Advanced Two-Step Synthesis of 2,6-Dichloro-4-Trifluoromethylaniline for Scalable Agrochemical Production
Advanced Two-Step Synthesis of 2,6-Dichloro-4-Trifluoromethylaniline for Scalable Agrochemical Production
The global demand for high-performance insecticides continues to drive innovation in the synthesis of critical agrochemical intermediates, specifically within the phenylpyrazole class. A pivotal development in this sector is detailed in Chinese Patent CN110845340B, which discloses a highly efficient preparation method for 2,6-dichloro-4-trifluoromethylaniline, a key building block for the synthesis of fipronil. This patent represents a significant technological leap forward by addressing long-standing issues related to reaction safety, raw material availability, and product purity that have historically plagued the industrial production of this compound. By shifting away from hazardous high-pressure protocols and expensive fluorinated precursors, this new methodology offers a streamlined pathway that aligns perfectly with modern green chemistry principles and cost-effective manufacturing strategies. For R&D directors and supply chain managers alike, understanding the nuances of this two-step chlorination and fluorination sequence is essential for securing a reliable agrochemical intermediate supplier capable of meeting stringent quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to synthesizing 2,6-dichloro-4-trifluoromethylaniline have been fraught with significant technical and economic hurdles that hinder large-scale industrialization. Prior art, such as the methods described in US5401882A and CN1436769A, relies on 2-chloro-4-trifluoromethyl-N,N-dimethylaniline as a starting material, which is not only difficult to source but also necessitates complex one-pot chlorination and demethylation reactions that generate substantial byproducts. Furthermore, alternative routes like those found in CN101289401B require the direct introduction of ammonia gas under extreme pressures exceeding 10MPa, creating severe safety risks and demanding specialized, high-cost pressure vessels that are not readily available in standard fine chemical facilities. Other methods utilizing gaseous trifluorobromotrifluoromethane, as seen in CN104072379B, introduce aggressive corrosion challenges that degrade standard metal reactor linings, leading to frequent maintenance downtime and potential contamination of the product stream with heavy metals. These conventional pathways collectively suffer from low selectivity, difficult purification processes, and yields that often struggle to meet the rigorous demands of modern pharmaceutical and agrochemical manufacturing.
The Novel Approach
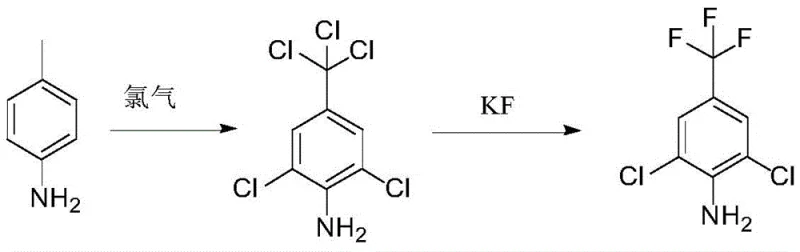
In stark contrast to these cumbersome legacy methods, the technology outlined in CN110845340B introduces a brand-new preparation strategy that fundamentally simplifies the synthetic architecture while enhancing overall process robustness. This novel approach begins with 4-methylaniline, an inexpensive and abundantly available commodity chemical, effectively bypassing the need for costly pre-fluorinated starting materials that traditionally inflate production costs. The process employs a controlled radical chlorination to install both the ring chlorines and the trichloromethyl group in a single operational step, followed by a nucleophilic fluorination that cleanly converts the trichloromethyl moiety into the desired trifluoromethyl group. This logical disconnection of the synthesis allows for milder reaction conditions, typically operating between 30°C and 120°C without the need for ultra-high pressure equipment, thereby drastically reducing capital expenditure requirements for plant infrastructure. The clarity of the reaction mechanism and the minimization of side reactions ensure that the final product is obtained with exceptional purity, facilitating easier downstream processing and ensuring consistent quality for the end-user.
Mechanistic Insights into Radical Chlorination and Halex Fluorination
The first stage of this synthesis involves a sophisticated free-radical chlorination mechanism where 4-methylaniline is subjected to chlorine gas in the presence of a radical initiator such as azodiisobutyronitrile (AIBN) or dibenzoyl peroxide. Under these conditions, the methyl group on the aniline ring undergoes exhaustive chlorination to form a trichloromethyl group, while the ortho positions on the aromatic ring are simultaneously chlorinated to yield 2,6-dichloro-4-trichloromethylaniline. The choice of solvent, typically an inert medium like dichloroethane or carbon tetrachloride, plays a critical role in stabilizing the radical intermediates and ensuring that the reaction proceeds with high regioselectivity to prevent over-chlorination or ring degradation. The use of a catalytic amount of radical initiator ensures a steady generation of chlorine radicals, maintaining a consistent reaction rate that can be precisely monitored via HPLC to stop the reaction once the starting material conversion exceeds 98%, thus preventing the formation of unwanted polychlorinated impurities.
Following the chlorination, the second stage employs a classic halogen exchange (Halex) reaction mechanism to transform the trichloromethyl group into the target trifluoromethyl functionality. This nucleophilic substitution is facilitated by the use of potassium fluoride or cesium fluoride in a polar aprotic solvent such as DMF or DMSO, which enhances the nucleophilicity of the fluoride ion by reducing solvation effects. The addition of a phase transfer catalyst like tetrabutylammonium bromide or a nitrogen-based catalyst like triethylenediamine is crucial for shuttling the fluoride anion into the organic phase where the reaction occurs, significantly accelerating the reaction kinetics. This step proceeds through a concerted mechanism where the strong carbon-fluorine bonds are formed at the expense of the weaker carbon-chlorine bonds, driven thermodynamically by the precipitation of insoluble chloride salts. The result is a clean conversion that preserves the integrity of the sensitive aniline nitrogen and the aryl chlorides, yielding the final 2,6-dichloro-4-trifluoromethylaniline with minimal structural degradation.
How to Synthesize 2,6-Dichloro-4-Trifluoromethylaniline Efficiently
Implementing this synthesis requires careful attention to reaction parameters to maximize yield and safety, particularly regarding the control of exothermic chlorination and the moisture sensitivity of the fluorination step. The patent provides a comprehensive framework for executing these transformations, emphasizing the importance of solvent selection and catalyst loading to achieve reproducible results on a commercial scale. Operators must ensure that the chlorination step is conducted with precise temperature control to manage the evolution of hydrogen chloride gas, while the subsequent fluorination requires rigorous drying of reagents to prevent hydrolysis of the fluoride source. The following guide outlines the standardized operational procedure derived from the patent examples, serving as a foundational protocol for process chemists aiming to replicate this high-efficiency route.
- Perform radical chlorination on 4-methylaniline using chlorine gas and a radical initiator like AIBN in an inert solvent to form 2,6-dichloro-4-trichloromethylaniline.
- Conduct a nucleophilic fluorination reaction on the trichloromethyl intermediate using potassium fluoride and a phase transfer catalyst in a polar aprotic solvent.
- Purify the final product through decompression desolventizing and high vacuum distillation to achieve purity greater than 99%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic benefits that extend far beyond simple chemical yield improvements. By eliminating the reliance on exotic, high-pressure reaction conditions and corrosive gaseous reagents, manufacturers can utilize standard glass-lined steel reactors that are already prevalent in most fine chemical production facilities, thereby avoiding the need for massive capital investment in specialized high-pressure autoclaves. This compatibility with existing infrastructure significantly reduces the barrier to entry for production and allows for rapid scale-up from pilot plant to commercial tonnage without the lengthy lead times associated with custom equipment fabrication. Furthermore, the use of commodity-grade starting materials like 4-methylaniline and inorganic fluoride salts insulates the supply chain from the volatility often seen in the market for specialized fluorinated building blocks, ensuring a more stable and predictable cost structure over the long term.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior because it replaces expensive pre-fluorinated raw materials with cheap, bulk-available 4-methylaniline, resulting in a drastic reduction in direct material costs per kilogram of product. Additionally, the elimination of high-pressure equipment and corrosive gas handling systems lowers both the depreciation costs of the plant and the ongoing maintenance expenses related to reactor lining repairs and safety monitoring. The high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, which reduces the consumption of solvents and energy during the purification and crystallization stages, further driving down the overall cost of goods sold. This holistic approach to cost optimization ensures that the final intermediate can be priced competitively while maintaining healthy margins for the manufacturer.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved as the key reagents, including chlorine gas and potassium fluoride, are produced globally in massive quantities for various industrial applications, reducing the risk of supply disruptions caused by niche vendor bottlenecks. The mild reaction conditions also mean that production is less susceptible to shutdowns caused by equipment failure or safety interlocks triggered by extreme operating parameters, leading to higher plant uptime and more consistent delivery schedules. By simplifying the synthetic route to just two main chemical transformations, the process reduces the number of unit operations required, which inherently decreases the probability of logistical delays or quality deviations occurring between steps. This reliability is critical for downstream customers who depend on a steady flow of high-quality intermediates to maintain their own continuous manufacturing campaigns.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, as the exothermic nature of the chlorination can be easily managed through controlled dosing in large-scale reactors, and the fluorination step operates at temperatures that are easily achievable with standard heating media. From an environmental perspective, the avoidance of toxic gaseous fluorinating agents and high-pressure ammonia significantly reduces the facility's environmental footprint and simplifies the permitting process for waste gas treatment systems. The use of recyclable polar aprotic solvents in the second step allows for effective solvent recovery loops, minimizing liquid waste generation and aligning with increasingly strict global regulations on volatile organic compound emissions. This combination of operational safety and environmental stewardship makes the technology highly attractive for production in regions with rigorous regulatory oversight.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, drawing directly from the comparative data and experimental results presented in the patent documentation. These insights are intended to clarify the operational advantages and resolve potential concerns regarding the transition from older, less efficient methods to this state-of-the-art protocol. Understanding these details is vital for technical teams evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: What are the primary safety advantages of this new synthesis method compared to prior art?
A: Unlike previous methods requiring high-pressure ammonia introduction (above 10MPa) or corrosive gaseous fluorinating agents, this process operates at atmospheric or low pressure using solid potassium fluoride, significantly reducing equipment corrosion and explosion risks.
Q: How does the raw material cost compare to traditional trifluoromethylation routes?
A: This route utilizes 4-methylaniline, a widely available and inexpensive commodity chemical, avoiding the need for expensive pre-fluorinated starting materials like 4-trifluoromethyl aniline which often exceed the cost of the final product.
Q: What purity levels can be achieved with this chlorination-fluorination sequence?
A: The optimized process conditions, including specific catalyst loading and vacuum distillation purification, consistently deliver product purity exceeding 99.0%, minimizing downstream purification burdens for API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,6-Dichloro-4-Trifluoromethylaniline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a supply chain for high-value agrochemical intermediates that balances technical excellence with commercial viability. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from laboratory validation to full-scale industrial output. Our facilities are equipped with the necessary corrosion-resistant reactors and advanced distillation columns required to execute the chlorination and fluorination steps described in CN110845340B, adhering to stringent purity specifications that exceed 99% to support your downstream synthesis needs. With our rigorous QC labs and commitment to process safety, we provide a secure foundation for your long-term manufacturing strategy.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific volume requirements and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this method compared to your current supply sources. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will enhance the competitiveness and resilience of your agrochemical product portfolio.
