Advanced Synthesis of Alpha-Substituted Diethyl Malonate Derivatives for Commercial Scale-Up
Advanced Synthesis of Alpha-Substituted Diethyl Malonate Derivatives for Commercial Scale-Up
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable, and environmentally compliant synthetic routes for critical building blocks like alpha-substituted diethyl malonate derivatives. A significant technological breakthrough in this domain is detailed in patent CN102531897A, which outlines a novel two-step methodology that fundamentally shifts the paradigm away from hazardous traditional chemistries. This innovative approach utilizes readily commercialized substituted ethyl acetates and diethyl oxalate as initial feedstocks, undergoing a sequential condensation and oxidation protocol to yield high-purity target molecules. By leveraging mild reaction conditions ranging from 20°C to 45°C and avoiding the use of剧毒 cyanides or heavy metal catalysts, this process offers a compelling value proposition for reliable pharmaceutical intermediate supplier networks aiming to enhance their green chemistry credentials. The technical robustness of this method ensures stable process conditions and high conversion rates, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing where supply chain continuity and regulatory compliance are paramount concerns for global procurement teams.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of alpha-substituted diethyl malonate derivatives has been plagued by significant safety and environmental challenges inherent to legacy synthetic pathways. The most prevalent conventional method involves a cyaniding esterification process, which relies on the displacement of halogenated acetic acids with highly toxic sodium cyanide to generate cyanoacetic acid sodium intermediates. This route not only poses severe risks to human health and the environment due to the handling of cyanide but also necessitates complex and costly waste treatment protocols to neutralize toxic byproducts before discharge. Alternative methods such as catalyzed carbonylation require severe reaction conditions involving carbon monoxide and specialized catalysts, creating substantial barriers to safe industrial implementation and increasing capital expenditure for pressure-rated equipment. Furthermore, ester-interchange methods often employ heavy metal catalysts like dibutyltin oxide, which introduce the risk of metal contamination in the final active pharmaceutical ingredients (APIs) and drive up raw material costs due to the expense of removing trace metals to meet stringent regulatory specifications.
The Novel Approach
In stark contrast to these hazardous legacy processes, the novel approach described in the patent utilizes a benign condensation-oxidation sequence that dramatically simplifies the operational landscape for chemical manufacturers. By selecting 2-substituted acetic acid ethyl esters and diethyl oxalate as the primary building blocks, the process initiates with a base-catalyzed condensation in ether solvents to form a 2-substituted-3-oxo diethyl succinate intermediate. This intermediate is subsequently subjected to an oxidative cleavage using persulfates in an acidic medium, effectively transforming the succinate backbone into the desired malonate derivative without generating toxic cyanide waste or requiring heavy metal removal steps. 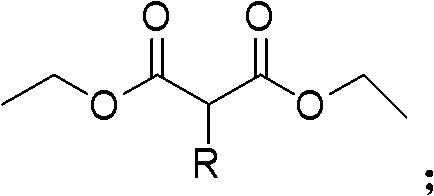 This streamlined two-step workflow operates under atmospheric pressure and moderate temperatures, significantly lowering the energy intensity of the production cycle. The elimination of toxic reagents and the use of commodity chemicals directly translates to enhanced supply chain reliability and reduced regulatory burden, positioning this technology as a superior choice for the commercial scale-up of complex pharmaceutical intermediates where safety and efficiency are non-negotiable requirements.
This streamlined two-step workflow operates under atmospheric pressure and moderate temperatures, significantly lowering the energy intensity of the production cycle. The elimination of toxic reagents and the use of commodity chemicals directly translates to enhanced supply chain reliability and reduced regulatory burden, positioning this technology as a superior choice for the commercial scale-up of complex pharmaceutical intermediates where safety and efficiency are non-negotiable requirements.
Mechanistic Insights into Condensation-Oxidation Cascade
The core of this synthetic strategy lies in the precise control of the condensation reaction between the substituted ethyl acetate and diethyl oxalate, facilitated by strong alkaline materials such as sodium tert-butoxide or potassium tert-butoxide. In this initial phase, the base deprotonates the alpha-carbon of the substituted ethyl acetate, generating a nucleophilic enolate that attacks the electrophilic carbonyl carbon of the diethyl oxalate. 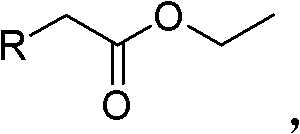 This Claisen-type condensation proceeds efficiently in ether solvents like THF or MTBE at temperatures maintained between 20°C and 35°C, ensuring high selectivity for the formation of the 2-substituted-3-oxo diethyl succinate intermediate while minimizing side reactions such as self-condensation of the ester. The subsequent workup involves careful pH adjustment to acidic conditions (pH 1-3) using hydrochloric acid, followed by silica gel treatment to adsorb tarry byproducts, which is a critical step for maintaining the high purity profile required for downstream applications. This meticulous control over the reaction environment prevents the formation of polymeric impurities that often plague ester condensations, thereby securing a clean intermediate stream for the subsequent oxidation step.
This Claisen-type condensation proceeds efficiently in ether solvents like THF or MTBE at temperatures maintained between 20°C and 35°C, ensuring high selectivity for the formation of the 2-substituted-3-oxo diethyl succinate intermediate while minimizing side reactions such as self-condensation of the ester. The subsequent workup involves careful pH adjustment to acidic conditions (pH 1-3) using hydrochloric acid, followed by silica gel treatment to adsorb tarry byproducts, which is a critical step for maintaining the high purity profile required for downstream applications. This meticulous control over the reaction environment prevents the formation of polymeric impurities that often plague ester condensations, thereby securing a clean intermediate stream for the subsequent oxidation step.
The second mechanistic pillar of this process is the oxidative decarboxylation of the succinate intermediate, which is achieved using potent oxidants like Potassium Persulfate or Sodium Persulfate in the presence of an acid solvent. The reaction mechanism involves the generation of sulfate radical anions that facilitate the cleavage of the carbon-carbon bond adjacent to the ketone functionality, effectively excising the extra carbonyl group introduced during the condensation phase. This transformation occurs in a biphasic system involving water and organic solubility promoters like ethanol or acetone, which ensure adequate mixing and mass transfer between the inorganic oxidant and the organic substrate. Temperature control is maintained strictly between 25°C and 45°C to manage the exothermic nature of the oxidation while preventing thermal degradation of the sensitive malonate product. The final isolation via ice-water separation and centrifugation allows for the precipitation of the product in high purity, with HPLC data from the patent examples confirming purity levels consistently exceeding 90%, demonstrating the efficacy of this mechanistic design in controlling the impurity profile throughout the synthesis.
How to Synthesize Alpha-Substituted Diethyl Malonate Efficiently
Implementing this synthesis route requires strict adherence to the stoichiometric ratios and temperature profiles outlined in the patent to ensure optimal yield and purity. The process begins with the preparation of the reaction vessel with the appropriate ether solvent and base, followed by the controlled addition of the diethyl oxalate and substituted acetate to manage the exotherm of the condensation. Following the isolation of the succinate intermediate, the oxidation step demands precise dosing of the persulfate oxidant and acid to drive the cleavage to completion without over-oxidizing the product. For a comprehensive understanding of the specific operational parameters, reagent grades, and workup procedures required to replicate this high-efficiency pathway, please refer to the standardized synthesis guide below.
- Perform condensation of 2-substituted acetic acid ethyl ester with diethyl oxalate in ether solvent using alkaline material at 20-35°C.
- Treat the intermediate 2-substituted-3-oxo diethyl succinate with acid solvent and oxidant like Potassium Persulfate at 25-45°C.
- Isolate the final alpha-substituted diethyl malonate derivative through extraction, washing, and rectification after ice-water separation.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this patented synthesis route offers profound advantages in terms of cost structure and supply chain resilience for manufacturers of fine chemical intermediates. By replacing toxic cyanide salts and expensive heavy metal catalysts with commodity chemicals like diethyl oxalate and persulfates, the raw material cost base is significantly stabilized and reduced. The elimination of hazardous reagents also removes the substantial overhead costs associated with specialized storage, handling protocols, and the disposal of toxic waste streams, which can often account for a significant portion of the total manufacturing cost in traditional processes. Furthermore, the simplicity of the operation, which does not require high-pressure reactors or extreme temperatures, lowers the barrier to entry for contract manufacturing organizations (CDMOs), thereby increasing the number of qualified suppliers in the market and enhancing competition. This diversification of the supply base is crucial for mitigating the risk of supply disruptions and ensuring long-term availability of these critical building blocks for the global pharmaceutical industry.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of high-cost, high-risk reagents with inexpensive, widely available commodity chemicals. The avoidance of sodium cyanide eliminates the need for expensive detoxification units and rigorous safety monitoring systems, leading to substantial operational expenditure savings. Additionally, the high conversion rates and purity levels achieved reduce the need for extensive downstream purification steps such as repeated recrystallizations or complex chromatographic separations, further driving down the cost of goods sold (COGS). The use of common solvents like THF and ethanol, which can often be recovered and recycled within the plant, adds another layer of cost efficiency that accumulates over large-scale production campaigns.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials such as substituted ethyl acetates and diethyl oxalate ensures a robust and resilient supply chain that is less susceptible to the bottlenecks often seen with specialty reagents. These feedstocks are produced by multiple global chemical manufacturers, reducing the dependency on single-source suppliers and minimizing the risk of raw material shortages. The mild reaction conditions also mean that the process can be executed in a wide range of standard chemical manufacturing facilities without the need for specialized infrastructure, allowing for greater flexibility in sourcing production capacity. This adaptability is essential for maintaining continuous supply to downstream API manufacturers, especially in times of market volatility or unexpected demand surges.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, as evidenced by the successful demonstration in 1000L reactors within the patent examples, proving its viability for multi-ton production campaigns. The absence of heavy metals and cyanides simplifies the environmental compliance landscape, making it easier to obtain necessary permits and maintain good standing with regulatory bodies. The waste streams generated are primarily aqueous salts and organic solvents, which are far easier to treat and manage than the hazardous sludge produced by cyanide-based routes. This environmental friendliness aligns perfectly with the increasing corporate sustainability goals of major pharmaceutical companies, making suppliers who adopt this technology more attractive partners for long-term contracts.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this alpha-substituted diethyl malonate synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the underlying patent documentation, providing a factual basis for evaluating the feasibility of this route for your specific production needs. Understanding these details is crucial for R&D and procurement teams when assessing the potential for technology transfer or outsourcing partnerships.
Q: How does this method improve safety compared to traditional cyanide routes?
A: This patented process eliminates the use of highly toxic sodium cyanide and heavy metal catalysts like dibutyltin oxide, significantly reducing environmental hazards and operator risk while simplifying waste treatment protocols.
Q: What is the expected purity level for the final derivatives?
A: The method consistently achieves liquid chromatography purity (HPLC) stabilized above 90.0%, with specific examples demonstrating purity levels reaching 91.6% for fluoro-derivatives without requiring complex purification trains.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly describes successful execution in 1000L reaction vessels with stable temperature control between 20-45°C, utilizing commercially available raw materials that support continuous and scalable manufacturing operations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Substituted Diethyl Malonate Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced, safe, and efficient synthetic routes to meet the evolving demands of the global pharmaceutical market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and robust. We are committed to delivering high-purity intermediates that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. By leveraging technologies like the one described in CN102531897A, we can offer our partners a secure supply of complex intermediates that are both cost-effective and environmentally responsible.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis route to your specific volume and quality requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this greener methodology. We encourage you to contact us today to索取 specific COA data and route feasibility assessments, allowing you to make informed decisions that will optimize your supply chain and accelerate your drug development timelines.
