Advanced Grignard Coupling for Rosuvastatin Calcium Intermediates: A Safer Industrial Scale-Up
Advanced Grignard Coupling for Rosuvastatin Calcium Intermediates: A Safer Industrial Scale-Up
The global demand for lipid-lowering agents continues to surge, driven by the increasing prevalence of hyperlipidemia and cardiovascular diseases worldwide. At the heart of this therapeutic class lies Rosuvastatin calcium, a potent statin whose market value has historically peaked at billions of dollars. The efficient and safe production of its key chiral side chain, specifically (4R,6R)-6-[(5-methyl-1,3,4-thiadiazole-2-yl)sulfonyl]methyl-2,2-dimethyl-1,3-dioxane-4-tert-butyl acetate, remains a critical bottleneck for generic manufacturers. Patent CN113387944B, published in late 2021, introduces a groundbreaking synthetic methodology that fundamentally shifts the paradigm from hazardous oxidation chemistry to a controlled organometallic coupling process. This technical insight report analyzes the proprietary data within this patent to demonstrate how this novel route offers a robust solution for reliable pharmaceutical intermediates supplier networks seeking to optimize their API manufacturing pipelines.
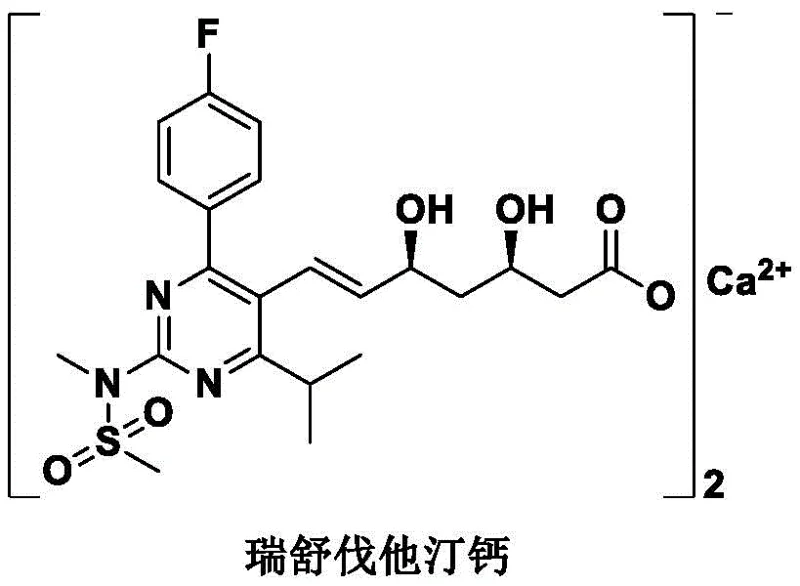
Rosuvastatin calcium represents a complex molecular architecture where the stereochemical integrity of the side chain is paramount for biological efficacy. The molecule features a fluorophenyl group, a pyrimidine nucleus, and a critical chiral dihydroxyheptenoic acid side chain linked via a sulfone bridge. As illustrated in the structural diagram, the sulfone linkage connects the heterocyclic core to the chiral lactone precursor. Traditional syntheses often struggle with the construction of this sulfone bond, frequently resorting to multi-step sequences that compromise overall yield and introduce significant safety liabilities. The patent CN113387944B addresses these challenges by focusing on the synthesis of the sulfone-containing side chain intermediate (Formula I), which serves as the pivotal building block for the subsequent Julia olefination that constructs the final drug substance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those disclosed in patents CN112028881 and CN111875538, rely heavily on a two-step sequence involving nucleophilic substitution followed by oxidation. In the first step, a chloro-acetate precursor (Compound D-4) undergoes an SN2 reaction with a thiadiazole thiol. This transformation is notoriously inefficient, requiring harsh reaction conditions with temperatures ranging from 100°C to 130°C to overcome the poor leaving group ability of the chlorine atom. Such high thermal energy input inevitably leads to substrate degradation, product carbonization, and the formation of dark-colored impurities that necessitate extensive decolorization and recrystallization steps. Furthermore, this process typically employs high-boiling polar aprotic solvents like dimethyl sulfoxide (DMSO) or N,N-dimethylformamide (DMF), which are energetically expensive to recover and contribute significantly to industrial waste streams.
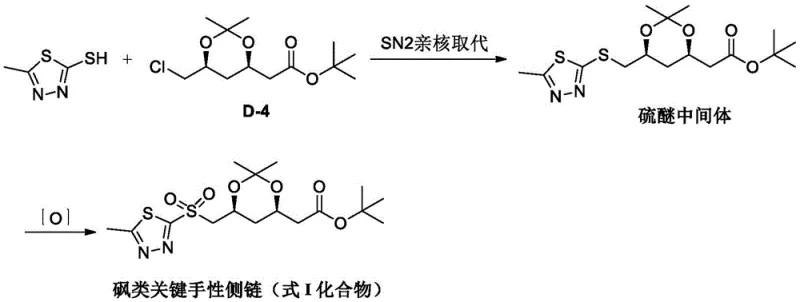
The second critical flaw in the conventional workflow is the oxidation step. To convert the resulting thioether into the required sulfone, the prior art utilizes peroxide oxidants. This specific chemical transformation is classified under the 18 dangerous supervision processes in chemical safety regulations due to the inherent instability of peroxy bonds (-O-O-). The bond energy of peroxides is low (approximately 80-160 kJ/mol), making them highly sensitive to heat, friction, and impact, posing a severe risk of explosive decomposition on a commercial scale. Additionally, the oxidative conditions often promote hydrolysis of the sensitive ester and acetonide protecting groups, leading to a complex impurity profile that includes sulfoxides and hydrolyzed by-products, thereby complicating downstream purification and reducing the overall viability of the process for cost reduction in API manufacturing.
The Novel Approach
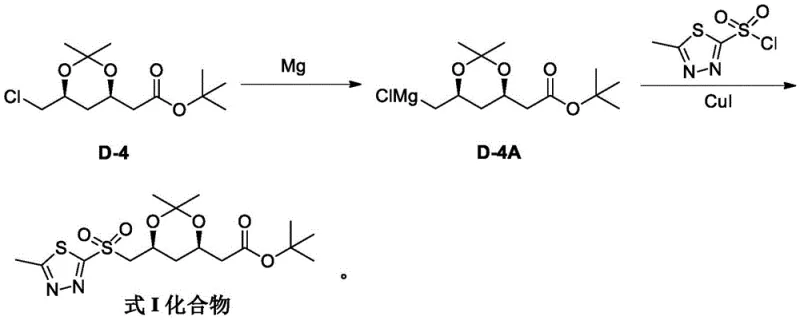
In stark contrast to the hazardous oxidation pathway, the invention detailed in CN113387944B proposes a direct construction of the sulfone bond via a Grignard-type coupling reaction. This innovative strategy bypasses the unstable thioether intermediate entirely. Instead, it involves the in situ generation of an organomagnesium species (Compound D-4A) from the chloro-acetate precursor using metallic magnesium. This organometallic reagent is then reacted directly with 5-methyl-1,3,4-thiadiazole-2-sulfonyl chloride. By shifting the synthetic logic from substitution-oxidation to direct metal-halogen exchange and coupling, the process eliminates the need for dangerous peroxide reagents and high-temperature SN2 conditions. The reaction proceeds under significantly milder thermal conditions, with the Grignard formation occurring at reflux (60-70°C) and the coupling step conducted at cryogenic temperatures (-30°C to -40°C), ensuring exceptional control over reaction selectivity and safety.
Mechanistic Insights into CuI-Catalyzed Grignard Coupling
The core mechanistic breakthrough of this patent lies in the stabilization and selective reactivity of the organomagnesium intermediate. When Compound D-4 reacts with magnesium turnings in an ether solvent system (such as tetrahydrofuran or a THF/MTBE mixture), it forms the Grignard reagent D-4A. However, organomagnesium compounds are prone to homocoupling (Wurtz-type coupling), where two equivalents of the Grignard reagent react with each other to form a dimer impurity. The patent data explicitly highlights that without intervention, this self-coupling dominates the reaction profile, drastically reducing the yield of the desired sulfone. The inventors discovered that the addition of a catalytic amount of Copper(I) Iodide (CuI), specifically at a loading of 0.03 to 0.05 molar equivalents, acts as a crucial transmetallation mediator. The copper salt facilitates the cross-coupling between the alkyl-magnesium species and the sulfonyl chloride electrophile, effectively suppressing the formation of the dimer by-product and channeling the reaction flux towards the target sulfone.
This mechanistic refinement has profound implications for impurity control. In the conventional peroxide oxidation route, the acidic environment and radical nature of the oxidation often lead to the cleavage of the acetonide protecting group, generating hydrolyzed impurities that are structurally similar to the product and difficult to separate. In the new Grignard protocol, the reaction environment is strictly anhydrous during the coupling phase, preserving the integrity of the acid-sensitive acetonide moiety. Furthermore, the absence of over-oxidation risks means that sulfoxide impurities (partial oxidation products) are virtually non-existent. The result is a crude product profile that is remarkably clean, often requiring only a simple pulping procedure with methanol and water to achieve purity levels exceeding 99%, as evidenced by the HPLC data in the patent examples. This level of purity is essential for meeting the stringent specifications required by regulatory bodies for high-purity pharmaceutical intermediates.
How to Synthesize Rosuvastatin Calcium Intermediate Efficiently
The implementation of this Grignard-based strategy offers a streamlined, one-pot operational protocol that is highly amenable to industrial scale-up. The process begins with the activation of magnesium metal in a dry ether solvent, followed by the slow addition of the chloro-acetate starting material to generate the organometallic species. Once the formation of the Grignard reagent is complete, indicated by the consumption of magnesium, the reaction mixture is cooled to sub-zero temperatures to minimize thermal side reactions. The sulfonyl chloride electrophile is then introduced in the presence of the copper catalyst. This sequence allows for the direct telescoping of steps without the isolation of unstable intermediates, maximizing throughput and minimizing equipment occupancy time. For a detailed breakdown of the specific stoichiometric ratios, temperature ramps, and workup procedures validated in the patent examples, please refer to the standardized synthesis guide below.
- Formation of Organomagnesium Compound: React compound D-4 with magnesium powder (1.2-1.3 eq) in THF or THF/MTBE at reflux (60-70°C) to generate the Grignard reagent D-4A.
- CuI Catalyzed Coupling: Cool the reaction mixture to -30°C to -40°C, add catalytic CuI (0.03-0.05 eq), and slowly add 5-methyl-1,3,4-thiadiazole-2-sulfonyl chloride.
- Quenching and Isolation: Warm to 0-10°C, quench with water, extract with ethyl acetate, and purify via methanol/water pulping to obtain the high-purity white powder intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from the legacy peroxide oxidation route to this novel Grignard coupling method represents a strategic opportunity to de-risk the supply of critical statin intermediates. The elimination of peroxide reagents removes a major regulatory and safety hurdle, simplifying the permitting process for manufacturing facilities and reducing insurance premiums associated with handling explosive materials. Moreover, the shift from high-boiling solvents like DMSO to volatile ethers like THF and MTBE dramatically improves the efficiency of solvent recovery systems. Ethers can be distilled and recycled with significantly lower energy input compared to the energy-intensive stripping required for DMSO, leading to substantial reductions in utility costs and waste disposal fees. This solvent optimization directly contributes to cost reduction in pharmaceutical intermediates manufacturing by lowering the variable cost per kilogram of the produced intermediate.
- Cost Reduction in Manufacturing: The new process eliminates the need for expensive phase transfer catalysts and stoichiometric oxidants, replacing them with catalytic amounts of copper iodide and inexpensive magnesium metal. By avoiding the high-temperature degradation of substrates, the process achieves significantly higher yields, meaning less raw material is wasted to produce the same amount of product. The simplified purification process, which replaces complex recrystallizations with simple pulping, further reduces labor and processing time, driving down the overall cost of goods sold (COGS) without compromising quality.
- Enhanced Supply Chain Reliability: The reliance on hazardous peroxides in the old method often creates bottlenecks due to strict transportation and storage regulations. By removing these controlled substances from the bill of materials, the supply chain becomes more resilient and less susceptible to regulatory delays. Additionally, the milder reaction conditions reduce wear and tear on reactor vessels and cooling systems, enhancing equipment longevity and minimizing unplanned downtime. This operational stability ensures a consistent and reliable flow of high-purity rosuvastatin intermediates to downstream API manufacturers, securing the continuity of the final drug supply.
- Scalability and Environmental Compliance: The one-pot nature of the Grignard coupling reduces the number of unit operations, shrinking the physical footprint required for production. The use of recyclable ether solvents aligns with green chemistry principles by minimizing the discharge of persistent organic pollutants. The absence of toxic tributylamine by-products (generated from the degradation of phase transfer catalysts in the old method) simplifies wastewater treatment protocols. These factors collectively make the process highly scalable, allowing manufacturers to ramp up production from pilot batches to commercial scale-up of complex pharmaceutical intermediates with confidence in meeting environmental, health, and safety (EHS) standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in patent CN113387944B, offering clarity on how this technology outperforms legacy methods in terms of safety, purity, and operational efficiency. Understanding these distinctions is vital for technical teams evaluating process transfers.
Q: Why is the Grignard route safer than the traditional SN2 oxidation method?
A: The traditional method requires hazardous peroxide oxidation, classified as one of the 18 dangerous supervision processes due to explosion risks. The new Grignard route eliminates peroxides entirely and operates at much milder temperatures (60-70°C vs 100-130°C), significantly reducing EHS risks.
Q: How does the new process improve impurity control?
A: By avoiding high-temperature SN2 conditions and acidic peroxide oxidation, the new method prevents the formation of sulfoxide impurities and hydrolysis by-products. The addition of CuI specifically suppresses the self-coupling dimerization of the Grignard reagent, ensuring higher chemical purity.
Q: What are the solvent advantages of this new synthetic route?
A: Unlike the conventional process which relies on high-boiling solvents like DMSO or DMF that are difficult to recover, this method utilizes THF and MTBE. These ether solvents are easily recyclable, drastically reducing three-waste discharge and lowering overall production costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rosuvastatin Calcium Intermediate Supplier
The technological advancements described in patent CN113387944B underscore the critical importance of adopting safer, more efficient synthetic routes in the competitive landscape of cardiovascular drug manufacturing. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle sensitive organometallic chemistries under strict inert atmospheres, ensuring that the delicate Grignard coupling steps are executed with precision. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced HPLC and chiral analysis to guarantee that every batch of intermediate meets the exacting standards required for global regulatory filings.
We invite pharmaceutical partners to collaborate with us to unlock the full potential of this optimized synthesis. By integrating this safer Grignard-based route into your supply chain, you can achieve significant operational efficiencies and risk mitigation. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data from our pilot runs and comprehensive route feasibility assessments to support your next-generation Rosuvastatin calcium projects, ensuring a seamless transition from development to commercial supply.
