Advanced Copper-Free Synthesis of Phenylethynyl Pyridine Derivatives for Commercial Scale
Advanced Copper-Free Synthesis of Phenylethynyl Pyridine Derivatives for Commercial Scale
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex heterocyclic scaffolds, particularly phenylethynyl pyridine derivatives which serve as critical building blocks for bioactive molecules and advanced materials. Patent CN108794385B introduces a transformative approach to this synthesis, leveraging a palladium-catalyzed Sonogashira coupling that fundamentally alters the operational landscape for manufacturers. This method utilizes palladium acetate in conjunction with the specialized Xantphos ligand and cesium carbonate in a methanol medium, achieving high conversion rates under remarkably mild conditions. By operating effectively at temperatures between 50°C and 60°C without the necessity for inert gas protection, this technology addresses long-standing inefficiencies in traditional cross-coupling reactions. For R&D directors and procurement leaders, this represents a significant opportunity to optimize production workflows while maintaining stringent quality standards for high-purity intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of phenylethynyl pyridine derivatives has relied heavily on traditional Sonogashira coupling protocols that impose significant operational burdens on manufacturing facilities. Conventional methods typically necessitate the use of copper salts as co-catalysts, which introduces complex downstream purification challenges to remove residual heavy metals to meet pharmaceutical grade specifications. Furthermore, these legacy processes often demand strictly anhydrous and anaerobic conditions, requiring expensive solvent drying systems and continuous nitrogen or argon purging throughout the reaction duration. The reliance on polar aprotic solvents such as DMF or DMAc in older methods further complicates waste management and solvent recovery, driving up both environmental compliance costs and overall production expenses. Additionally, the sensitivity of traditional catalyst systems to oxygen and moisture often leads to inconsistent batch-to-batch reproducibility, creating supply chain vulnerabilities for companies relying on these intermediates for critical drug synthesis pipelines.
The Novel Approach
The innovative methodology disclosed in the patent data offers a robust alternative that circumvents these historical bottlenecks through a carefully engineered catalyst system. By employing a palladium acetate and Xantphos ligand combination, the reaction achieves high efficiency without the need for copper co-catalysts, thereby eliminating a major source of metallic impurities from the outset. The use of methanol as a solvent is particularly advantageous, as it is a commodity chemical that is easier to handle, recover, and dispose of compared to traditional high-boiling polar solvents. Perhaps most critically, this system demonstrates remarkable tolerance to air and moisture, allowing reactions to proceed under ambient conditions without the need for rigorous degassing or inert atmosphere protection. This simplification of reaction conditions not only reduces capital expenditure on specialized equipment but also significantly shortens the operational cycle time, enhancing the overall throughput of the manufacturing facility.
Mechanistic Insights into Pd-Catalyzed Sonogashira Coupling
The core of this technological advancement lies in the specific interaction between the palladium center and the Xantphos ligand, which creates a highly active catalytic species capable of facilitating the cross-coupling of 2-iodopyridine and phenylacetylene derivatives. The wide bite angle of the Xantphos ligand stabilizes the palladium complex, promoting the oxidative addition and reductive elimination steps essential for the catalytic cycle while preventing catalyst deactivation. This stability allows the reaction to proceed efficiently at moderate temperatures of 50-60°C, reducing thermal stress on the substrates and minimizing the formation of thermal degradation byproducts. The absence of copper eliminates the competing Glaser homocoupling side reaction, which is a common impurity profile issue in traditional Sonogashira protocols, thus ensuring a cleaner crude reaction profile. This mechanistic precision translates directly to higher isolated yields and reduced purification burden, which is a key metric for R&D teams evaluating process viability.
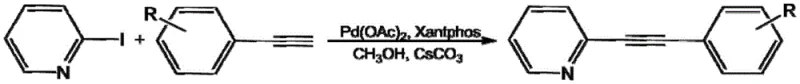
Impurity control is further enhanced by the choice of cesium carbonate as the base, which provides a mild alkaline environment sufficient to activate the terminal alkyne without promoting excessive decomposition of sensitive functional groups. The reaction mechanism tolerates a variety of substituents on the phenylacetylene ring, including electron-withdrawing nitro groups and electron-donating methoxy groups, demonstrating broad substrate applicability. This versatility is crucial for medicinal chemistry campaigns where diverse analog libraries need to be synthesized rapidly. The robust nature of the catalytic cycle ensures that even with slight variations in reagent quality or environmental conditions, the reaction maintains high conversion rates. For quality assurance teams, this means a more consistent impurity profile across different production batches, simplifying the validation process for regulatory filings and ensuring a reliable supply of high-purity phenylethynyl pyridine intermediates for downstream applications.
How to Synthesize Phenylethynyl Pyridine Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting involves a straightforward procedure that aligns with standard operational protocols while offering superior performance metrics. The process begins with the precise weighing of palladium acetate and Xantphos ligand, which are then introduced to a reaction vessel containing the substrate 2-iodopyridine and the specific phenylacetylene derivative. Methanol is added as the reaction medium, followed by the introduction of cesium carbonate to initiate the catalytic cycle under ambient air conditions. The mixture is then heated to a controlled temperature range of 50-60°C and maintained for a period of 6 to 10 hours depending on the specific electronic nature of the substituents. Upon completion, the reaction mixture is cooled to room temperature and subjected to standard workup procedures, typically involving column chromatography to isolate the target phenylethynyl pyridine derivative with high purity.
- Prepare the reaction mixture by combining 2-iodopyridine and phenylacetylene derivative in methanol solvent with cesium carbonate base.
- Add palladium acetate catalyst and Xantphos ligand to the mixture under ambient air conditions without inert gas protection.
- Heat the reaction to 50-60°C for 6-10 hours, then purify the crude product via column chromatography to obtain high-purity derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers substantial strategic advantages for procurement managers and supply chain heads looking to optimize their sourcing strategies for heterocyclic intermediates. The elimination of copper co-catalysts and the use of mild reaction conditions directly translate to a simplified manufacturing process that requires less specialized infrastructure and lower operational overhead. The ability to run reactions in air without strict moisture control reduces the complexity of the production environment, allowing for greater flexibility in facility utilization and scheduling. Furthermore, the use of methanol as a solvent significantly lowers raw material costs and simplifies solvent recovery systems, contributing to a more sustainable and cost-effective production model. These factors combined create a more resilient supply chain capable of responding quickly to market demands without compromising on quality or compliance standards.
- Cost Reduction in Manufacturing: The removal of copper salts from the reaction formulation eliminates the need for expensive and time-consuming heavy metal scavenging steps during downstream processing. This reduction in purification complexity leads to significant savings in consumables such as silica gel and scavenger resins, while also reducing the volume of hazardous waste generated. Additionally, the lower energy requirements due to mild heating conditions (50-60°C) compared to traditional high-temperature reflux methods result in reduced utility costs over the lifecycle of the product. The overall process efficiency allows for higher throughput per batch, effectively lowering the cost of goods sold and improving margin potential for commercial scale-up of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The robustness of this catalytic system against air and moisture ensures that production is less susceptible to interruptions caused by equipment failure in inert gas systems or solvent drying units. This reliability is critical for maintaining continuous supply lines to downstream customers who depend on just-in-time delivery models. The use of widely available commodity chemicals like methanol and cesium carbonate reduces the risk of raw material shortages that can plague supply chains reliant on specialized or niche reagents. Consequently, manufacturers can offer more stable lead times and pricing structures, strengthening their position as a reliable phenylethynyl pyridine supplier in the global market.
- Scalability and Environmental Compliance: Scaling this reaction from laboratory to industrial production is facilitated by the simple equipment requirements and the safety profile of the solvents and reagents involved. Methanol is easier to handle and recycle on a large scale compared to high-boiling amide solvents, reducing the environmental footprint of the manufacturing process. The absence of copper waste simplifies effluent treatment protocols, ensuring easier compliance with increasingly stringent environmental regulations. This scalability ensures that the process can meet the growing demand for high-purity pyridine derivatives in the pharmaceutical and electronic materials sectors without requiring massive capital investment in new reactor infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this synthesis technology. These answers are derived directly from the patent specifications and are intended to provide clarity for technical teams evaluating the feasibility of this route for their specific applications. Understanding these details is essential for making informed decisions about process adoption and supply chain integration.
Q: Does this synthesis method require copper co-catalysts?
A: No, this patented method eliminates the need for copper salts, significantly reducing heavy metal impurity risks and simplifying downstream purification processes.
Q: What are the reaction conditions regarding moisture and oxygen?
A: The process is robust under ambient air conditions and uses methanol without strict dehydration, removing the need for expensive inert gas protection.
Q: Is this method suitable for large-scale manufacturing?
A: Yes, the mild temperatures (50-60°C) and use of commodity solvents like methanol make it highly scalable and cost-effective for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenylethynyl Pyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes in the development of next-generation pharmaceuticals and fine chemicals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN108794385B can be successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of phenylethynyl pyridine derivative meets the exacting standards required by global regulatory bodies. Our commitment to technical excellence allows us to offer customized solutions that align with your specific project timelines and quality requirements.
We invite you to collaborate with us to leverage this advanced technology for your upcoming projects. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis to understand how this copper-free route can optimize your specific manufacturing budget. We encourage potential partners to contact us to obtain specific COA data and route feasibility assessments tailored to your target molecules. Let us help you reduce lead time for high-purity pyridine derivatives and secure a competitive advantage in your market sector through superior chemical manufacturing solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
