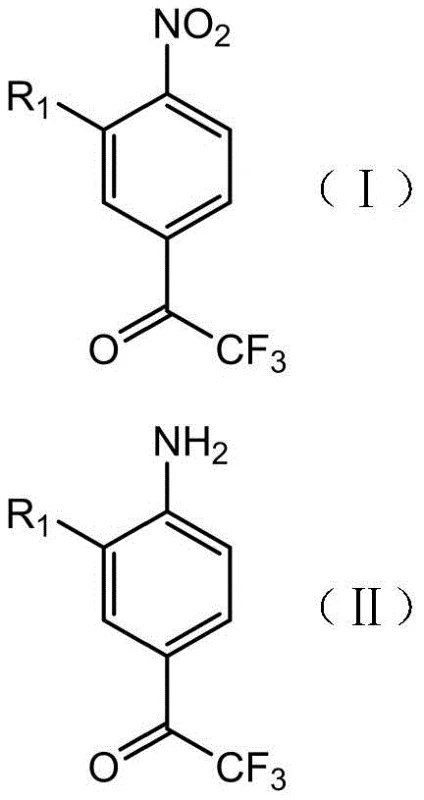
Mechanistic Insights into Catalytic Reduction and Diazo-Substitution
The core innovation of this synthesis lies in the clever manipulation of substituent effects through the temporary installation of an amino group. In Step S1, the nitro group of the starting material is reduced to an amine using heterogeneous catalysts such as platinum carbon, palladium carbon, or Raney nickel under hydrogen pressure. This transformation is critical because the resulting amino group acts as a strong activator, overcoming the deactivating influence of the trifluoroacetyl moiety and facilitating the subsequent electrophilic attack in Step S2. The halogenation step utilizes reagents like sulfuryl chloride or N-chlorosuccinimide (NCS) to introduce the second halogen atom specifically at the position ortho to the amino group, ensuring high regioselectivity. This mechanistic pathway ensures that the difficult 3,5-dihalo pattern is constructed with precision, avoiding the formation of isomeric impurities that are notoriously difficult to separate in fluorochemical synthesis.
Furthermore, the final substitution step (S3) demonstrates remarkable versatility through diazonium chemistry. By converting the amino group into a diazonium salt using nitrous acid reagents like sodium nitrite, the process opens multiple pathways for functionalization depending on the reaction conditions and copper salts employed. For instance, using hypophosphorous acid and cuprous oxide allows for reductive deamination to yield the unsubstituted 3,5-dihalo derivative, while the use of cuprous chloride or cuprous bromide facilitates the Sandmeyer reaction to install chlorine or bromine atoms respectively. Even the introduction of a hydroxyl group is achievable by heating the diazonium intermediate in acidic copper sulfate solutions. This modularity means that a single intermediate (Compound III) can be diverged into multiple high-value products, maximizing the utility of the production line and reducing lead time for high-purity specialty chemicals.
How to Synthesize 3,5-Dihalo Trifluoroacetophenone Efficiently
The practical implementation of this synthesis requires careful control of reaction parameters to maximize yield and purity, as evidenced by the extensive experimental data provided in the patent. The process begins with the dissolution of the nitro-starting material in a solvent such as ethanol or toluene, followed by catalytic hydrogenation at moderate temperatures (40-50°C) and pressures (0.1-1.0 MPa). Following filtration of the catalyst, the crude amine is subjected to halogenation, where temperature control is vital to prevent poly-halogenation; typically, reactions are maintained between 20°C and 70°C depending on the halogenating agent used. The final diazotization step requires low temperatures (-10°C to 0°C) to stabilize the diazonium intermediate before warming to effect substitution. For a detailed breakdown of the specific molar ratios, solvent choices, and workup procedures optimized for different derivatives, please refer to the standardized synthesis guide below.
- Perform catalytic hydrogenation on 3-halogen-4-nitro trifluoroacetophenone using Pd/C or Raney Nickel to reduce the nitro group to an amino group.
- Conduct electrophilic halogenation on the resulting amine using agents like sulfuryl chloride or NCS to introduce a second halogen ortho to the amino group.
- Execute diazotization followed by substitution (Sandmeyer-type reaction) using cuprous salts to replace the amino group with hydrogen, halogen, or hydroxyl groups.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this preparation method offers profound advantages in terms of cost stability and operational reliability. The elimination of cryogenic conditions and pyrophoric reagents significantly reduces the capital expenditure required for reactor infrastructure, allowing manufacturers to utilize standard glass-lined or stainless steel vessels without specialized cooling jackets. Moreover, the raw materials, such as 3-halogen-4-nitro trifluoroacetophenone, are either commercially available byproducts or easily synthesized, ensuring a robust upstream supply chain that is less susceptible to market volatility. The use of conventional solvents like ethanol, toluene, and acetonitrile further simplifies solvent recovery and recycling processes, contributing to a greener manufacturing profile that aligns with increasingly stringent environmental regulations.
- Cost Reduction in Manufacturing: The transition from organolithium chemistry to catalytic hydrogenation and diazotization eliminates the need for expensive, moisture-sensitive reagents and the energy-intensive maintenance of -78°C environments. This shift results in substantial cost savings by lowering utility consumption and reducing the complexity of waste treatment, as the byproducts are primarily inorganic salts that are water-soluble and easily separated from the organic phase.
- Enhanced Supply Chain Reliability: By utilizing widely available catalysts like Pd/C and Raney Nickel, along with commodity chemicals like sodium nitrite and sulfuric acid, the process mitigates the risk of supply disruptions associated with specialized reagents. The robustness of the reaction conditions ensures consistent batch-to-batch quality, which is critical for maintaining long-term contracts with downstream pharmaceutical clients who require rigorous adherence to purity specifications.
- Scalability and Environmental Compliance: The patent data explicitly confirms the scalability of this method through 10-fold scale-up experiments (Examples 16, 17, 22, 23) which maintained high yields and purity levels comparable to laboratory scale. The aqueous workup procedures facilitate the removal of impurities without generating hazardous organic waste streams, simplifying the environmental compliance burden and enabling smoother permitting for large-scale production facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology, derived directly from the experimental findings and comparative analysis within the document. These insights are intended to clarify the operational benefits and chemical versatility of the described synthesis route for potential partners and technical evaluators.
Q: What are the advantages of this new preparation method over traditional lithiation routes?
A: Unlike traditional methods requiring cryogenic temperatures (-78°C) and expensive organolithium reagents which yield only 38.4%, this patent utilizes mild catalytic hydrogenation and diazotization, achieving yields over 90% with significantly lower energy consumption and operational costs.
Q: Can this process be scaled for commercial manufacturing?
A: Yes, the patent explicitly demonstrates successful scale-up experiments (Examples 16, 17, 22, 23) where material quantities were increased tenfold without compromising yield or purity, proving its viability for metric-ton scale production.
Q: What specific substituents can be introduced at the 4-position?
A: Through varying the diazotization and substitution conditions in Step S3, the process allows for the introduction of hydrogen, fluorine, chlorine, bromine, or hydroxyl groups at the 4-position, providing versatile access to diverse chemical scaffolds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,5-Dihalo Trifluoroacetophenone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced intermediate technologies like CN112110804B in driving innovation for our global partners. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant manufacturing environment. Our state-of-the-art facilities are equipped to handle the specific catalytic hydrogenation and diazotization steps safely and efficiently, supported by rigorous QC labs that enforce stringent purity specifications to meet the exacting standards of the pharmaceutical and agrochemical sectors.
We invite you to collaborate with us to leverage this cutting-edge synthesis route for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this method can optimize your bill of materials. Please contact us today to request specific COA data for our existing inventory or to discuss route feasibility assessments for your custom synthesis needs, ensuring a seamless integration of this high-performance intermediate into your supply chain.
