Scaling Vitamin A Intermediates: Continuous Flow Synthesis of 4-Bromo-3-Methyl-2-Buten-1-Ol Acetate
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for synthesizing critical vitamin precursors, and patent CN110240543B presents a significant advancement in the production of 4-bromo-3-methyl-2-buten-1-ol acetate. This compound serves as a pivotal intermediate in the industrial synthesis of Vitamin A and its derivatives, making its efficient manufacture a priority for global supply chains. The disclosed technology shifts away from traditional batch processing towards a sophisticated continuous flow system, utilizing tubular reactors to enhance safety and throughput. By integrating bromohydrination and subsequent esterification-rearrangement into a seamless pipeline operation, the method addresses longstanding issues regarding raw material waste and hazardous handling of volatile reagents like isoprene. This technical evolution not only promises higher purity profiles but also establishes a foundation for reliable agrochemical intermediate supplier capabilities, ensuring that downstream manufacturers receive consistent quality materials essential for complex multistep syntheses.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this key intermediate relied on batch processes described in older literature, such as US4175204, which involved adding isoprene to glacial acetic acid followed by the batch-wise addition of N-bromosuccinimide. This conventional approach suffered from severe inefficiencies, including the requirement for a large excess of isoprene to drive the reaction, leading to substantial raw material waste and increased separation burdens. Furthermore, the batch addition of brominating agents to flammable isoprene created significant safety hazards, lowering the safe operation coefficient and necessitating rigorous containment measures. Post-reaction processing was equally cumbersome, requiring multiple extraction, washing, and drying steps with organic solvents like dichloromethane, generating large volumes of wastewater and complicating the recovery of the product. Additionally, the formation of the unwanted 1,2-addition isomer required a separate, distinct rearrangement step using liquid acids, further elongating the production cycle and depressing the overall yield to approximately 78%.
The Novel Approach
In stark contrast, the novel methodology outlined in the patent employs a continuous pumping strategy where a mixture of isoprene and water is introduced directly into a reactor pre-loaded with N-bromosuccinimide. This continuous bromoalcoholation reaction occurs under controlled temperatures between 0°C and 10°C, allowing for precise thermal management that mitigates the risks associated with exothermic reactions. The resulting mixture is rectified to isolate the bromohydrin intermediates, while the succinimide byproduct remains trapped within the pipeline, effectively eliminating a major solid waste stream. Subsequently, the purified intermediates are continuously pumped with acetic anhydride into a second reactor containing a solid acid catalyst, where esterification and rearrangement occur simultaneously. This integrated approach not only simplifies the workflow by removing the need for separate rearrangement stages but also elevates the total yield to over 85%, demonstrating a clear pathway for cost reduction in electronic chemical manufacturing and related high-value sectors.
Mechanistic Insights into Solid Acid Catalyzed Esterification and Rearrangement
The core chemical innovation lies in the dual-function capability of the solid acid catalyst, typically a strong acid cation exchange resin such as type 732 or D001, which facilitates both esterification and allylic rearrangement. In the second reactor, maintained at temperatures between 80°C and 100°C, the hydroxyl group of the bromohydrin intermediate reacts with acetic anhydride to form the acetate ester. Crucially, the acidic sites on the resin also promote the migration of the double bond and the bromine atom, converting the thermodynamically less stable 1,2-addition isomer into the desired conjugated product. 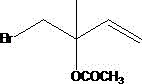 This mechanistic pathway ensures that isomeric impurities, which would otherwise require difficult chromatographic separation or result in yield loss, are chemically transformed into the target molecule. The use of a heterogeneous catalyst allows the reaction to proceed without the contamination of liquid acid residues, thereby simplifying the downstream purification process and enhancing the final purity of the high-purity OLED material or pharmaceutical intermediate.
This mechanistic pathway ensures that isomeric impurities, which would otherwise require difficult chromatographic separation or result in yield loss, are chemically transformed into the target molecule. The use of a heterogeneous catalyst allows the reaction to proceed without the contamination of liquid acid residues, thereby simplifying the downstream purification process and enhancing the final purity of the high-purity OLED material or pharmaceutical intermediate.
Furthermore, the continuous flow architecture inherently improves mass and heat transfer characteristics compared to static batch vessels. The tubular reactor design ensures that the residence time distribution is narrow, meaning all molecules experience nearly identical reaction conditions, which minimizes the formation of side products caused by localized hot spots or concentration gradients. This uniformity is critical for maintaining a clean impurity profile, as it prevents the over-reaction or degradation of the sensitive allylic bromide functionality. By controlling the pumping flow rates precisely, operators can tune the residence time to optimize the conversion of the starting materials while ensuring complete consumption of the reactive intermediates. This level of process control is essential for commercial scale-up of complex polymer additives and fine chemicals, where reproducibility and batch-to-batch consistency are paramount for regulatory compliance and customer satisfaction.
How to Synthesize 4-Bromo-3-Methyl-2-Buten-1-Ol Acetate Efficiently
To implement this synthesis effectively, one must adhere to the specific parameters defined in the patent examples, which demonstrate the robustness of the continuous flow setup. The process begins with the careful preparation of the isoprene and water mixture, which is then metered into the first tubular reactor containing solid N-bromosuccinimide particles. Following the initial bromohydrination, the crude stream undergoes rectification to separate the volatile bromohydrins from the non-volatile succinimide, which stays behind in the equipment. The detailed standardized synthesis steps see the guide below for exact operational protocols.
- Continuously pump a mixture of isoprene and water into a tubular reactor containing N-bromosuccinimide (NBS) at 0-10°C to perform bromohydrination.
- Rectify the crude mixture to isolate the bromohydrin intermediates, removing succinimide byproducts retained in the pipeline.
- Pump the purified bromohydrins with acetic anhydride into a second tubular reactor packed with solid acid resin (e.g., D001) at 80-100°C for simultaneous esterification and rearrangement.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from batch to continuous flow chemistry represented in this patent offers profound strategic benefits beyond mere technical elegance. The elimination of liquid acid catalysts and the retention of solid byproducts within the reactor pipeline drastically simplify the waste treatment infrastructure required at the manufacturing site. This reduction in effluent complexity translates directly into lower environmental compliance costs and reduced downtime for cleaning and maintenance between production runs. Moreover, the ability to run the process continuously rather than in discrete batches allows for a more consistent output rate, smoothing out inventory fluctuations and enabling just-in-time delivery models that are highly valued by downstream pharmaceutical manufacturers.
- Cost Reduction in Manufacturing: The substitution of traditional liquid acids with reusable solid acid resins eliminates the need for neutralization steps and the associated salt waste disposal costs. By integrating the esterification and rearrangement into a single unit operation, the process reduces energy consumption and labor hours required for multiple reaction setups and workups. The higher overall yield of over 85% compared to the historical 78% means that less raw material is required per kilogram of finished product, significantly lowering the cost of goods sold. Additionally, the recovery of unreacted acetic anhydride and acetic acid via distillation allows these valuable reagents to be recycled back into the process, further enhancing the economic efficiency of the manufacturing campaign.
- Enhanced Supply Chain Reliability: The continuous nature of the process, utilizing tubular reactors and metering pumps, ensures a steady and predictable production rate that is less susceptible to the variability inherent in batch processing. The use of readily available starting materials like isoprene and acetic anhydride, combined with the robustness of the solid acid catalyst, minimizes the risk of supply disruptions caused by specialized reagent shortages. The simplified post-treatment process, which avoids complex extractions and drying steps, reduces the turnaround time for each production cycle, allowing the facility to respond more agilely to sudden increases in market demand. This reliability is crucial for maintaining the continuity of supply for critical vitamin intermediates that feed into high-volume global supply chains.
- Scalability and Environmental Compliance: Scaling up this process is inherently safer and more straightforward because it relies on numbering up tubular reactors or increasing their length rather than increasing the volume of a single large vessel, which often introduces heat transfer limitations. The containment of hazardous reagents like isoprene within a closed piping system significantly reduces the risk of fugitive emissions and exposure to personnel, aligning with stringent modern safety regulations. The reduction in wastewater generation, due to the absence of aqueous washes and neutralization steps, lowers the burden on wastewater treatment facilities and supports sustainability goals. This environmentally friendly profile makes the technology attractive for production in regions with strict environmental enforcement, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this continuous flow technology for producing 4-bromo-3-methyl-2-buten-1-ol acetate. These answers are derived directly from the experimental data and technical disclosures found within the patent documentation, providing clarity on process capabilities and advantages. Understanding these details is essential for evaluating the feasibility of adopting this method for large-scale commercial production.
Q: How does the continuous flow method improve safety compared to batch processing?
A: The continuous flow method eliminates the need for large batches of volatile isoprene and avoids the hazardous batch-wise addition of N-bromosuccinimide, significantly reducing explosion risks and improving operational safety coefficients.
Q: What is the advantage of using a solid acid catalyst in this synthesis?
A: Using a solid acid cation exchange resin replaces traditional liquid acids like p-toluenesulfonic acid, allowing the catalyst to remain fixed in the reactor pipeline. This eliminates neutralization steps, reduces wastewater generation, and simplifies product isolation.
Q: Can this process handle the rearrangement of isomeric byproducts?
A: Yes, the process integrates esterification and rearrangement in a single step. The solid acid catalyst effectively converts the unwanted 1,2-addition isomer into the desired 4-bromo-3-methyl-2-buten-1-ol acetate, boosting overall yield above 85%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Bromo-3-Methyl-2-Buten-1-Ol Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful synthesis of complex active ingredients. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 4-bromo-3-methyl-2-buten-1-ol acetate performs consistently in your downstream reactions. Our expertise in continuous flow chemistry allows us to optimize these advanced manufacturing techniques for maximum yield and safety.
We invite you to collaborate with us to leverage these technological advancements for your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how implementing this continuous flow route can reduce your overall manufacturing expenses. Please contact our technical procurement team to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring a partnership built on transparency and technical excellence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
