Revolutionizing Alminoprofen Intermediate Production: A Safer Two-Step Synthetic Route for Global Supply Chains
Introduction to Advanced Intermediate Synthesis
The pharmaceutical industry continuously seeks robust manufacturing pathways for nonsteroidal anti-inflammatory drug (NSAID) precursors, particularly for complex aromatic structures like those found in alminoprofen synthesis. Patent CN113135831A introduces a transformative technical route for preparing ethyl 2-(4-amino-phenyl) propionate, a critical building block that addresses longstanding inefficiencies in traditional four-step syntheses. This innovation focuses on telescoping reaction steps and replacing hazardous reagents, thereby offering a compelling value proposition for reliable alminoprofen intermediate suppliers aiming to optimize their production portfolios. By integrating alkylation and hydrolysis into a single operational sequence and utilizing a safer catalytic reduction system, the technology not only enhances throughput but also aligns with modern green chemistry principles essential for sustainable cost reduction in pharmaceutical intermediates manufacturing.
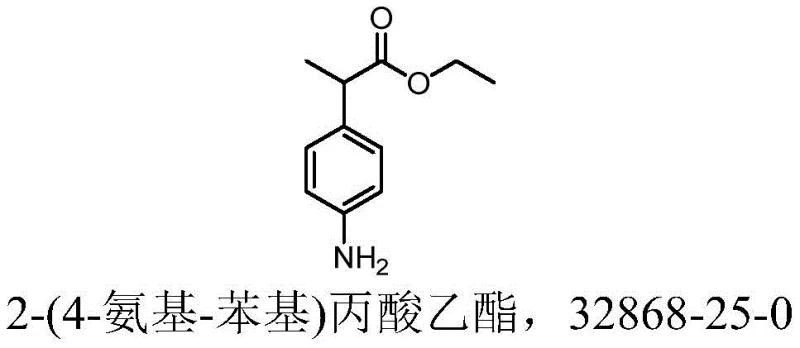
The strategic importance of this intermediate lies in its role as the direct precursor to alminoprofen, a potent analgesic and anti-inflammatory agent. Traditional methods often suffer from prolonged cycle times and significant safety liabilities due to the use of pyrophoric catalysts. The disclosed methodology mitigates these risks by employing a ferric chloride-hydrazine hydrate system, which operates under milder conditions while maintaining high conversion rates. For procurement and supply chain leaders, understanding these mechanistic shifts is vital, as they directly translate to improved commercial scale-up of complex aromatic amino esters and more predictable delivery schedules for global API manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ethyl 2-(4-amino-phenyl) propionate has relied on a cumbersome four-step sequence that introduces multiple points of failure and inefficiency. The initial step typically involves the reaction of p-fluoronitrobenzene with diethyl malonate in dimethyl sulfoxide (DMSO), necessitating the use of powdered potassium carbonate which creates a highly viscous sludge that is notoriously difficult to filter on an industrial scale. Following this, the intermediate undergoes hydrolysis, esterification, and finally reduction using active nickel under hydrogen pressure. This conventional pathway is plagued by excessive unit operations, including repeated concentration, extraction, and pH adjustments that consume vast quantities of hydrochloric acid and generate substantial volumes of saline wastewater. Furthermore, the reliance on active nickel in the final reduction step presents a severe safety hazard, as the material is spontaneously flammable upon exposure to air, requiring specialized handling protocols that increase operational costs and insurance liabilities for chemical plants.
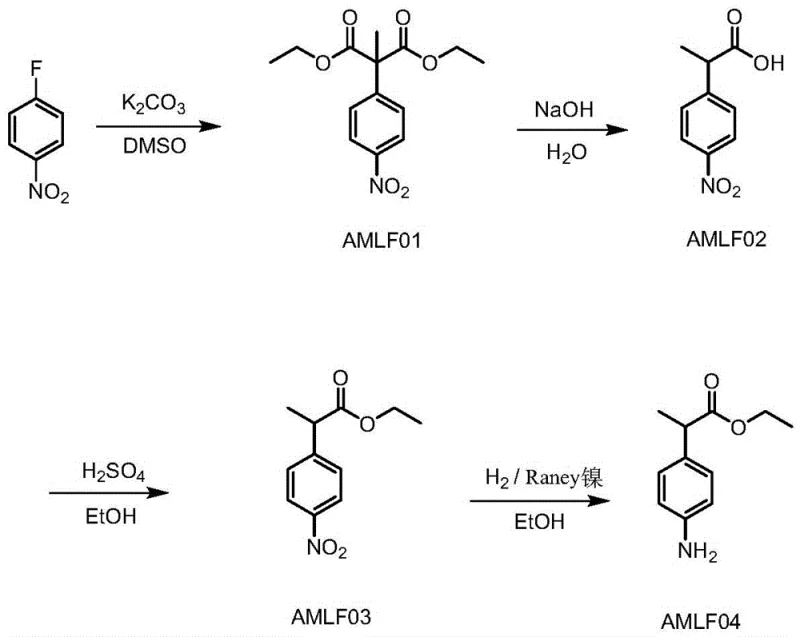
The Novel Approach
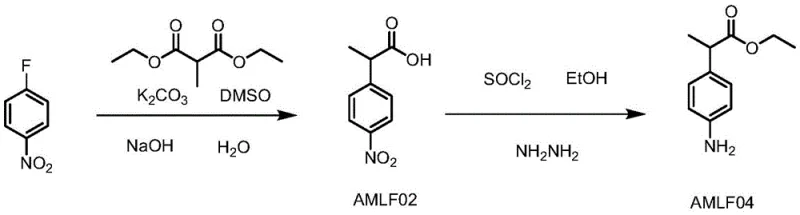
In stark contrast, the innovative process described in the patent streamlines the synthesis into two primary stages, effectively telescoping the alkylation and hydrolysis reactions to eliminate the problematic filtration of inorganic salts. By reacting p-fluoronitrobenzene directly with diethyl methylmalonate in the presence of anhydrous alkali, followed by immediate alkaline hydrolysis in the same vessel, the process allows for direct phase separation of the high-salt aqueous layer, bypassing the need for filtering solid potassium carbonate entirely. The subsequent stage combines esterification with thionyl chloride and a novel catalytic reduction using hydrazine hydrate and ferric chloride. This approach not only reduces the total reaction time significantly but also minimizes the consumption of neutralizing acids, leading to a drastic reduction in the treatment difficulty of three wastes. The elimination of extraction steps between the esterification and reduction phases further simplifies the workflow, making it an ideal candidate for reducing lead time for high-purity pharmaceutical intermediates in a competitive market.
Mechanistic Insights into FeCl3-Catalyzed Reduction
The core chemical innovation in this pathway lies in the replacement of heterogeneous hydrogenation with a homogeneous catalytic transfer hydrogenation system utilizing ferric chloride and hydrazine hydrate. In this mechanism, hydrazine serves as the hydrogen donor, decomposing on the surface of the in situ generated iron species to release nascent hydrogen which selectively reduces the nitro group to an amine without affecting the ester functionality. The presence of activated carbon in the catalyst mixture plays a crucial dispersing role, preventing the aggregation of iron particles and ensuring uniform contact with the substrate in the ethanol solvent. This catalytic cycle operates efficiently at temperatures between 70-80°C, providing a kinetic profile that matches or exceeds that of traditional high-pressure hydrogenation while operating at atmospheric pressure. The selectivity of this system is paramount, as it avoids the over-reduction or hydrogenolysis of the benzylic position that can sometimes occur with aggressive nickel catalysts, thereby preserving the structural integrity of the propionate side chain.
From an impurity control perspective, the telescoped alkylation-hydrolysis step offers distinct advantages by minimizing the exposure of the intermediate to harsh acidic conditions prior to the final workup. In traditional routes, multiple pH swings can lead to the formation of decarboxylation byproducts or polymeric tars that are difficult to remove. By maintaining a controlled alkaline environment during the hydrolysis and utilizing a mild acidification strategy only when necessary for product extraction, the new process limits the formation of these degradation products. Additionally, the use of dichloromethane or ethyl acetate for selective impurity extraction prior to final acidification ensures that non-polar organic impurities are removed early in the sequence. This rigorous control over the reaction environment results in a crude product purity that consistently exceeds 98.5%, reducing the burden on downstream purification units and ensuring that the final high-purity ethyl 2-(4-amino-phenyl) propionate meets the strict specifications required for GMP-grade API production.
How to Synthesize Ethyl 2-(4-Amino-Phenyl) Propionate Efficiently
The implementation of this synthesis requires precise control over stoichiometry and temperature profiles to maximize the benefits of the telescoped design. The process begins with the nucleophilic aromatic substitution of p-fluoronitrobenzene, where the choice of solvent (preferably DMSO) and base (powdered potassium carbonate) is critical for driving the alkylation to completion before the introduction of the hydrolysis agent. Following the alkylation, the direct addition of sodium hydroxide solution initiates the hydrolysis of the diester to the mono-acid, which is then isolated via phase separation rather than filtration. The second stage involves converting this acid to the ethyl ester using thionyl chloride, followed immediately by the reduction step without isolating the nitro-ester intermediate. Detailed standardized operating procedures regarding reagent addition rates, exotherm management, and workup protocols are essential for safe scale-up.
- React p-fluoronitrobenzene with diethyl methylmalonate in DMSO using potassium carbonate, followed by direct alkaline hydrolysis to obtain AMLF02 without filtration.
- Perform esterification of AMLF02 with thionyl chloride in ethanol, followed by reduction using hydrazine hydrate and a ferric chloride catalyst to yield AMLF04.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers tangible strategic benefits that extend beyond simple yield improvements. The most significant advantage is the enhancement of operational safety, which directly correlates to reduced insurance premiums and lower regulatory compliance burdens. By eliminating the use of pyrophoric active nickel, facilities can avoid the costly infrastructure requirements associated with high-pressure hydrogenation units and specialized fire suppression systems. This shift allows for more flexible manufacturing scheduling and reduces the risk of unplanned shutdowns due to safety incidents, thereby ensuring a more consistent supply of critical intermediates to downstream API producers. Furthermore, the reduction in unit operations translates to lower utility consumption, as fewer distillation and concentration cycles are required, contributing to a leaner and more cost-effective manufacturing footprint.
- Cost Reduction in Manufacturing: The elimination of the filtration step for potassium carbonate and the reduction in acid consumption for pH adjustment lead to substantial savings in raw material costs and waste disposal fees. By avoiding the use of expensive Raney nickel catalysts and the associated safety handling costs, the overall cost of goods sold (COGS) is significantly optimized. The telescoping of reaction steps also reduces labor hours and equipment occupancy time, allowing for higher throughput within existing facility constraints without the need for capital-intensive expansion.
- Enhanced Supply Chain Reliability: The simplified process flow reduces the number of potential bottlenecks, such as slow filtration or complex extractions, which often cause delays in batch release. The use of readily available reagents like hydrazine hydrate and ferric chloride, which are stable and easy to source globally, mitigates the risk of supply disruptions associated with specialized catalysts. This reliability ensures that production timelines are met consistently, fostering stronger partnerships with API manufacturers who depend on just-in-time delivery models for their own production schedules.
- Scalability and Environmental Compliance: The drastic reduction in wastewater volume and organic solvent waste simplifies the environmental permitting process and lowers the cost of effluent treatment. The process is inherently scalable, as the removal of solid filtration steps eliminates one of the primary challenges in moving from pilot plant to commercial tonnage production. This scalability, combined with a greener chemical profile, positions manufacturers to meet increasingly stringent environmental regulations while maintaining competitive pricing structures in the global marketplace.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis route. These insights are derived directly from the comparative data and experimental examples provided in the patent literature, offering a transparent view of the technology's capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this method for large-scale production of alminoprofen intermediates.
Q: How does the new process improve safety compared to traditional Raney Nickel reduction?
A: The novel route replaces pyrophoric active nickel with a safer hydrazine hydrate and ferric chloride catalytic system, eliminating the high fire risk associated with handling active nickel in large-scale hydrogenation reactors.
Q: What are the purity specifications achievable with this synthetic method?
A: Experimental data indicates that the optimized process consistently yields the final intermediate with a purity exceeding 98.5%, meeting stringent requirements for downstream API synthesis without extensive recrystallization.
Q: Does this method reduce environmental waste generation?
A: Yes, by omitting the filtration of solid potassium carbonate and reducing the amount of acid required for pH adjustment during workup, the process significantly lowers the volume of wastewater and organic solvent waste per kilogram of product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ethyl 2-(4-Amino-Phenyl) Propionate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer and more efficient synthetic routes is critical for the long-term sustainability of the pharmaceutical supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the FeCl3-catalyzed reduction of nitro-aromatics are translated into robust, GMP-compliant manufacturing processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of ethyl 2-(4-amino-phenyl) propionate meets the exacting standards required for NSAID synthesis, providing our partners with the confidence needed to streamline their own API production lines.
We invite global pharmaceutical companies and contract manufacturers to engage with our technical procurement team to discuss how this optimized route can be integrated into your supply network. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the specific economic benefits applicable to your operation. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to delivering high-quality intermediates through cutting-edge chemical engineering.
